COMECAUSE IN-Y500 Automated Antibacterial Zone Diameter & Potency Analyzer
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Region Classification | Domestic (China) |
| Model | IN-Y500 |
| Price | USD 11,400 (approx. based on ¥82,000 at 7.2 exchange rate) |
Overview
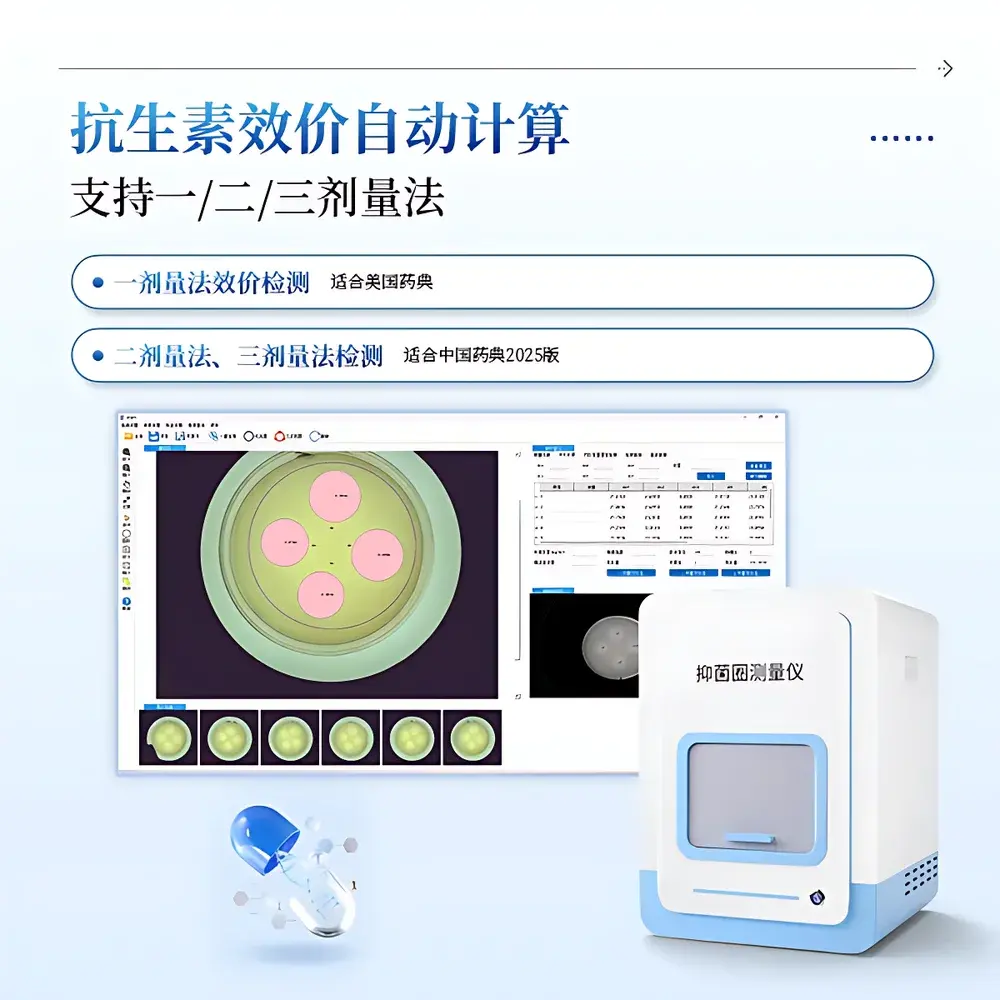
The COMECAUSE IN-Y500 Automated Antibacterial Zone Diameter & Potency Analyzer is a CE-compliant, GxP-aligned digital imaging system engineered for precise, reproducible measurement of inhibition zone diameters in agar diffusion assays (e.g., Kirby-Bauer, cylinder-plate, and disc-diffusion methods). It operates on the principle of high-resolution optical imaging combined with algorithm-driven edge detection and geometric fitting—enabling traceable, objective quantification of antimicrobial activity without manual calipers or subjective visual interpretation. Designed for regulatory environments, the IN-Y500 supports pharmacopoeial compliance workflows including USP , EP 5.1.3, and ChP 2025 (Chinese Pharmacopoeia, Volume IV), particularly for antibiotic potency determination via one-, two-, and three-dose assay designs. Its fully enclosed dark chamber architecture eliminates ambient light interference, while dual independent illumination pathways—top-mounted 360° quad-color visible LED array and bottom-transmitted white light—ensure optimal contrast across diverse agar formulations, colony morphologies, and disc/blank/cylinder-based test configurations.
