COMECAUSE IN-Y400 High-Precision Antibiotic Zone-of-Inhibition Analyzer
| Brand | COMECAUSE |
|---|---|
| Model | IN-Y400 |
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Category | Domestic Instrument |
| Price | USD 10,000 (FOB) |
| Camera Resolution | 20 MP color CMOS sensor |
| Lens | 12 mm focal length, high-definition macro lens |
| Petri Dish Compatibility | 90–110 mm standard diameter |
| Illumination | Dual independent LED lighting system (top & bottom), 360° quad-color visible-light ring + 254 nm UV sterilization lamp |
| Brightness Control | Software-adjustable in 1% increments |
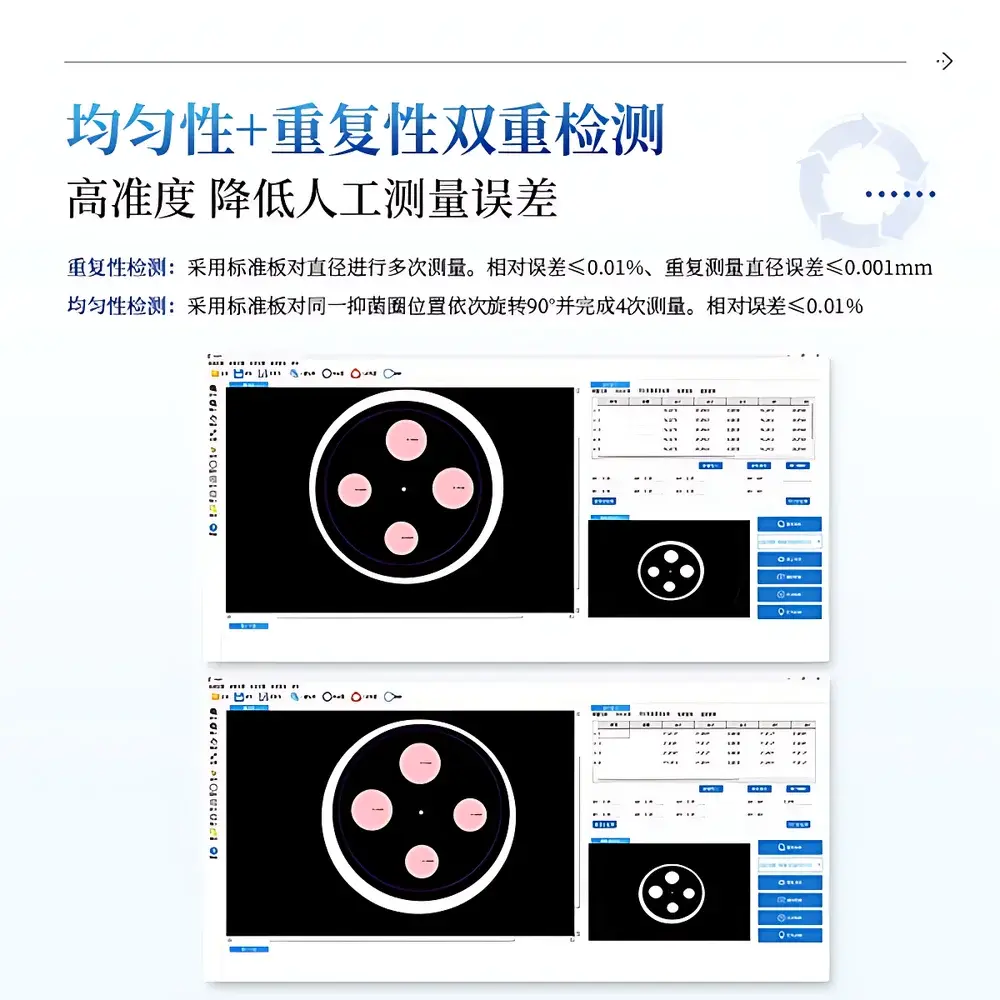
| Calibration Accuracy | ≤0.001 mm diameter measurement error |
| Repeatability | Relative error ≤0.01% |
| Uniformity | Relative error ≤0.02% after 90° rotational sampling |
| Detection Modes | Auto-edge detection, circle-fitting (for irregular zones), manual three-point circle definition |
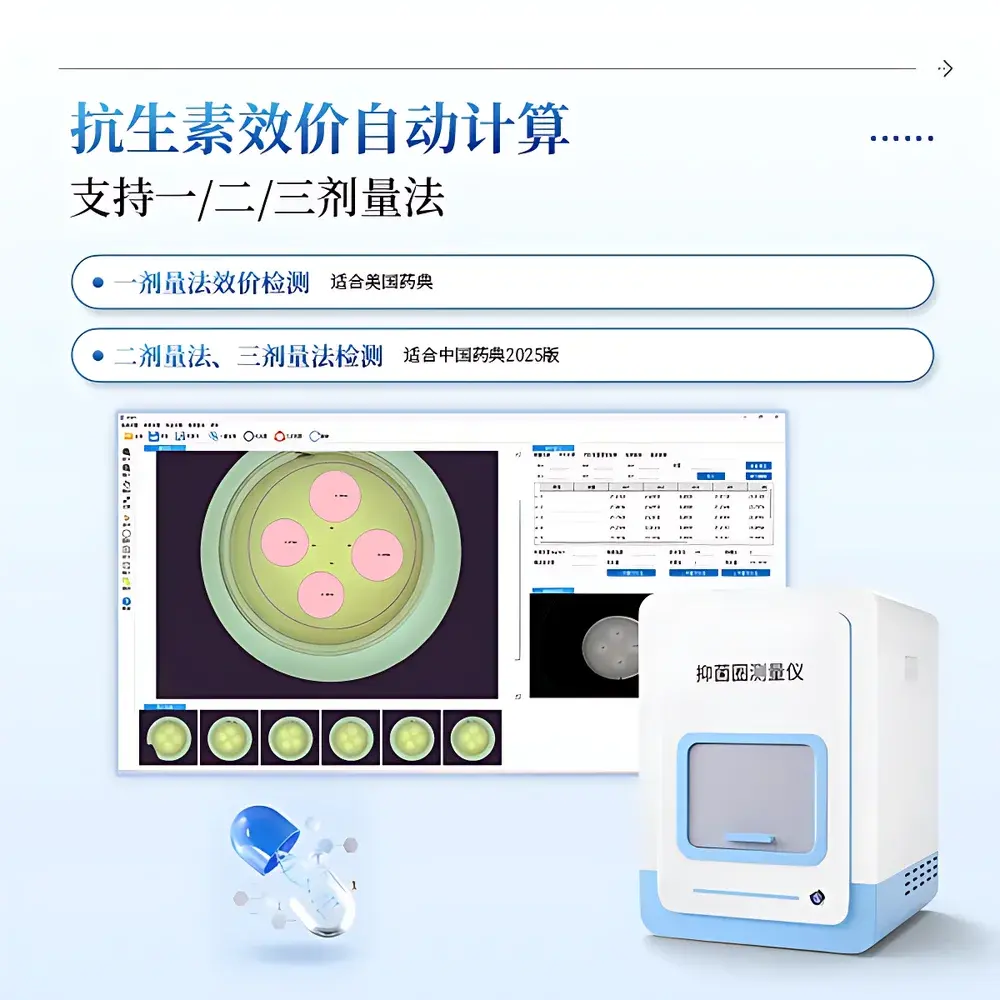
| Assay Compliance | USP <81>, Chinese Pharmacopoeia 2025 (2-dose & 3-dose methods), CLSI M02-A13 |
| Data Security | Role-based multi-user access, immutable raw data storage, FDA 21 CFR Part 11–compliant audit trail, PDF export with digital signature support |
| Cloud Integration | Secure HTTPS-based cloud sync with TLS 1.3 encryption |
Overview
The COMECAUSE IN-Y400 High-Precision Antibiotic Zone-of-Inhibition Analyzer is a fully automated, image-based microbiological assay platform engineered for quantitative measurement of inhibition zones in agar diffusion assays—primarily the Kirby-Bauer disk diffusion method and pharmacopoeial antibiotic potency testing. It operates on the principle of high-fidelity digital image acquisition followed by algorithmic segmentation and geometric quantification of clear zones surrounding antimicrobial agents embedded in solidified agar media. Unlike subjective visual interpretation or manual caliper-based measurement, the IN-Y400 applies calibrated pixel-to-millimeter transformation to deliver traceable, operator-independent measurements of zone diameter (in mm) and derived potency values (e.g., IU/mL or µg/disk). Its optical architecture—featuring a sealed dark-field imaging chamber, dual independently controllable illumination paths, and a 20-megapixel color CMOS sensor—ensures consistent contrast, minimal distortion, and reproducible edge detection across diverse microbial strains and agar formulations. Designed for compliance-critical environments, the system supports both qualitative susceptibility classification and quantitative bioassay validation per international regulatory frameworks.
Key Features
- Sealed dark-box imaging enclosure eliminates ambient light interference and ensures stable illumination geometry.
