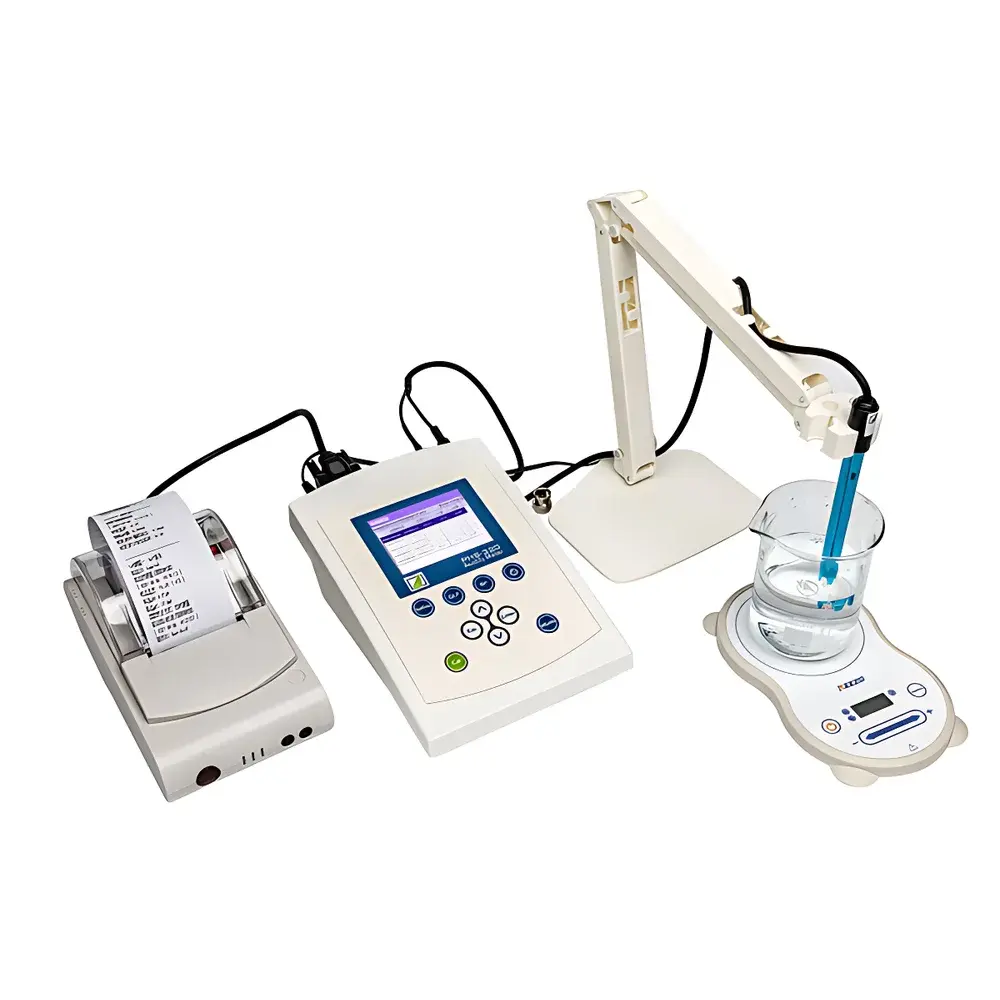
FangZhou PHS-320 Biological Pharmaceutical pH Meter
| Brand | FangZhou |
|---|---|
| Origin | Sichuan, China |
| Model | PHS-320 Biological Pharmaceutical pH Meter |
| Instrument Type | Multifunctional Benchtop pH/mV/ISE Meter |
| Measurement Accuracy | ±0.002 pH |
| pH Range | (−5.000 to 20.000) pH |
| Adjustable Resolution | 0.001 / 0.01 / 0.1 pH |
| mV Range | (−2000.00 to +2000.00) mV |
| mV Resolution | 0.01 / 0.1 / 1 mV |
| Temperature Range | (−20.0 to 135.0) °C |
| Temp. Resolution | 0.1 °C |
| Temp. Accuracy | ±0.2 °C |
| Input Impedance | ≥3 × 10¹² Ω |
| Calibration Points | 1–5 point automatic buffer recognition |
| Supported Buffers | 33 NIST-traceable standards across 7 buffer groups (JJG119, DIN 19266/19267, JIS Z8802, USP, Merck, Mettler Toledo) |
| GLP Compliance | Full audit trail with sample/personnel ID (10 each), timestamped data logging, and electronic record integrity |
| Data Storage | ≥100,000 measurements with time/date stamp |
| Interface | RS232 with three operational modes (printer output, PC data export to Excel/Word/Notepad, bidirectional instrument control) |
| Power | DC 9 V, 500 mA (center-positive) |
Overview
The FangZhou PHS-320 Biological Pharmaceutical pH Meter is a microprocessor-controlled, benchtop electrochemical analyzer engineered for high-precision pH, mV, ORP, ISE, and temperature measurements in regulated life science and pharmaceutical environments. It operates on the fundamental principle of potentiometric measurement—detecting the potential difference between a pH-sensitive glass electrode and a stable reference electrode immersed in an aqueous solution—while compensating for thermal drift via integrated ATC (Automatic Temperature Compensation) or manual input. Designed specifically for biological and pharmaceutical applications—including cell culture media, parenteral solutions, bioprocess buffers, and sterile water systems—the instrument meets stringent requirements for traceability, repeatability, and regulatory documentation. Its architecture supports rigorous quality assurance workflows under GMP, GLP, and ISO/IEC 17025 frameworks, with built-in compliance features aligned with FDA 21 CFR Part 11 expectations for electronic records and signatures.
