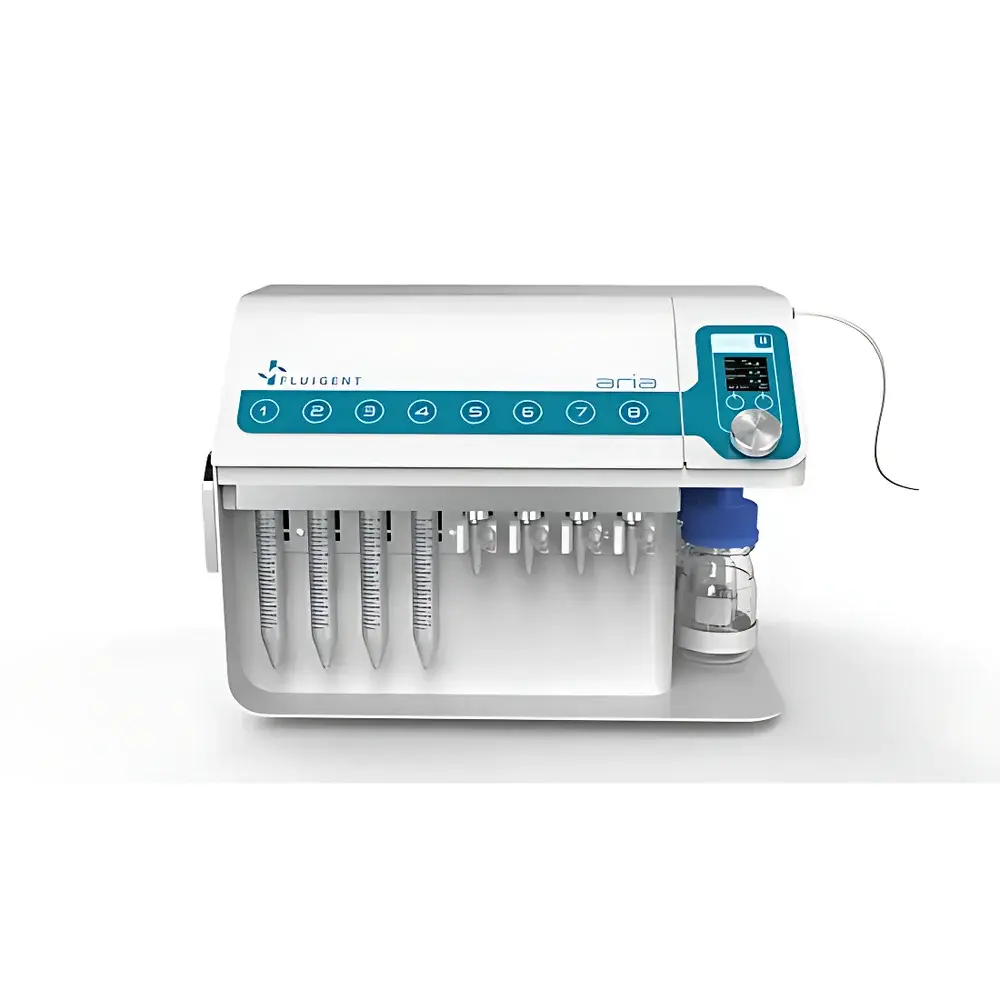
Fluigent ARIA Automated Microfluidic Perfusion System
| Brand | Fluigent |
|---|---|
| Origin | France |
| Model | ARIA |
| Instrument Type | Automated |
| Reservoir Capacity | 2 mL & 15 mL (8 positions) + 100 mL rinse reservoir |
| Flow Rate Range | 40 µL/min to 1 mL/min (water, typical) |
| Pressure Supply | Up to 2 bar |
| Capillary ID/OD | 250 µm / 1.59 mm, FEP material |
| Software | Windows 7 or later |
| Compliance | Designed for GLP-compliant live-cell imaging workflows, compatible with FDA 21 CFR Part 11–ready data logging when integrated with validated software environments |
Overview
The Fluigent ARIA Automated Microfluidic Perfusion System is an engineered platform for precise, programmable, and contamination-minimized fluid delivery in live-cell microscopy and electrophysiology applications. Based on pressure-driven microfluidic control—leveraging Fluigent’s proprietary Flow-EZ™ pneumatic actuation technology—the ARIA delivers stable, pulseless flow across a broad dynamic range (40 µL/min to 1 mL/min), enabling both slow, physiological perfusion and rapid solution exchange required for time-resolved functional assays. Its modular architecture integrates seamlessly with inverted microscopes, patch-clamp rigs, and microfluidic chips, supporting long-term (multi-hour to multi-day) imaging experiments without manual intervention. Unlike peristaltic or syringe-based systems, the ARIA eliminates mechanical pulsation and backflow risk through closed-loop pressure regulation and passive valve switching, ensuring high reproducibility and minimal shear stress on sensitive cell cultures.
