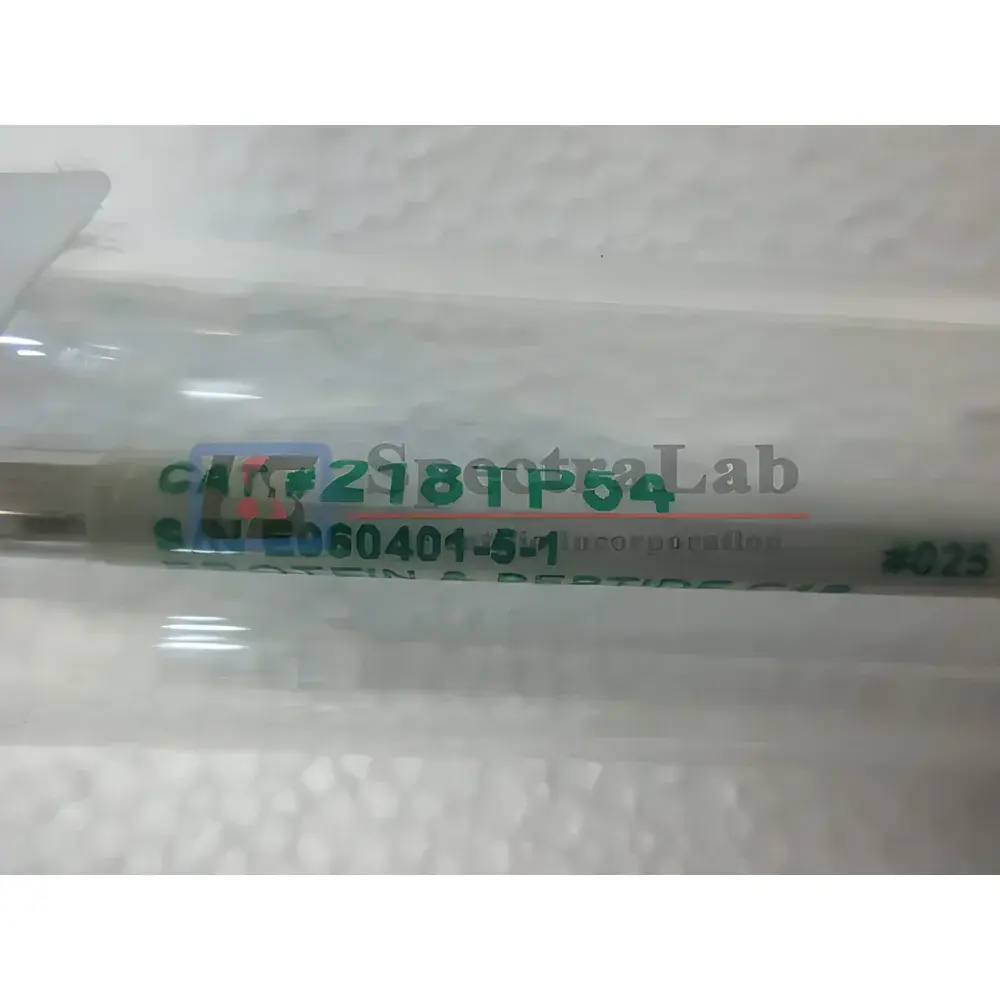
Grace Vydac Protein & Peptide C18 HPLC Column
| Origin | Canada |
|---|---|
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Protein & Peptide C18 |
| Price Range | USD 150–7,500 (FOB) |
| Product Category | Chromatography Column |
| Service Life | 4–5 years |
| Warranty | 6 months |
| Part Number | 218TP54 |
Overview
The Grace Vydac Protein & Peptide C18 HPLC Column is a high-performance reversed-phase analytical column engineered for the robust and reproducible separation of intact proteins, large peptides, and hydrophobic biomolecules under denaturing or semi-denaturing conditions. Built on Vydac’s proprietary wide-pore (300 Å) silica support with a fully endcapped C18 ligand, this column delivers superior peak shape, resolution, and recovery for analytes ranging from 1 kDa to >150 kDa. Its design adheres to established chromatographic principles for biomolecule analysis—leveraging hydrophobic interaction chromatography (HIC) in conjunction with gradient elution using aqueous/organic mobile phases (typically water/acetonitrile with 0.1% trifluoroacetic acid). The column is compatible with standard high-pressure liquid chromatography systems operating up to 400 bar and supports both analytical and semi-preparative applications in QC, process development, and structural biology workflows.
