Fluigent OMI Microfluidic Organ-on-a-Chip Platform
| Brand | Fluigent |
|---|---|
| Origin | France |
| Model | OMI |
| Instrument Type | Fully Automated, Battery-Powered |
| Reservoir Capacity | 4 × 4 mL |
| Device Height | 6 cm |
| Footprint | < 12 cm × 8 cm |
| Weight | < 800 g |
| Flow Rate Range | 0.1–100 µL/min (adjustable per channel) |
| Flow Control Precision | ±2% CV |
| Operating Environment | Standard CO₂ incubator (37 °C, 5% CO₂) |
| Power | Rechargeable Li-ion battery (2 h continuous operation) |
| Connectivity | Wi-Fi & Bluetooth 5.0 |
| Control Interface | Web-based dashboard & tablet app |
| Data Storage | Encrypted cloud sync with local backup |
| Compliance | Designed for GLP-compliant workflows |
Overview
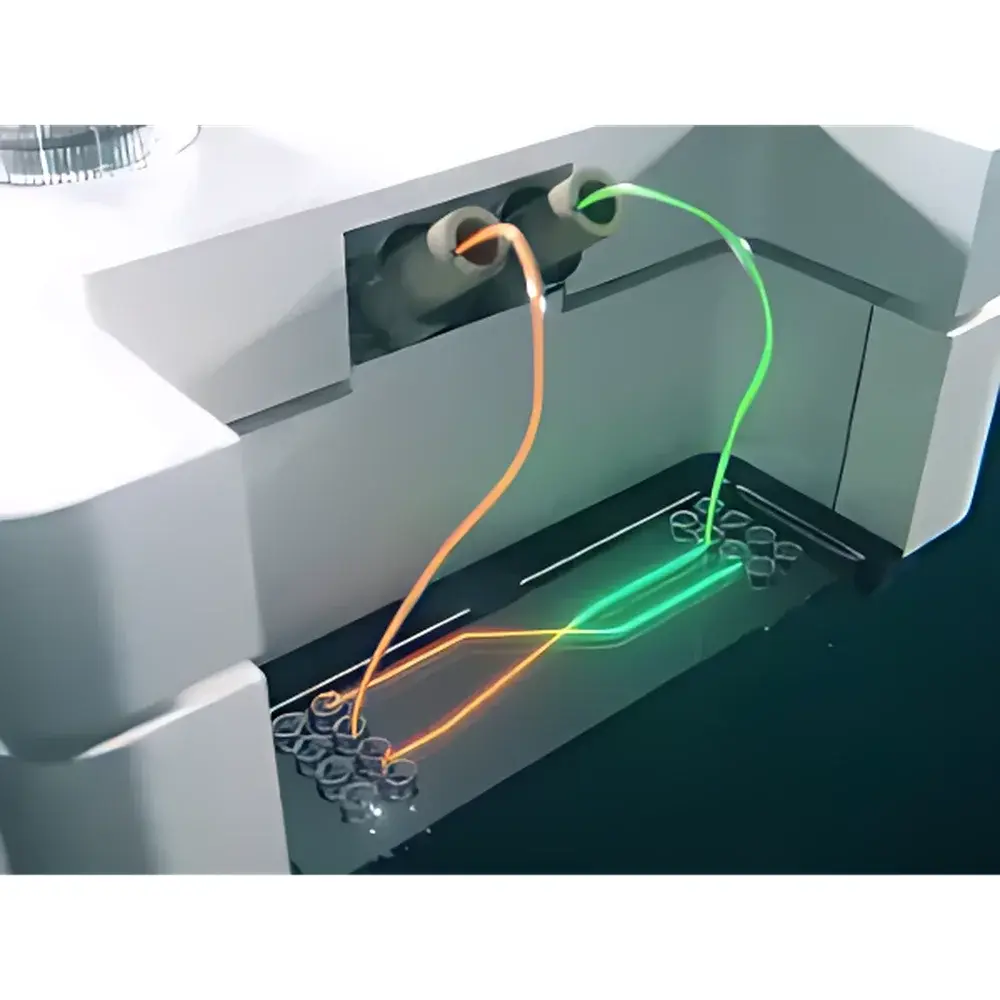
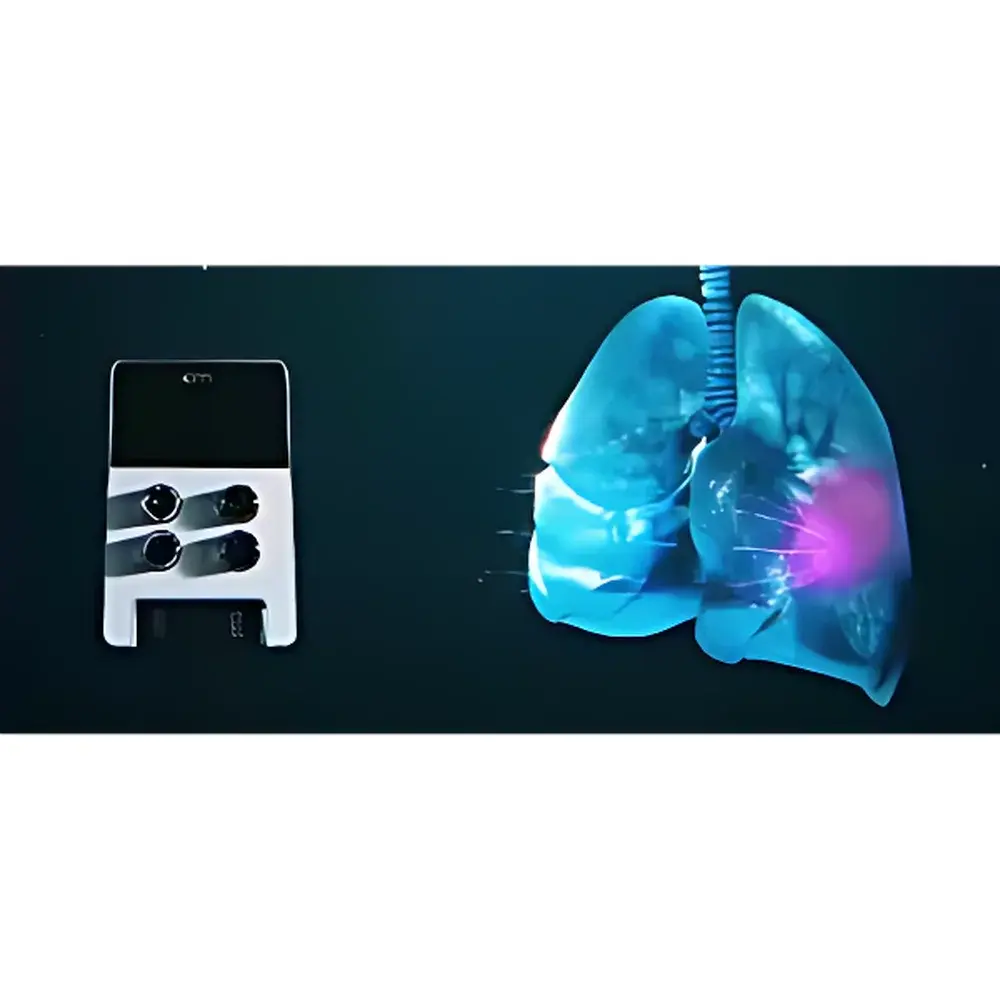
The Fluigent OMI is a compact, fully automated microfluidic organ-on-a-chip (OoC) perfusion platform engineered for precise, long-term, physiologically relevant cell culture under controlled incubator conditions. Unlike conventional benchtop perfusion systems, the OMI integrates pressure-driven flow control, multi-reservoir fluid handling, and embedded intelligence into a palm-sized (12 cm × 8 cm × 6 cm), incubator-compatible device. It operates on the principle of regulated pneumatic pressure modulation—leveraging Fluigent’s proprietary Flow-EZ™ microfluidic actuation technology—to deliver stable, pulseless, low-shear flow across organ chip microchannels. Its architecture eliminates external tubing, syringe pumps, and compressed air sources, enabling true “plug-and-perfuse” deployment inside standard 40 cm × 40 cm CO₂ incubators—up to six units simultaneously. The system is purpose-built for dynamic, multi-day experiments requiring reproducible shear stress, cyclic stretch mimicry (via programmable flow profiles), and inter-compartmental barrier modeling (e.g., blood–brain barrier, gut–vascular interface).