Key Features
- 25 MP ultra-high-resolution color CMOS camera coupled with a 12 MP, 12 mm focal length macro lens for sub-pixel spatial fidelity
- Adjustable dual-lighting system: software-controlled upper 360° quad-color visible LED (450/525/590/625 nm) and lower broadband white LED; brightness adjustable in 1% increments per channel
- Integrated 254 nm UV-C germicidal lamp for in-chamber sterilization between runs
- Three-tier detection algorithm: (1) Auto-edge detection for sharp, circular zones; (2) Circle-fitting mode for elliptical or irregular zones; (3) Manual 3-point circle definition for low-contrast or fragmented zones
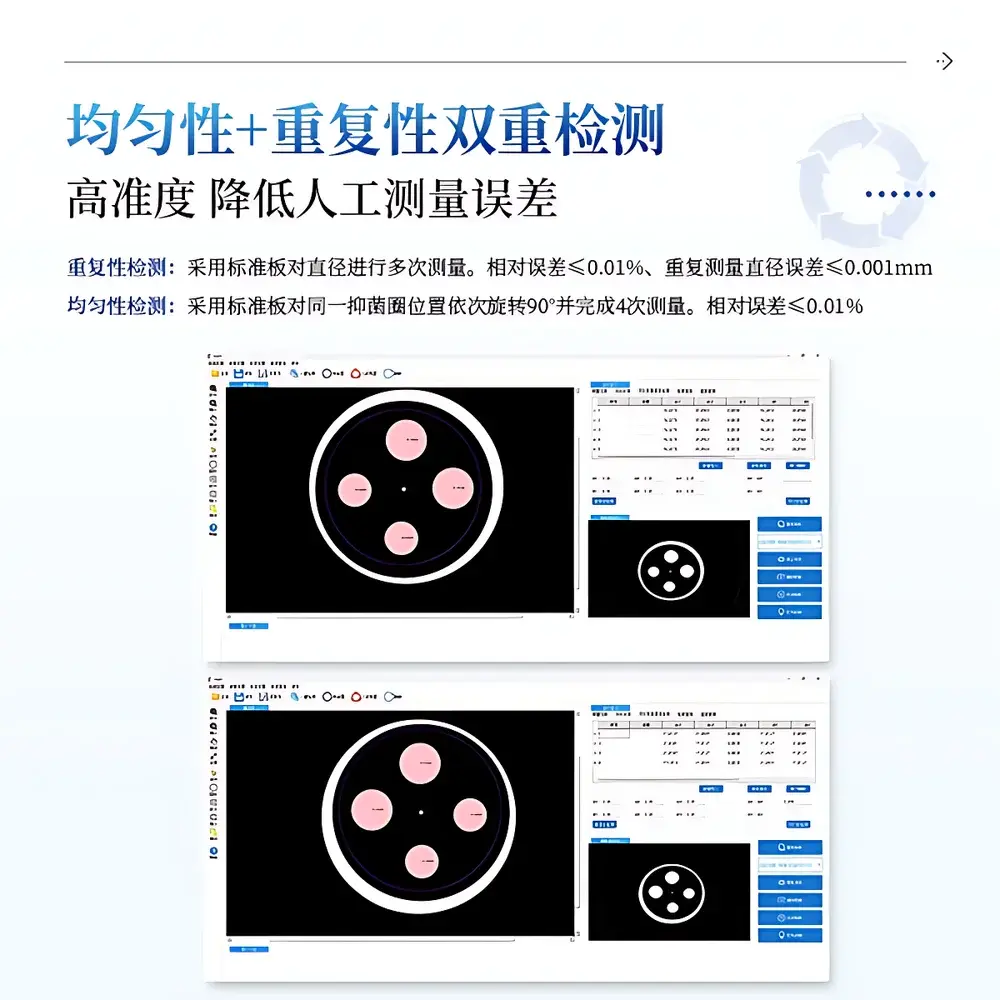
- Validated measurement repeatability: ≤0.001 mm absolute deviation across ≥10 repeated measurements on NIST-traceable calibration plate; uniformity error ≤0.01% across 4 rotational positions (0°/90°/180°/270°)
- Onboard calibration engine supporting both auto-calibration (via lens distortion mapping and sensor geometry) and manual standard-plate-based mm/pixel conversion
Sample Compatibility & Compliance
The IN-Y500 accommodates standard Petri dishes (90–110 mm diameter); smaller plates (e.g., 35 mm or 60 mm) are centered within the field-of-view using the adjustable stage insert. It supports all common antimicrobial assay formats: paper discs, stainless-steel cylinders, agar wells, and gradient strips. The system meets ISO/IEC 17025:2017 requirements for test method validation and is configured to support GLP and GMP audit readiness—including full 21 CFR Part 11 compliance via electronic signatures, role-based access control, immutable raw image archiving, and time-stamped audit trails for every user action (login, image acquisition, measurement, report export, parameter change). All calibration and verification records are retained in encrypted local storage with optional cloud synchronization.
Software & Data Management
The proprietary IN-Analyzer v4.2 software provides bilingual (English/Chinese) UI with context-aware tooltips and embedded pharmacopoeial guidance. It includes FDA-validated reporting modules generating PDF reports compliant with ICH M4Q(R2) specifications. Raw images (.TIFF, 16-bit depth) and metadata (timestamp, operator ID, assay parameters, calibration log) are stored separately from processed