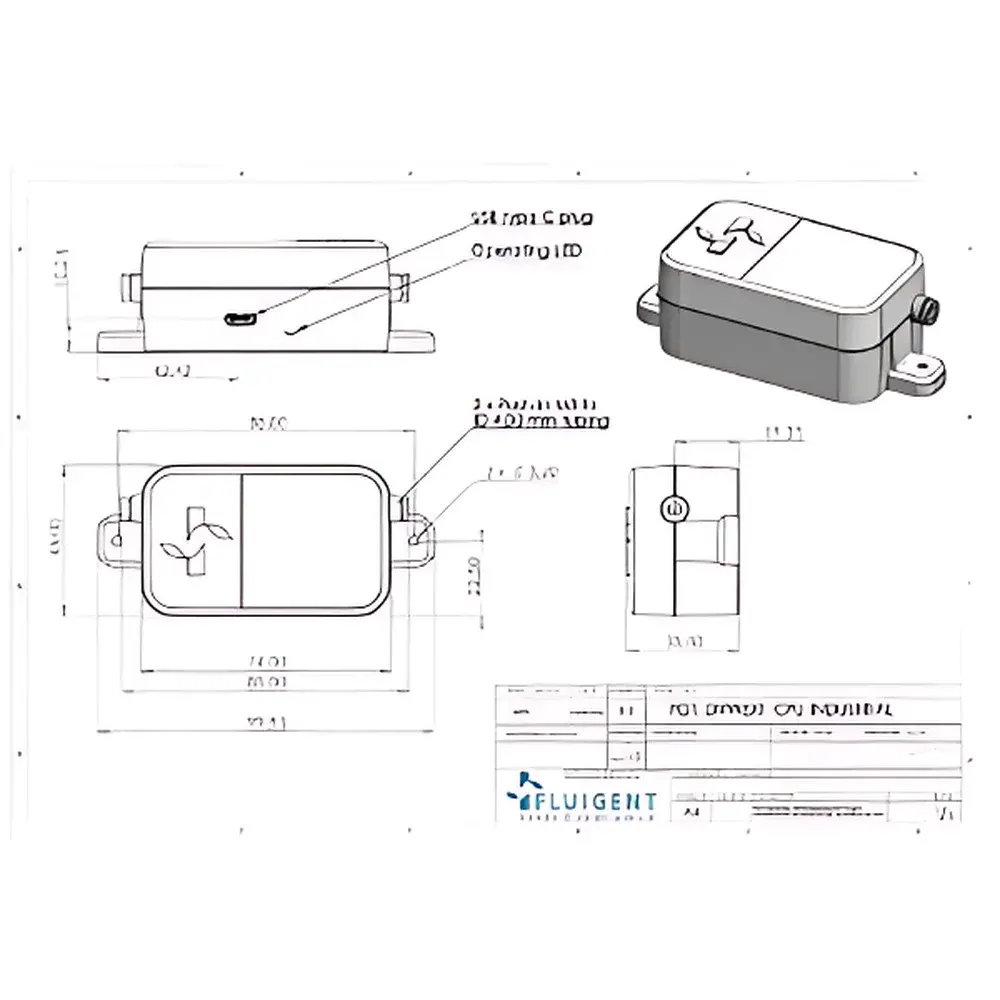
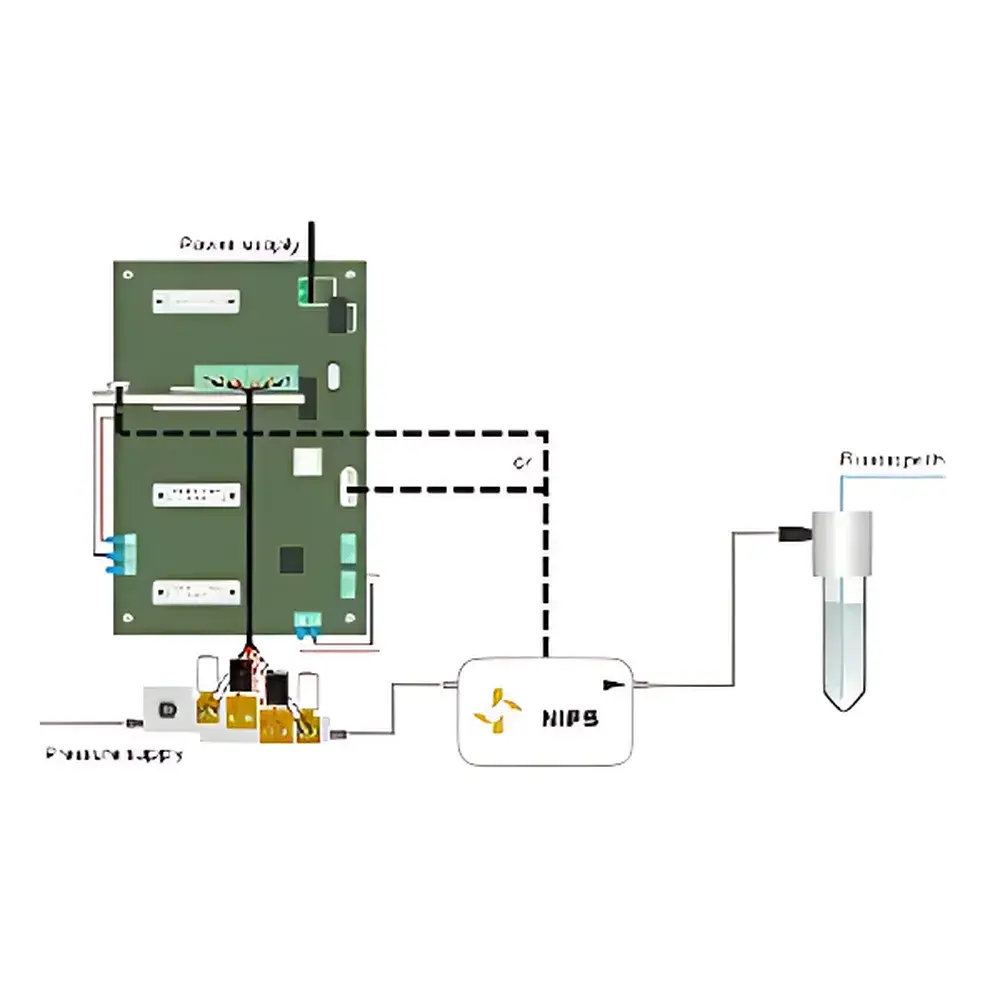
Fluigent NIFS Non-Invasive Industrial OEM Mass Flowmeter
| Brand | Fluigent |
|---|---|
| Origin | France |
| Model | NIFS |
| Liquid Mass Flow Accuracy | ±5% of measured value |
| Flow Range | 100 µL/min to 10 mL/min |
| Minimum Detectable Flow Increment | 5 µL |
| Response Time (Reading) | ~10 s |
| Calibration Time (if required) | 10–30 s |
| Contactless Operation | Yes |
| Bidirectional Flow Monitoring | Yes |
| Compliance | CE, RoHS, ISO 9001-certified manufacturing |
Overview
The Fluigent NIFS is a non-invasive, industrial-grade OEM mass flowmeter engineered for precise, maintenance-free liquid flow monitoring in microfluidic systems. Unlike conventional thermal or Coriolis-based flowmeters, the NIFS operates on an indirect pneumatic principle: it measures real-time gas flow variations induced by liquid displacement within a sealed fluidic path—enabling fully contactless liquid flow quantification. This architecture eliminates physical sensor exposure to process fluids, thereby removing risks associated with fouling, clogging, chemical incompatibility, and cross-contamination. Designed specifically for integration into automated, sterile, or multi-reagent microfluidic platforms—including organ-on-chip, droplet generation, and perfusion bioreactors—the NIFS delivers robust performance without routine recalibration or cleaning protocols. Its operational envelope spans 100 µL/min to 10 mL/min, with a typical accuracy of ±5% of measured value (m.v.) under stable temperature and pressure conditions. The device supports bidirectional flow detection and is optimized for extended-duration experiments (>1 hr), where temporal resolution requirements are secondary to long-term stability and system hygiene.
