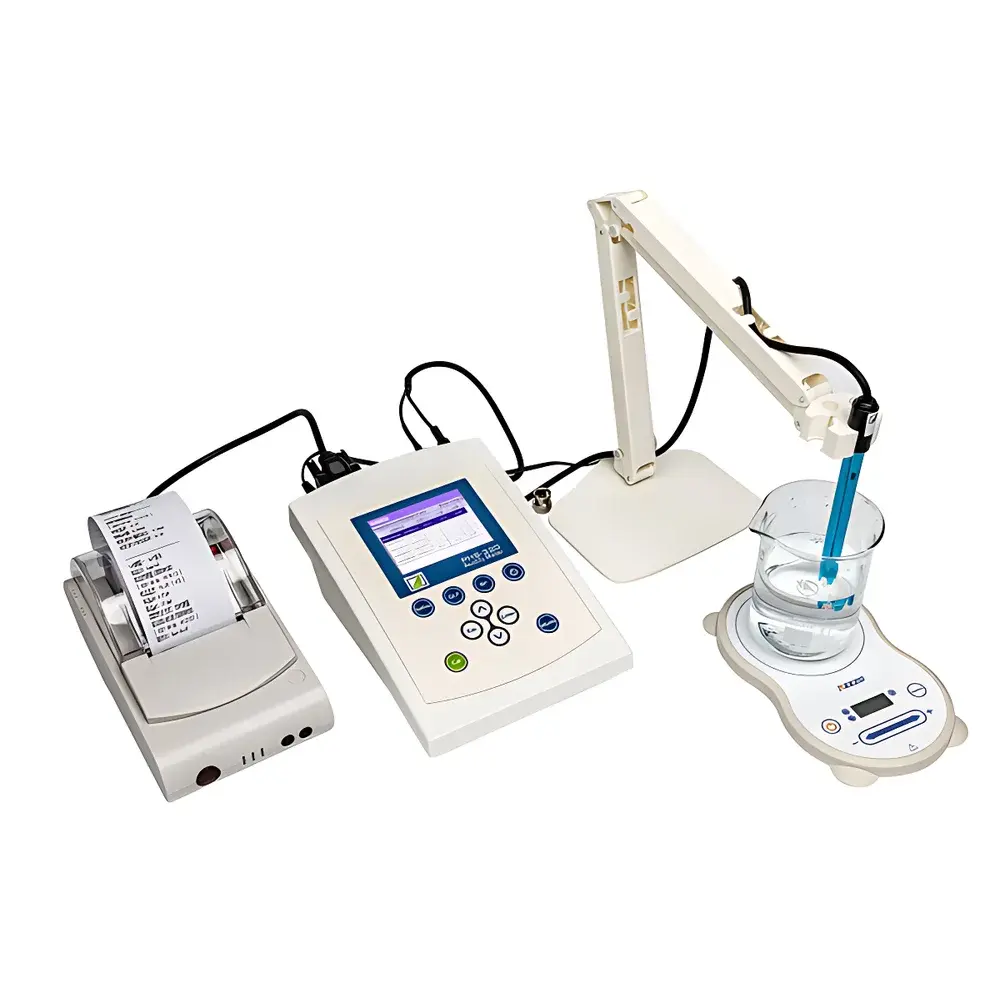
FangZhou PHS-320 Pharmaceutical-Grade pH Meter for Organic Compound Solutions
| Brand | FangZhou |
|---|---|
| Origin | Sichuan, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | PHS-320 Pharmaceutical-Grade pH Meter |
| Instrument Type | Multiparameter |
| Measurement Accuracy | ±0.002 pH |
| pH Range | (−5.000 to 20.000) pH |
| Resolution | 0.001 / 0.01 / 0.1 pH (selectable) |
| mV Range | (−2000.00 to +2000.00) mV |
| mV Resolution | 0.01 / 0.1 / 1 mV (selectable) |
| Temperature Range | (−20.0 to 135.0) °C |
| Temp. Resolution | 0.1 °C |
| Temp. Accuracy | ±0.2 °C ±1 digit |
| Input Impedance | ≥3 × 10¹² Ω |
| Calibration | Up to 5-point, auto-buffer recognition (NIST/USP/DIN/JIS/Merck/MTT standards) |
| GLP Compliance | Full audit trail, user & sample ID (10 each), timestamped data logging (≥100,000 entries) |
| Interface | RS232 with printer export (GLP/simple/custom formats), PC data transfer (Excel/Word/Notepad), bidirectional control |
| Power | DC 9 V, 500 mA (center-positive) |
Overview
The FangZhou PHS-320 Pharmaceutical-Grade pH Meter is a microprocessor-controlled, high-precision electrochemical instrument engineered for rigorous measurement of hydrogen ion activity in aqueous and semi-aqueous matrices containing organic compounds—particularly relevant to pharmaceutical formulation, bioprocess monitoring, and regulatory-compliant quality control. It operates on the fundamental principle of potentiometric pH measurement: the potential difference between a glass pH electrode and a reference electrode is measured under zero-current conditions and converted to pH via the Nernst equation (E = E⁰ − (RT/F) ln(10)·pH). Designed for environments where trace-level organic interference, low ionic strength, or non-aqueous solvents may compromise electrode response stability, the PHS-320 integrates advanced signal conditioning, high-input-impedance circuitry (≥3 × 10¹² Ω), and configurable isopotential adjustment to maintain accuracy across challenging sample types—including protein-rich buffers (e.g., TRIS, PBS), viscous formulations, suspensions, and ultra-pure water systems. Its extended pH range (−5.000 to 20.000) supports both strongly acidic and alkaline process streams encountered in API synthesis and cleaning validation.