results to preserve data integrity. Cloud integration enables secure HTTPS-based upload to private AWS S3 buckets or on-premise NAS systems; users may configure automated backup schedules and retention policies. Audit trail logs capture user identity, timestamp, action type (e.g., “Zone diameter recalculated using circle-fit mode”), and pre/post-value states—fully exportable as CSV for internal QA review or regulatory submission.
Applications
The IN-Y500 serves quality control laboratories in pharmaceutical manufacturing (antibiotic API release testing), contract research organizations (CROs) performing microbiological assay development, academic microbiology departments conducting resistance surveillance, and food safety labs validating preservative efficacy. It is routinely deployed for: potency testing of β-lactams, aminoglycosides, and macrolides per USP and ChP 2025; antimicrobial susceptibility profiling in clinical isolates; preservative challenge testing in cosmetics (ISO 11930); and environmental bioburden assessment in cleanroom monitoring programs. Its quantitative output feeds directly into statistical process control (SPC) dashboards and LIMS integrations via RESTful API.
FAQ
Does the IN-Y500 support multi-dose assay calculations per Chinese Pharmacopoeia 2025?
Yes—it implements validated two-dose and three-dose parallel-line assay algorithms with built-in slope-ratio and validity checks per ChP IV Section 1101.
Can raw images be modified after acquisition?
No—acquired TIFF files are write-protected at the OS level upon saving; only annotated overlays (e.g., zone labels, scale bars) are editable layers stored separately.
Is UV sterilization cycle validated for microbial reduction?
The 254 nm UV lamp delivers ≥10 mJ/cm² dose over 5 minutes, validated against Bacillus atrophaeus spores per ISO 15714, achieving ≥3-log reduction.
How is measurement traceability ensured?
Each measurement links to a unique calibration certificate ID; pixel-to-mm conversion is logged with date, operator, standard plate ID, and residual error (RMS ≤0.005 mm).
Does the system integrate with laboratory information management systems (LIMS)?
Yes—via configurable JSON/REST API endpoints supporting HL7 ADT and ASTM E1384 message structures; custom middleware connectors available upon request.