Key Features
- Contactless operation: No wetted parts; sensor remains isolated from all process liquids
- Zero calibration dependency: Factory-calibrated for broad fluid compatibility—no user-side fluid-specific recalibration required
- No maintenance or cleaning: Eliminates downtime, sterilization cycles, and consumable replacement
- Bidirectional real-time flow monitoring: Captures forward and reverse flow events without hardware reconfiguration
- OEM-optimized interface: Compact form factor with analog (0–10 V) and digital (RS-485/Modbus RTU) outputs for seamless PLC or DAQ integration
- Chemical inertness: Compatible with aqueous buffers, organic solvents (e.g., ethanol, isopropanol), oils, surfactant solutions, and biological media including whole blood and plasma
Sample Compatibility & Compliance
The NIFS is compatible with all Newtonian and weakly non-Newtonian liquids commonly used in life science and industrial microfluidics—including cell culture media, serum-containing solutions, hydrogels (pre-gelation), emulsions, and low-viscosity polymer precursors. Its non-contact design inherently satisfies stringent hygienic requirements for aseptic processing and single-use systems. While not certified under FDA 21 CFR Part 11, the NIFS supports GLP/GMP-aligned workflows when integrated with validated control software (e.g., Fluigent’s FLOW-EZ or third-party SCADA platforms with audit-trail capabilities). Manufacturing complies with ISO 9001:2015, and the device bears CE marking per EU Directive 2014/30/EU (EMC) and 2011/65/EU (RoHS).
Software & Data Management
The NIFS communicates via standard industrial protocols—primarily RS-485 Modbus RTU—for direct integration into LabVIEW, Python (PyModbus), MATLAB, or custom HMI environments. Analog output enables plug-and-play connection to data acquisition systems without driver installation. Fluigent provides open communication documentation and example scripts for rapid validation. For traceability-critical applications, users may configure external logging tools to record timestamps, flow values, and direction flags at configurable intervals (default 1 Hz). Though the NIFS itself does not store historical data or implement onboard encryption, its deterministic response behavior ensures high reproducibility across repeated experimental runs—facilitating post-hoc statistical analysis of flow stability over time.
Applications
- Organ-on-chip perfusion systems requiring continuous, contamination-free media exchange
- Droplet microfluidics platforms switching between immiscible phases (e.g., aqueous/organic/surfactant blends) without recalibration delays
- Cell encapsulation workflows involving sequential injection of hydrogel precursors, crosslinkers, and cell suspensions
- In-line quality monitoring during microscale synthesis or analytical sample preparation (e.g., enzymatic assays, nucleic acid purification)
- Automated bioprocessing modules where sterilizable, disposable fluid paths must remain intact across multiple batches
FAQ
Does the NIFS require periodic recalibration?
No—factory calibration covers the full specified flow range and fluid types. Recalibration is unnecessary unless subjected to extreme mechanical shock or ambient temperature shifts exceeding ±10 °C from nominal operating conditions.
Can the NIFS measure viscous or particulate-laden fluids?
It monitors flow indirectly via gas dynamics; therefore, viscosity changes do not affect accuracy—as long as the liquid remains pumpable and does not generate excessive backpressure beyond system specifications. Suspended particles <5 µm pose no risk due to zero fluid contact.
What is the recommended upstream/downstream configuration?
The NIFS must be installed between a regulated gas source (e.g., Fluigent PX, F-OEM pressure controller) and the headspace of a sealed reservoir—never in-line with liquid flow. Refer to Fluigent Application Note AN-NIFS-01 for piping schematics and pressure drop guidelines.
Is the NIFS suitable for regulatory submissions (e.g., ISO 13485, FDA filings)?
While the device itself lacks formal medical device certification, its design principles—contactless operation, repeatability, and compatibility with validated pressure controllers—support inclusion in Class I/IIa system-level validations when accompanied by documented risk assessments and IQ/OQ protocols.