Heguan HGK-81 Sulfur Dioxide Residue Analyzer for Food and Pharmaceutical Applications
| Brand | Heguan |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Domestic (China) |
| Model | HGK-81 |
| Pricing Range | USD 9,500 – 15,200 (FOB Shanghai) |
| Product Type | Intelligent Automated SO₂ Distillation Analyzer |
| Detection Limit | RSD ≤ 5% (repeatability), Parallel Difference ≤ 10% (with 5 mL standard alkali consumption) |
| Detection Range | 0–50 mg SO₂ per sample |
Overview
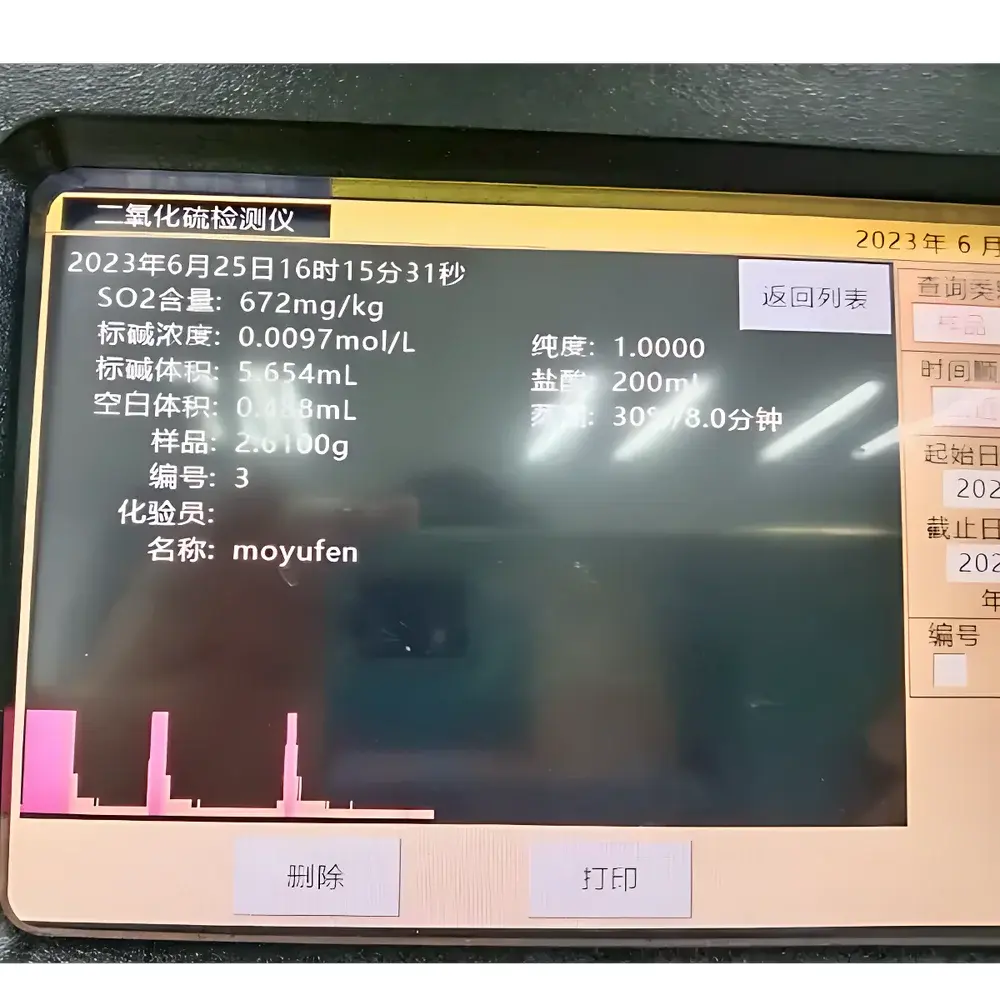
The Heguan HGK-81 Sulfur Dioxide Residue Analyzer is an automated, micro-distillation-based analytical instrument engineered for precise quantification of sulfur dioxide (SO₂) residues in food, herbal medicine, and pharmaceutical raw materials. It implements the official titrimetric distillation method specified in GB 5009.34–2022 (National Food Safety Standard for Determination of Sulfur Dioxide in Foods) and aligns with relevant monographs in the Chinese Pharmacopoeia (ChP). The system operates on the principle of acid liberation followed by steam distillation under controlled conditions, where liberated SO₂ is selectively trapped in a hydrogen peroxide or formaldehyde absorption solution and subsequently titrated with standardized iodine solution. Designed for routine QC laboratories in food safety testing centers, GMP-compliant pharmaceutical manufacturers, and herbal product quality control units, the HGK-81 delivers high reproducibility, operator safety, and method compliance without requiring manual intervention during critical distillation steps.