Key Features
- Wide-pore (300 Å) high-purity silica base matrix optimized for macromolecular diffusion and reduced steric hindrance
- Densely bonded, fully endcapped C18 phase ensuring high surface coverage and minimized silanol activity
- Batch-tested for lot-to-lot consistency in retention time, resolution (Rs ≥ 1.5 for insulin A/B chain pair), and column efficiency (N ≥ 35,000 plates/m)
- Validated for use with common denaturants including 0.1% TFA, 0.1% formic acid, and up to 20% isopropanol in mobile phase
- Robust mechanical stability—rated for repeated injections (>2,000 injections) under standard gradient conditions at flow rates ≤1.0 mL/min
- Pre-conditioned and performance-verified prior to resale; includes certificate of inspection with backpressure, asymmetry factor (As), and resolution test data
Sample Compatibility & Compliance
This column demonstrates consistent performance across diverse protein classes—including monoclonal antibodies (mAbs), antibody-drug conjugates (ADCs), recombinant enzymes, and synthetic peptide libraries. It complies with method requirements outlined in USP , ICH Q5E, and ASTM D8078 for biomolecular purity assessment. While not supplied with full GMP documentation by default, the column supports GLP-compliant workflows when integrated into validated HPLC systems with audit-trail-enabled software (e.g., Waters Empower, Thermo Chromeleon). All units undergo post-refurbishment testing per internal QA protocol aligned with ISO/IEC 17025 calibration traceability principles for column performance verification.
Software & Data Management
No embedded firmware or proprietary software is associated with the column itself; however, its performance metrics are fully integrable into industry-standard chromatography data systems (CDS). Retention time stability, peak symmetry, and resolution values are directly reportable in compliance-ready formats supporting FDA 21 CFR Part 11 requirements when used with validated CDS configurations. Spectralab Scientific provides optional post-purchase technical documentation—including method transfer templates, system suitability criteria checklists, and column regeneration protocols—for seamless integration into regulated environments.
Applications
- Intact mass analysis and purity profiling of therapeutic proteins and biosimilars
- Peptide mapping support (pre- or post-enzymatic digestion) in structural characterization studies
- Stability-indicating assays for forced degradation studies (thermal, oxidative, acidic)
- Process intermediate monitoring during purification campaigns (e.g., eluate pool analysis)
- Quality control release testing per pharmacopeial monographs (e.g., EP 2.2.46, USP )
- Support for LC-MS coupling where desolvation efficiency and ion suppression minimization are critical
FAQ
Is this column suitable for use with aqueous mobile phases only?
No—this column requires organic modifiers (typically acetonitrile or methanol) in combination with ion-pairing agents (e.g., TFA) for effective protein/peptide elution. Pure aqueous conditions result in irreversible adsorption and loss of resolution.
What is the recommended storage solvent after use?
Store in ≥80% acetonitrile or isopropanol with 0.1% TFA at ambient temperature. Avoid prolonged exposure to pure water or high-pH buffers (>7.5).
Can this column be regenerated after performance decline?
Yes—standard regeneration protocols involving stepwise flushing with increasing organic strength (water → 20% ACN → 50% ACN → 100% ACN → 100% IPA) are effective for restoring backpressure and peak shape, provided no covalent fouling has occurred.
Does the 6-month warranty cover performance drift under routine use?
Yes—the warranty includes re-testing and replacement if resolution, asymmetry, or backpressure deviates beyond ±10% of initial certified values under documented, manufacturer-recommended operating conditions.
Are lot-specific chromatograms and test reports available upon request?
Yes—Spectralab Scientific provides digital copies of the original QC chromatogram, system suitability summary, and batch-specific retention time data for each unit shipped.