FOUR E’S SCIENTIFIC Precipette™ Single-Channel Adjustable Volume Pipette
| Brand | FOUR E'S SCIENTIFIC |
|---|---|
| Origin | Guangdong, China |
| Manufacturer Type | OEM/ODM Manufacturer |
| Country of Origin | China |
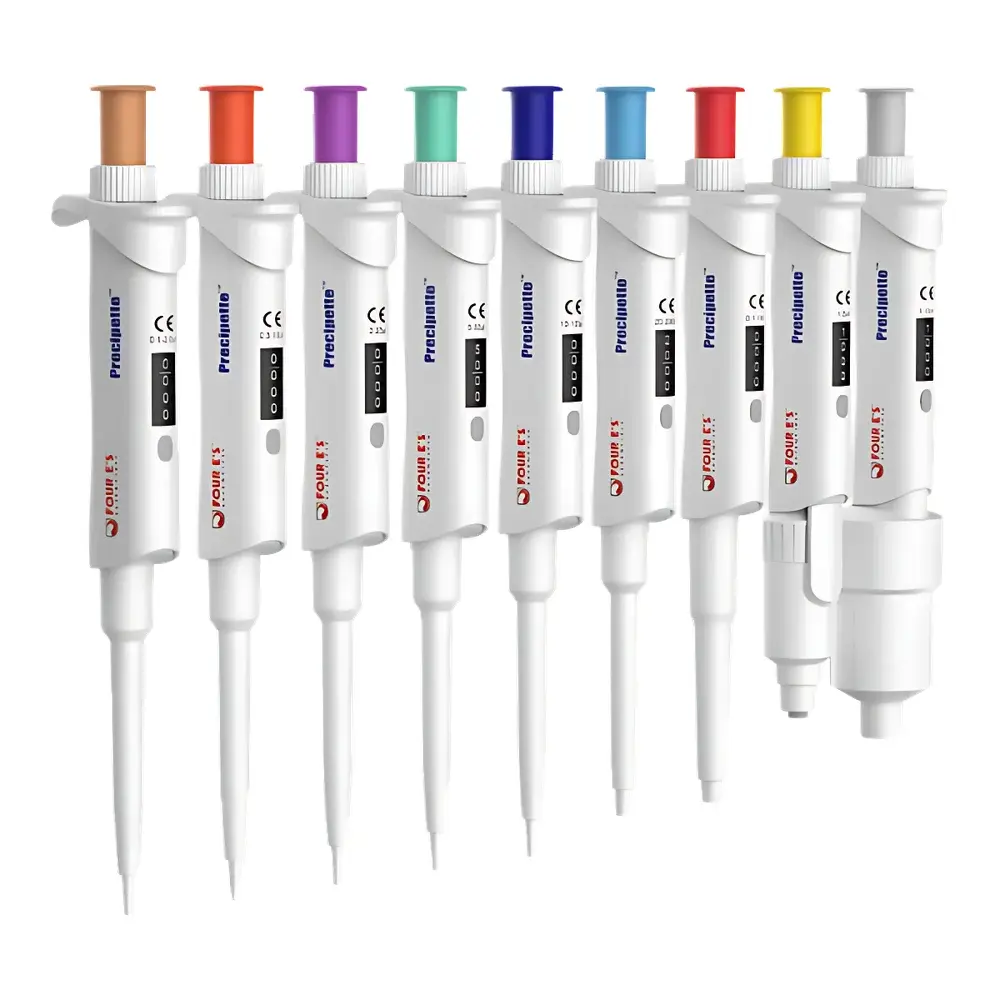
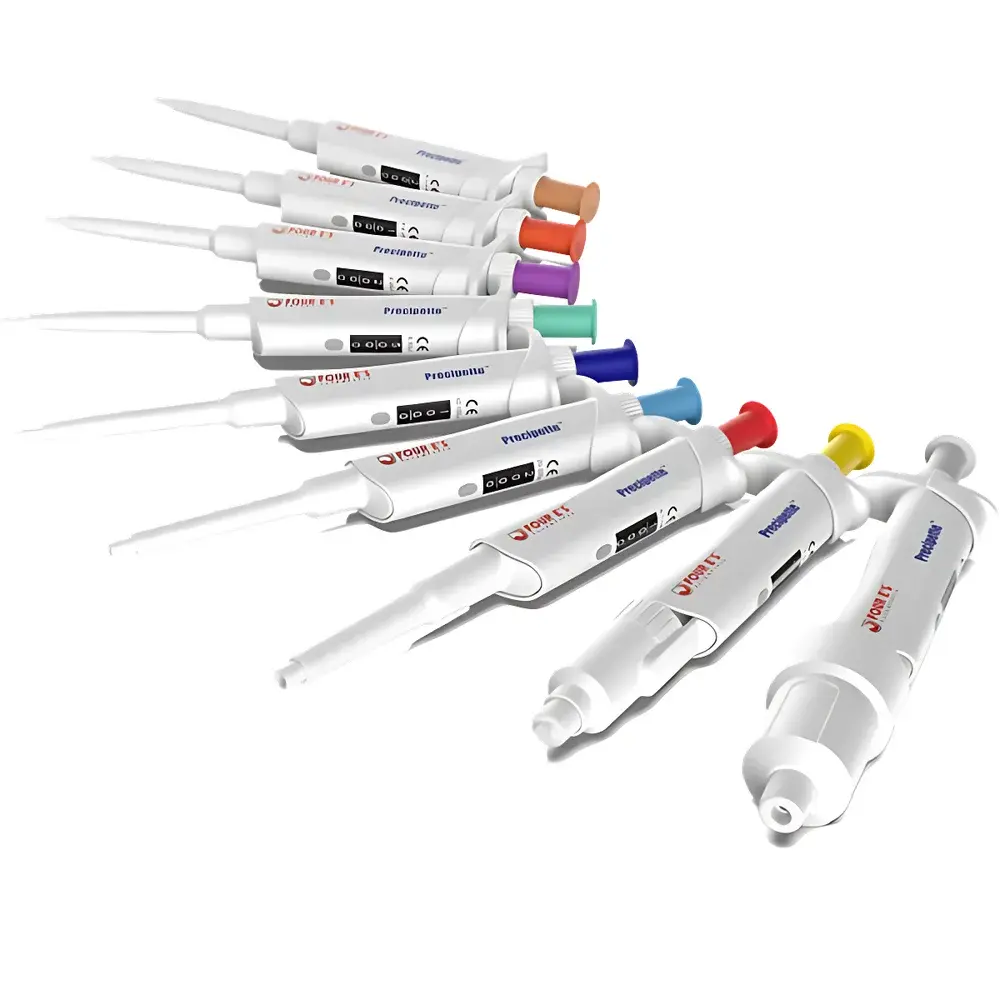
| Model Range | Preci2 / Preci10 / Preci20 / Preci50 / Preci100 / Preci200 / Preci1K / Preci5K / Preci10K |
| Pipette Type | Manual Single-Channel Adjustable Volume Pipette |
| Volume Range | 0.1 µL – 10 mL |
| Maximum Capacity | 10 mL |
| Accuracy (Max System Error) | ±0.60% (varies by model |
| Precision (Max Random Error) | ≤0.20% (model-dependent) |
| Calibration Standard | ISO 8655 compliant |
| Tip Compatibility | Universal fit for standard conical polypropylene tips (e.g., Gilson, Eppendorf, Rainin, Thermo Fisher) |
Overview
The FOUR E’S SCIENTIFIC Precipette™ Single-Channel Adjustable Volume Pipette is a precision-engineered manual liquid handling instrument designed for reproducible volumetric transfer in academic research laboratories, pharmaceutical quality control (QC), clinical diagnostics, and industrial R&D environments. Built upon the fundamental air displacement principle, the Precipette™ employs a tightly sealed piston-cylinder system where vertical plunger movement creates controlled negative pressure to aspirate and dispense liquids via disposable polypropylene tips. Its mechanical design adheres strictly to ISO 8655-1 (2022) and ISO 8655-2 (2022) standards for piston-operated volumetric apparatus, ensuring traceable accuracy and long-term metrological integrity across nine calibrated volume ranges—from ultra-low-volume 0.1–2.0 µL (Preci2) to high-capacity 1–10 mL (Preci10K). Each unit undergoes individual gravimetric calibration at three points (low, mid, and high volume) under controlled temperature (20–25 °C) and humidity (40–60% RH), with full calibration certificates provided per ISO/IEC 17025 requirements.
Key Features
- Ergonomic lightweight housing with balanced center-of-gravity design reduces thumb fatigue during repetitive pipetting cycles—validated through ISO 13485-aligned human factors assessment.
- Two-stage tip ejection mechanism: soft first-stage for gentle tip release; firm second-stage for secure disposal without aerosol generation or finger contact contamination.