Key Features
- Palm-sized form factor (12 × 8 × 6 cm) and sub-800 g weight—designed for seamless transfer between incubator, microscope stage, and imaging rig without flow interruption
- Fully autonomous operation powered by integrated rechargeable lithium-ion battery (2 h runtime; hot-swappable in multi-unit configurations)
- Four independent 4 mL reservoirs per unit—enabling sequential or parallel delivery of media, drugs, cytokines, or wash buffers without manual intervention
- Multi-device orchestration: up to 12 OMI units can be synchronized via Fluigent’s CloudLink™ protocol for scalable, multi-organ system studies (e.g., liver–gut–kidney axis)
- Web-native control interface with real-time flow monitoring, historical log export (CSV/JSON), and customizable protocol scheduling (start/stop/pause/resume)
- On-device data encryption and automatic cloud synchronization (AWS-hosted, HIPAA- and GDPR-compliant infrastructure); optional local NAS integration
- No external gas supply or high-voltage connections required—complies with IEC 61010-1 safety standards for Class II laboratory equipment
Sample Compatibility & Compliance
The OMI platform is mechanically agnostic: its universal cartridge slot accepts industry-standard organ chips from Emulate, Mimetas, TissUse, AxoSys, and custom PDMS/glass/SU-8 devices (chip footprint ≤ 25 mm × 50 mm). It supports both open-channel and closed-loop recirculation modes—critical for metabolite accumulation studies (e.g., hepatic clearance assays) and barrier integrity assessment (TEER-compatible when paired with integrated electrodes). All fluidic pathways are biocompatible (USP Class VI-certified elastomers) and sterilizable via 70% ethanol or autoclave (20 min at 121 °C, non-battery modules only). The system supports ALARA (As Low As Reasonably Achievable) fluid volumes—minimizing reagent consumption while maintaining physiological flow Reynolds numbers (Re < 100) across capillary-scale channels. For regulatory submissions, OMI-generated datasets include timestamped metadata (flow rate, reservoir level, error flags), satisfying FDA 21 CFR Part 11 requirements for electronic records and signatures when configured with user authentication and audit trail enablement.
Software & Data Management
Fluigent’s OMI Control Suite (v3.2+) runs as a responsive web application (Chrome/Firefox/Safari), eliminating OS-specific installation. Protocols are defined using a drag-and-drop workflow editor supporting conditional logic (e.g., “if reservoir A drops below 0.5 mL, switch to reservoir B”), time-based ramping (linear/logarithmic flow gradients), and event-triggered sampling (e.g., “collect effluent at t = 24 h post-dose”). All raw sensor data (pressure, temperature, valve state) and derived metrics (cumulative volume, shear stress τ = 6μQ/πw h²) are stored with SHA-256 checksums. Export options include FAIR-aligned .jsonld manifests, MIAME-compliant metadata templates, and direct API access for integration with LIMS (LabVantage, Thermo Fisher SampleManager) or analysis platforms (Python/Pandas, MATLAB). Audit trails record every user action—including parameter edits, firmware updates, and emergency stop events—with immutable timestamps and IP addresses.
Applications
The OMI platform serves as a foundational infrastructure for next-generation in vitro models demanding spatiotemporal control and experimental scalability. Key use cases include: high-content ADME-Tox screening across human-relevant tissue barriers (BBB penetration, intestinal absorption, hepatobiliary transport); longitudinal tumor–stroma co-culture under perfused hypoxia; immune cell trafficking assays across endothelialized lung or lymph node chips; and multi-organ pharmacokinetic modeling (e.g., linking gut absorption to hepatic metabolism to renal excretion). Its low dead-volume design (95% inter-unit flow reproducibility over 72 h (n = 18 units, CV < 1.8%), supporting multicenter preclinical trials aligned with IQ Consortium guidelines.
FAQ
Can OMI be used inside a standard humidified CO₂ incubator?
Yes—its sealed electronics, passive thermal management, and absence of heat-generating components allow uninterrupted operation at 37 °C and 5% CO₂ for ≥7 days.
Does OMI support real-time TEER measurement?
Not natively—but it is electrically compatible with commercial TEER probes (e.g., ECIS, CellSens) via standardized electrode ports on supported organ chips.
How is calibration performed?
Factory-calibrated using NIST-traceable microflow standards; end-users may perform quick verification using gravimetric collection (±0.5 µL accuracy) via included calibration kit.
Is cloud data storage mandatory?
No—local operation with offline protocol execution and USB-C data dump is fully supported; cloud sync is opt-in and configurable per experiment.
What maintenance is required?
Annual firmware updates and biannual reservoir seal inspection; no consumables beyond standard organ chips and sterile tubing (optional for external reservoir extension).