Key Features
- True-color 4.3-inch TFT LCD display with graphical curve plotting, intuitive menu navigation, and real-time trend visualization
- Selectable resolution modes: 0.001 pH / 0.01 mV for high-fidelity research; 0.01 pH / 0.1 mV for routine QC applications
- Five adaptive measurement modes: Real-time, Timed, Auto-Fast, Auto-Medium, and Auto-Slow—optimized for dynamic equilibration in viscous, colloidal, or low-conductivity samples
- Isopotential adjustment (−20.000 to +20.000 pH) compensates for variable zero-potential electrodes used in specialized applications (e.g., solid-contact or polymer membrane sensors)
- Auto-temperature compensation (ATC) with dual-mode support: automatic recognition of Pt1000 or thermistor probes, or manual entry within −20.0–135.0 °C
- Comprehensive self-diagnostic suite: continuous assessment of electrode slope, asymmetry potential, and internal resistance; alerts for dry junction, clogged frit, or degraded glass membrane
- Onboard clock and calendar synchronization ensure ISO/IEC 17025 and FDA 21 CFR Part 11–compliant timestamping for all measurements and calibrations
Sample Compatibility & Compliance
The PHS-320 is validated for use with diverse sample categories requiring pharmacopeial-grade metrology: purified water (USP , EP 2.2.38), cell culture media, protein solutions, emulsions, soil extracts, and aggressive chemical matrices (e.g., HF-containing etchants, caustic soda solutions). Its modular electrode architecture supports 12 purpose-engineered probe configurations—including FZ-600T general-purpose electrodes, low-ionic-strength pure-water sensors, food-grade stainless-steel penetration probes, planar surface electrodes, and HF-resistant antimony electrodes—each selected per ICH Q2(R2) method suitability criteria. The instrument fully conforms to GLP (Good Laboratory Practice) requirements: all calibration events, measurement records, user actions, and system diagnostics are stored with immutable timestamps, digital signatures (via optional USB authentication), and full audit-trail export capability. Data integrity meets FDA 21 CFR Part 11 Annex 11 and ISO/IEC 17025:2017 clause 7.7.1 specifications for electronic records.
Software & Data Management
Data handling is governed by an embedded GLP framework supporting 10 user IDs and 10 sample IDs, with hierarchical access control and password-protected configuration changes. Internal memory retains ≥100,000 timestamped entries—including raw mV, temperature, slope, offset, buffer ID, and calibration history—with customizable storage intervals (0–6000 s). RS232 interface enables three interoperable workflows: (1) Direct printing to thermal printers in GLP-compliant report format (including instrument ID, operator, sample ID, calibration certificate, uncertainty statement); (2) Bidirectional PC communication using ASCII protocol compatible with Microsoft Excel, Word, and Notepad—enabling automated statistical analysis (RSD, confidence intervals) and trending per ASTM E2554; (3) Remote instrument control via terminal emulation for integration into LIMS or MES platforms. All exported files retain cryptographic hash verification to prevent tampering.
Applications
- Pharmaceutical QC: pH verification of injectables, ophthalmic solutions, and buffer preparations per USP and EP 2.2.3
- Biotechnology: Monitoring of fermentation broths, cell lysates, and chromatography eluents where organic solvents or surfactants affect junction potential
- Food & Beverage: Rapid pH profiling of dairy products, fermented foods, and acidic beverages using food-grade penetration electrodes
- Environmental Testing: Soil pore-water analysis, wastewater effluent monitoring, and leachate characterization under EPA Method 9040C
- Academic Research: Electrode characterization studies, pKa determination of weak acids in mixed-solvent systems, and corrosion inhibitor evaluation
- Power & Semiconductor: Ultrapure water (UPW) monitoring in semiconductor fab ultrapure water loops per SEMI F63
FAQ
Does the PHS-320 support NIST-traceable calibration verification?
Yes—its auto-buffer recognition engine includes 33 certified standard solutions aligned with NIST SRM, DIN 19266/19267, JIS Z8802, and USP reference materials, with documented uncertainty budgets for each.
Can it measure pH in non-aqueous or mixed-solvent systems?
It is optimized for aqueous-organic mixtures (e.g., ethanol/water, acetonitrile/water up to 40% v/v) when paired with solvent-compatible electrodes; however, absolute pH values require matrix-matched calibration per ISO 17558.
Is the instrument compliant with FDA 21 CFR Part 11 for electronic records?
Yes—when used with enabled audit-trail logging, user authentication, and secure RS232 export, it satisfies electronic signature, record retention, and change-control requirements outlined in Subpart B.
What electrode options are available for low-conductivity samples like distilled water?
The dedicated ultra-low-ionic-strength electrode (model FZ-PW-ULS) features a double-junction design with KCl-free electrolyte and ceramic frit to minimize diffusion potential errors in conductivity <5 µS/cm matrices.
How does the isopotential function improve measurement reliability?
It allows manual offset correction of the reference electrode’s zero-potential drift—critical when using alternative reference systems (e.g., Ag/AgCl in non-aqueous media) or aged electrodes exhibiting asymmetric potentials beyond ±30 mV.