Key Features
- True-color TFT LCD display with graphical interface, real-time curve plotting, and intuitive menu navigation
- Adjustable resolution: 0.001 pH / 0.01 mV for critical applications requiring metrological rigor
- Five measurement modes: Real-time, Timed, Auto-Fast, Auto-Medium, and Auto-Slow—optimized for dynamic or equilibrium conditions
- Advanced electrode diagnostics: continuous self-check of electrode slope, offset, and asymmetry potential; supports isoelectric adjustment (−20.000 to +20.000 pH)
- Multi-point calibration with auto-buffer recognition across 33 NIST-traceable standards, including USP, DIN, JIS, and Chinese JJG119 specifications
- Robust input impedance (>3 × 10¹² Ω) ensures minimal loading error during low-conductivity measurements (e.g., purified water, serum, or low-ionic-strength biological matrices)
- Configurable GLP parameters: 10 user-defined sample IDs, 10 operator IDs, timestamped logging, and editable metadata fields
- Flexible data handling: internal storage capacity ≥100,000 entries with date/time stamp; RS232 port supporting printer output (GLP-formatted reports), direct PC export to Excel/Word/Notepad, and bidirectional command control
Sample Compatibility & Compliance
The PHS-320 accommodates diverse sample types through application-specific electrode configurations—including low-conductivity pure water electrodes, food-grade stainless-steel penetration probes, semi-micro and micro-volume electrodes (≥15 µL), planar surface sensors, and chemically resistant electrodes for HF, strong alkalis, or sulfide-rich suspensions. All configurations maintain metrological integrity across pH −5.000 to 20.000 and temperature −20.0 to 135.0 °C. The system complies with international standardization requirements for analytical instrumentation: calibration traceability to NIST SRMs, adherence to ISO 3696 (water purity), USP (pH measurement), and EP 2.2.3 (potentiometric determination). Its GLP implementation includes immutable audit trails, electronic signature support, and full data export capability—enabling compliance with FDA 21 CFR Part 11, EU Annex 11, and ISO/IEC 17025 clause 7.7.
Software & Data Management
Data integrity is enforced via embedded firmware that logs every measurement with UTC-synchronized timestamps, operator ID, sample ID, electrode serial number, calibration history, and environmental metadata. Exported files retain native formatting and are compatible with LIMS integration protocols. The RS232 interface operates in three mutually exclusive modes: (1) GLP-compliant report printing with customizable templates (simple, standard, GLP, or user-defined); (2) ASCII-based streaming to spreadsheet or text editors without proprietary software dependencies; and (3) SCPI-like command-response protocol for automated test sequencing in QC laboratories. No cloud connectivity or remote access is implemented—ensuring data sovereignty and alignment with air-gapped facility policies common in pharmaceutical manufacturing.
Applications
- Pharmaceutical QA/QC: pH verification of WFI (Water for Injection), Purified Water, and buffer solutions per USP
- Biotechnology: monitoring of mammalian cell culture media, fermentation broths, and downstream purification steps
- Clinical diagnostics: stability testing of reagent kits, serum pH profiling, and electrophoresis buffer validation
- Food & beverage R&D: acidification kinetics in dairy, meat, and fermented products using food-grade penetration electrodes
- Environmental testing: pH analysis of wastewater, sludge extracts, and soil leachates with specialized suspended-solid electrodes
- Academic research: precise ISE-based ion quantification (e.g., Na⁺, K⁺, Ca²⁺, F⁻) in physiological and enzymatic assays
FAQ
Does the PHS-320 support 21 CFR Part 11-compliant electronic signatures?
Yes—through configurable user authentication, immutable audit trails, and exportable raw data with full metadata, it satisfies predicate rule requirements for electronic records.
Can the instrument perform simultaneous pH and temperature measurements with automatic compensation?
Yes—integrated ATC uses either built-in or external Pt1000 sensors, applying the Nernst equation correction in real time across −20.0 to 135.0 °C.
Is calibration traceable to national standards?
All supported buffer sets include NIST-traceable certificates; users may load custom calibration curves with documented uncertainty budgets.
What electrode options are validated for ultra-pure water measurement?
The FZ-600T and dedicated low-conductivity electrodes feature double-junction reference systems and LiCl electrolyte to minimize junction potential drift in <1 µS/cm water.
How is data security maintained during RS232 transmission?
No encryption is applied—but the protocol is deterministic and ASCII-based, enabling secure transfer within validated local networks without third-party drivers or cloud intermediaries.