- Dual-path LED illumination system: top-mounted 360° quad-color visible-light ring (3500–8500 K tunable white balance) and bottom-transmitted diffuse light, each software-controlled with 1% brightness resolution.
- Integrated 254 nm UV-C lamp enables in-chamber sterilization between runs, reducing cross-contamination risk without requiring external disinfection protocols.
- Three-tier detection algorithm suite: auto-edge detection for crisp circular zones; least-squares circle fitting for fragmented or asymmetric inhibition halos; and manual three-point circle definition for low-contrast or diffused boundaries.
- Pharmacopoeia-compliant potency calculation engine supporting USP <81> single-dose assay, Chinese Pharmacopoeia 2025 two-dose and three-dose parallel-line analysis, and CLSI M02-A13 interpretive criteria.
- Sub-micron calibration stability: ≤0.001 mm absolute diameter repeatability verified via NIST-traceable stainless-steel standard plate under ISO/IEC 17025-aligned verification procedures.
Sample Compatibility & Compliance
The IN-Y400 accommodates standard 90–110 mm Petri dishes (including polystyrene, glass, and disposable plastic variants) and supports off-center placement of smaller plates using central aperture alignment. It is validated for use with Mueller-Hinton agar, Iso-Sensitest agar, and other CLSI-recommended media formulations. All measurement outputs—including zone diameters, confidence intervals, and potency estimates—are generated in accordance with ISO 17025–aligned uncertainty budgets. The instrument meets essential requirements for GLP and GMP environments: role-based user authentication, immutable audit trails compliant with FDA 21 CFR Part 11, electronic signatures, and full traceability from raw image capture to final report generation. Regulatory documentation—including Design Qualification (DQ), Installation Qualification (IQ), and Operational Qualification (OQ) templates—is provided as part of the delivery package.
Software & Data Management
The proprietary IN-Analyzer v4.2 software provides a bilingual (English/Chinese) GUI with configurable workflow modules. Image processing pipelines include adaptive thresholding, morphological noise suppression, and sub-pixel edge interpolation to minimize discretization artifacts. Statistical modules compute intra-plate and inter-plate variability, coefficient of variation (CV%), and regression linearity for dose-response curves. Data integrity is enforced through write-once-read-many (WORM) storage architecture: raw TIFF images and unprocessed measurement logs are cryptographically hashed upon acquisition and stored separately from editable analysis reports. Export options include PDF/A-2b (archival-grade), CSV (for LIMS integration), and XML (for SDTM-compliant clinical trial submissions). Cloud synchronization uses end-to-end AES-256 encryption and adheres to ISO/IEC 27001-certified infrastructure standards.
Applications
- Antibiotic susceptibility testing (AST) in clinical microbiology laboratories, aligned with CLSI M100 and EUCAST breakpoints.
- Pharmaceutical quality control: potency verification of bulk antibiotics, finished dosage forms, and sterile ophthalmic solutions per USP <81> and ChP 2025.
- Food safety assessment: evaluation of natural preservatives (e.g., nisin, lysozyme), organic acid efficacy, and antimicrobial packaging film performance.
- Environmental monitoring: detection of residual biocides in wastewater effluents and surface water samples using selective enrichment protocols.
- Cosmetic microbiological testing: preservative efficacy testing (PET) per ISO 11930 and CTFA guidelines.
- Academic research: high-throughput screening of novel antimicrobial peptides, phage lysins, and plant-derived phytochemicals.
FAQ
Does the IN-Y400 support FDA 21 CFR Part 11 compliance?
Yes—the system implements electronic signatures, audit trail logging with tamper-evident hashing, and role-based access controls required for regulated environments.
Can it process overlapping or merged inhibition zones?
The software includes watershed segmentation and contour-splitting algorithms optimized for resolving partially confluent zones, though manual intervention is recommended when zone fusion exceeds 30% of total perimeter.
Is calibration required before each run?
No—once validated using the included NIST-traceable calibration plate, the system retains calibration parameters across sessions; however, daily verification using a control plate is recommended per ISO/IEC 17025.
What file formats are supported for raw image export?
TIFF (uncompressed, 16-bit grayscale or RGB), PNG (lossless compression), and JPEG2000 (with optional wavelet compression).
How does the system handle agar thickness variation across plates?
Users may perform custom height-based scaling via the “Agar Height Compensation” module, which adjusts Z-axis depth weighting during edge detection to mitigate refraction-induced boundary distortion.
Is remote maintenance or software update capability available?
Yes—secure SSH-based remote diagnostics and over-the-air firmware updates are enabled via TLS-encrypted connection to COMECAUSE’s authorized support portal.