- Tool-free disassembly: upper body, piston assembly, and tip cone separate in <15 seconds using only the included hex key—enabling full internal cleaning, lubrication, and visual inspection per GLP maintenance logs.
- Chemically resistant tip cone and O-ring materials (FKM fluoroelastomer and PTFE-coated stainless steel) withstand exposure to alcohols, acetone, dilute acids/bases, and common organic solvents.
- Color-coded upper housings (brown to gray) provide immediate visual identification of nominal volume range—supporting error-proofing in multi-pipette workflows and ISO 15190-compliant lab zoning protocols.
- Integrated dual-stop plunger travel: primary stop for forward dispensing; secondary stop for blow-out mode—ensuring complete delivery of viscous or volatile samples without over-pressurization.
Sample Compatibility & Compliance
The Precipette™ series accommodates aqueous solutions, buffers, cell suspensions, serum, ethanol/water mixtures, and low-viscosity reagents up to 10 mPa·s. It is not recommended for highly viscous (e.g., glycerol >30%), corrosive (e.g., concentrated HCl, HF), or volatile (e.g., chloroform, ether) liquids without protocol validation. All models comply with ISO 8655-1 (General Requirements), ISO 8655-2 (Specifications for Air Displacement Pipettes), and ISO 8655-5 (Testing and Calibration Methods). Units are supplied with CE marking per EU Directive 2014/30/EU (EMC) and 2014/35/EU (LVD), and meet RoHS 2011/65/EU substance restrictions. Traceable calibration data supports FDA 21 CFR Part 11 readiness when paired with FOUR E’S SCIENTIFIC’s optional electronic calibration logbook software.
Software & Data Management
While the Precipette™ is a fully manual device, its calibration documentation integrates seamlessly into digital laboratory quality systems. Each pipette ships with a unique serial number-linked calibration certificate (PDF + CSV), containing gravimetric test results, uncertainty budgets (k=2), environmental conditions, and technician signatures. Optional FOUR E’S SCIENTIFIC PipetteTrack™ cloud module enables centralized inventory management, scheduled recalibration alerts, audit trail generation (aligned with ALCOA+ principles), and export to LIMS or ELN platforms. All calibration records retain full metadata for GLP/GMP inspections and ISO/IEC 17025 accreditation audits.
Applications
- Quantitative PCR (qPCR) setup requiring sub-microliter precision in master mix preparation.
- Cell culture passaging and viability assays where consistent seeding density depends on accurate 10–100 µL transfers.
- Pharmaceutical dissolution testing per USP <711>, including standard solution dilution across 0.5–1000 µL ranges.
- Environmental water analysis (EPA Method 525.3) involving SPE cartridge elution and LC-MS/MS sample prep.
- Diagnostic assay development under CLIA and CAP guidelines, where inter-operator variability must remain <1.5% CV.
- High-throughput screening (HTS) plate replication where tip-to-tip consistency across multiple Preci200 units ensures assay robustness.
FAQ
Is the Precipette™ compatible with third-party pipette tips?
Yes—its conical tip interface conforms to ISO 8655-3 dimensional tolerances and achieves leak-tight seals with all major global tip brands (e.g., Axygen, Sarstedt, VWR, Bio-Rad) tested per ISO 8655-6 tip compatibility protocols.
What is the recalibration interval recommendation?
FOUR E’S SCIENTIFIC recommends recalibration every 6 months for routine use, or after 10,000 actuations—whichever occurs first—as defined in ISO 8655-5 Annex C. More frequent verification is advised for critical GxP applications.
Can the Precipette™ be autoclaved?
No—the entire unit is not autoclavable. Only the tip cone assembly (excluding piston and spring) may be sterilized via dry heat (160 °C, 2 h) or ethylene oxide (EtO); full disassembly and re-lubrication with ISO 6743-17 Class HXES synthetic grease is required post-sterilization.
Does it support reverse pipetting?
Yes—reverse pipetting mode is enabled by aspirating 20–30% above the set volume and dispensing only to the first stop, minimizing residual volume errors for proteins, glycerol solutions, or surfactant-containing buffers.
How is traceability maintained across calibration events?
Each recalibration generates a new certificate with updated uncertainty values, reference standard IDs (traceable to NIM, China), and digital signature—stored in PipetteTrack™ with immutable timestamps and user-access logs compliant with 21 CFR Part 11 §11.10(a).