Key Features
- 7-inch full-color capacitive touchscreen interface with embedded operational guidance, multilingual support (English/Chinese), and voice-prompted workflow navigation
- Robust housing fabricated from high-impact, chemically resistant ABS engineering plastic—certified for sustained exposure to concentrated HCl, H₃PO₄, and alkaline reagents
- Custom-specified high-temperature, acid/alkali-resistant silicone tubing (supplied exclusively by Heguan) used throughout all fluidic pathways—rated for >36 months continuous service life under routine laboratory conditions
- Volumetric precision dosing via calibrated peristaltic pumps for acid, distilled water, and absorbent solution—eliminating volumetric error from manual pipetting or graduated cylinder use
- Integrated 750 mL borosilicate glass distillation flask with optimized vapor path geometry to minimize SO₂ adsorption loss and ensure quantitative recovery
- Pressure-free, low-noise liquid displacement pump system enabling fully automated sequence control: acid addition → water replenishment → absorption solution delivery → steam distillation initiation → endpoint detection
- User-configurable distillation time (0–99 min) and reagent volume parameters; real-time level sensing with auto-compensation for evaporation losses during extended runs
- Programmable post-run cleaning cycle using hot water and dilute acid rinse—reducing carryover risk between samples and supporting GLP-aligned documentation practices
- Comprehensive hardware interlock system: automatic shutdown upon low-level detection of acid, absorbent, or coolant; thermal cutoff at distillation head; overpressure relief valve
- Standardized 8 mm OD nitrogen inlet port compatible with optional on-site nitrogen generators (500–2000 mL/min flow rate) for inert atmosphere-assisted distillation
- Adjustable heating power (0–1500 W) with PID-controlled temperature regulation to maintain consistent steam generation across varying ambient and load conditions
- Heguan Smart-Cool™ feature: condenser water supply automatically activates only during active distillation and deactivates upon completion—reducing lab utility consumption by ~65% versus continuous-flow systems
Sample Compatibility & Compliance
The HGK-81 accommodates solid matrices up to 100 g (e.g., dried fruits, medicinal herbs, starch-based additives) and liquid samples up to 100 mL (e.g., wine, fruit juices, herbal extracts). Its validated performance meets the repeatability (RSD ≤ 5%) and recovery (>85%) criteria stipulated in GB 5009.34–2022. Instrument operation supports audit-ready workflows compliant with ISO/IEC 17025:2017 requirements for testing laboratories. Data integrity safeguards—including electronic logbooks, user access levels, and timestamped parameter changes—are configurable to meet FDA 21 CFR Part 11 readiness expectations when paired with Heguan’s optional LIMS-integrated software module.
Software & Data Management
The embedded operating system stores up to 1,000 customizable distillation protocols, each retaining complete metadata: date/time stamp, operator ID, sample ID, reagent lot numbers, distillation duration, power profile, and final titration volume. All method parameters are password-protected and version-controlled. Raw data export is supported via USB 2.0 to CSV or PDF formats. Optional Heguan Analytical Suite v3.2 provides advanced features including trend analysis across batch runs, calibration curve management, outlier detection algorithms, and automated report generation aligned with ISO 10576-1 statistical guidelines for analytical uncertainty estimation.
Applications
- Quantitative SO₂ residue testing in sulfited dried fruits (apricots, raisins), preserved vegetables, and candied ginger per GB 5009.34–2022
- Residual sulfite verification in traditional Chinese medicinal materials (e.g., Paeonia lactiflora, Glycyrrhiza uralensis) prior to formulation
- Wine and cider SO₂ monitoring for compliance with OIV and EU Regulation No. 1308/2013 labeling thresholds
- Pharmaceutical excipient screening for SO₂ contamination in starches, lactose, and carrageenan derivatives
- Method validation studies supporting ICH Q2(R2) requirements for specificity, linearity, accuracy, and robustness
- Reference laboratory participation in proficiency testing schemes coordinated by CNAS or FAPAS
FAQ
What regulatory standards does the HGK-81 directly support?
It is validated for GB 5009.34–2022 and referenced in ChP 2020 General Chapter 0832. Method equivalence to AOAC 990.28 and ISO 5776:1991 can be demonstrated through laboratory-specific validation.
Can the HGK-81 be integrated into a LIMS environment?
Yes—via optional RS-232/USB-to-Ethernet gateway and Heguan DataBridge middleware, enabling bidirectional transfer of sample IDs, results, and instrument status logs.
Is third-party certification available for IQ/OQ/PQ protocols?
Heguan provides factory-issued Installation Qualification (IQ) and Operational Qualification (OQ) documentation templates; Performance Qualification (PQ) must be executed onsite by the end-user per ISO/IEC 17025 Clause 7.7.
What maintenance intervals are recommended?
Quarterly verification of pump calibration and tubing integrity; annual replacement of heating element gaskets and condenser O-rings; no scheduled replacement of core fluidic tubing required within first 36 months under normal usage.
Does the system support dual-titration modes (iodometric and para-rosaniline)?
The base configuration supports iodometric titration only. Para-rosaniline colorimetric mode requires optional UV-Vis spectrophotometer integration and separate reagent kit (HGK-SO₂-COLOR).