Key Features
- Automated sequential perfusion of up to 10 distinct reagents via integrated 2-Switch or M-Switch modules—supporting single-chip or multi-chip configurations (up to 4 chips simultaneously)
- Single-output design with universal compatibility: connects directly to any standard perfusion chamber, microfluidic device, or recording setup via FEP capillaries (250 µm ID, 1.59 mm OD)
- “Dark Room Mode”: tactile buttons with low-intensity LED backlighting enable silent, non-phototoxic manual operation under light-sensitive experimental conditions (e.g., fluorescence recovery after photobleaching, optogenetics)
- Dual-reservoir system: eight 15 mL or 2 mL reagent vials plus dedicated 100 mL rinse reservoir for automated cleaning between protocols
- Zero-cross-contamination architecture: chemically inert FEP tubing, non-wetting surfaces, and programmable “Stop-Flow” function prevent carryover during reagent transitions
- Compact footprint (230 × 170 × 120 mm) and plug-and-play USB interface reduce lab space requirements and integration complexity
Sample Compatibility & Compliance
The ARIA supports a wide range of biological samples—including primary neurons, iPSC-derived cardiomyocytes, organoids, and adherent or suspension cell lines—without compromising viability during extended perfusion. Its pressure-limited operation (max 2 bar) ensures compatibility with delicate microfluidic chips (e.g., OrganoPlate®, μ-Slides, custom PDMS devices) and glass-bottom dishes. The system conforms to core principles of Good Laboratory Practice (GLP): full audit-trail capability via timestamped protocol logs, user-access controls, and versioned method storage. When paired with validated third-party acquisition software (e.g., MetaMorph, NIS-Elements, or PatchMaster), it meets traceability requirements aligned with ISO/IEC 17025 and FDA 21 CFR Part 11 for regulated preclinical studies.
Software & Data Management
ARIA is controlled via Fluigent’s intuitive ARIA Control Software (Windows 7+), which enables graphical programming of multi-step perfusion sequences—including timed injections, concentration gradients, flow-rate ramps, and conditional triggers based on external TTL signals. Protocols are saved as encrypted .aria files with embedded metadata (user ID, timestamp, hardware configuration). The software supports batch execution, real-time flow monitoring (via optional pressure sensor feedback), and export of CSV-formatted logs for downstream analysis in MATLAB, Python (Pandas), or GraphPad Prism. Integration with LabVIEW and Python APIs allows customization for high-content screening pipelines and closed-loop stimulation paradigms.
Applications
- Long-term live-cell imaging: Sustained nutrient/gas exchange over 7+ hours with sub-minute temporal resolution—reducing experimenter workload by >60% versus manual pipette-based approaches
- Electrophysiological recordings: Rapid solution exchange (<500 ms rise time) for voltage-clamp or current-clamp experiments requiring precise agonist/antagonist application
- Immunofluorescence workflow automation: Sequential fixation, permeabilization, blocking, and antibody incubation steps without sample removal from the microscope stage
- Microphysiological system (MPS) support: Continuous perfusion of organ-on-chip platforms to maintain barrier integrity and metabolic activity
- High-throughput drug screening: Reproducible dosing across 96-well plate-compatible microfluidic arrays using M-Switch expansion
FAQ
Does the ARIA require an external air compressor?
Yes—it operates with clean, oil-free, dry compressed gas (N₂ or shop air) regulated to ≤2 bar; a standard laboratory-grade compressor with 0.01 µm filtration is recommended.
Can I use the ARIA with non-Fluigent microfluidic chips?
Absolutely—the single-output port uses standard 1/4″-28 UNF threaded fittings and accepts any FEP or PTFE tubing with compatible outer diameter (1.59 mm); adapter kits for common chip interfaces (e.g., Elveflow, Micronit) are available.
Is flow calibration required before each experiment?
No—factory-calibrated pressure-to-flow transfer functions are embedded in firmware; however, users may perform optional in-line flow verification using Fluigent’s Flow Unit sensor for critical quantitative applications.
How does the “Stop-Flow” function enhance imaging stability?
It halts fluid motion without releasing pressure, eliminating mechanical vibration and meniscus drift during acquisition—critical for high-magnification time-lapse or super-resolution imaging.
What level of maintenance does the ARIA demand?
Minimal: routine capillary flushing with ethanol/water and monthly inspection of O-rings; no pumps, motors, or disposable cartridges to replace—designed for >5 years of continuous lab use.