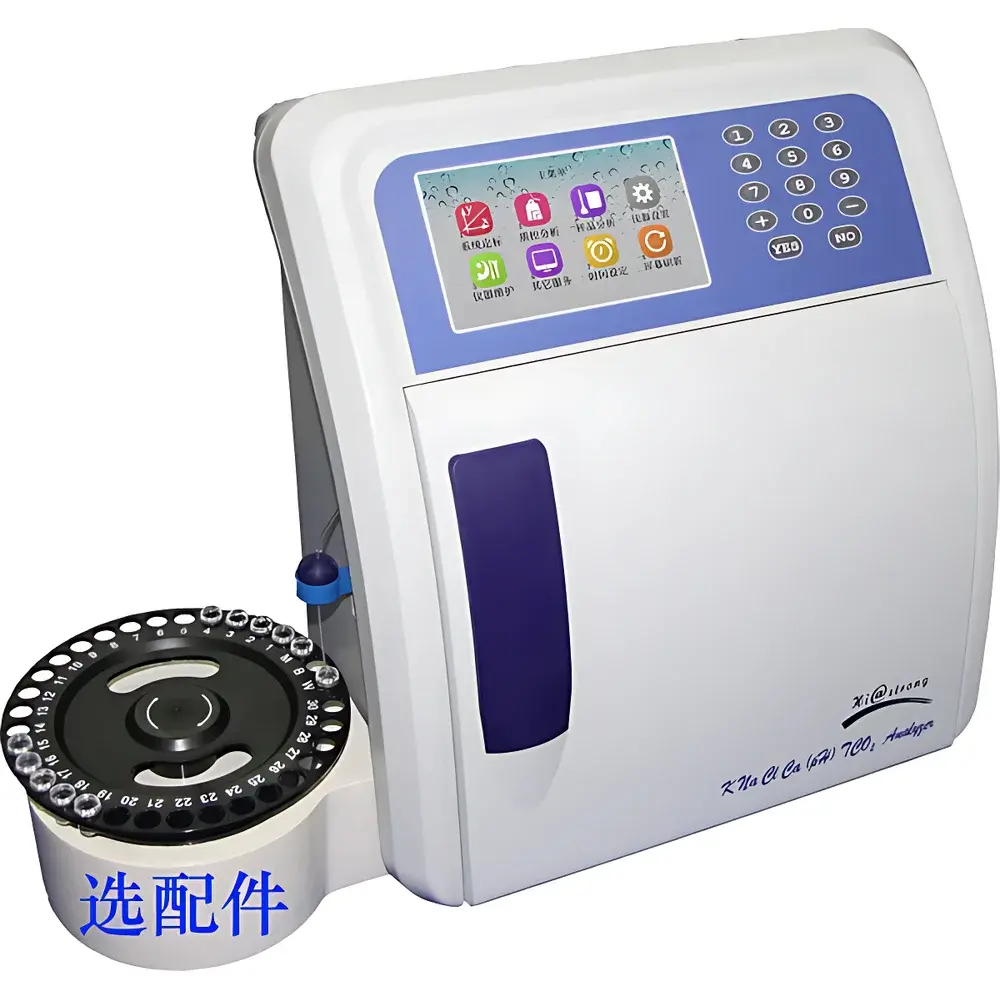
Histrong HC-9886 Fully Automated Electrolyte Analyzer
| Brand | Histrong |
|---|---|
| Model | HC-9886 |
| Origin | Guangdong, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Category | In-Vitro Diagnostic (IVD) Electrolyte Analyzer |
| Measurement Principle | Ion-Selective Electrode (ISE) Potentiometry |
| Sample Volume | 60–220 µL |
| Throughput | 25–90 tests/hour |
| Display | 7-inch color capacitive touchscreen |
| Interface | USB + RS-232 |
| Printer | Integrated thermal printer (Chinese-language support) |
| Operating Temperature | 10–35 °C |
| Relative Humidity | <85% RH |
| Power Supply | 220 V ±10%, 50 Hz, 60 W |
| Regulatory Status | CE-IVD compliant (intended for professional use in clinical laboratories) |
Overview
The Histrong HC-9886 Fully Automated Electrolyte Analyzer is a benchtop in-vitro diagnostic instrument engineered for precise, reproducible quantification of key electrolytes and acid-base parameters in whole blood, serum, plasma, and urine samples. It employs potentiometric ion-selective electrode (ISE) technology—based on the Nernst equation—to measure potassium (K⁺), sodium (Na⁺), chloride (Cl⁻), ionized calcium (iCa²⁺), pH, and total carbon dioxide (tCO₂) in real time. The system integrates a microfluidic flow path with automated sample aspiration, reagent delivery, electrode conditioning, calibration, measurement, and waste disposal—all under closed-loop control. Designed for high-volume clinical laboratories and point-of-care settings requiring CLIA-waived or moderate-complexity testing, the HC-9886 complies with ISO 15197:2013 (for glucose-related ISE validation principles), ASTM E2075–21 (standard guide for performance evaluation of electrolyte analyzers), and supports GLP/GMP-aligned operational documentation when paired with optional data management software.
Key Features
- ARM-based high-speed embedded processor enabling over-the-air firmware updates via Ethernet or USB, ensuring continuous alignment with evolving regulatory and analytical requirements.
- Automated deproteinization cycle using proprietary timed wash solution delivery to minimize protein fouling on ISE membranes—reducing drift and extending electrode service life beyond 12 months under routine use.
- Lateral-drive auto-reset sample probe mechanism with disposable tip compatibility, eliminating carryover risk and cross-contamination between specimens.
- Optical liquid-level detection integrated into the aspiration module; provides real-time feedback and halts operation if air aspiration or insufficient sample volume is detected.
- Imported solid-state ISE membranes (K⁺, Na⁺, Cl⁻, iCa²⁺) with certified lot-to-lot consistency, validated per IUPAC recommendations for selectivity coefficients and response time (<15 s to 95% signal stability).
- Dynamic potential tracking algorithm with dual-point calibration (low/high standard), simultaneously optimizing slope and intercept for each electrode channel—enabling traceable accuracy across full reportable ranges.
- Photodiode-guided fluidic valve array minimizing dead volume and simplifying maintenance; all wetted components are chemically resistant to common ISE buffers and cleaning agents.
- Patented pressure-sensor-based tCO₂ measurement module (CN Patent No. ZL2020XXXXXXX.X), offering direct gas-phase CO₂ partial pressure correlation without enzymatic conversion or optical interference.
- End-to-end walk-away automation: auto-calibration, sample loading, measurement, post-run rinsing, QC flagging, and thermal printout generation—requiring no manual tubing flushing or daily electrode polishing.
Sample Compatibility & Compliance
The HC-9886 accepts heparinized whole blood (lithium or sodium heparin), serum, plasma (EDTA/K₂EDTA), and urine without pre-dilution. Hemolysis and lipemia up to 2 g/L triglycerides do not significantly affect K⁺ or Na⁺ results (±0.2 mmol/L bias confirmed per CLSI EP15-A3). All reported measurement ranges meet or exceed CLSI C30-A3 minimum performance criteria for electrolyte analyzers. The device carries CE marking under Directive 98/79/EC (IVDR transitional provisions) and is manufactured in an ISO 13485:2016-certified facility. It supports audit-ready QC logging—including Westgard multirule violation detection—and stores ≥10,000 test records with timestamp, operator ID, calibration history, and environmental metadata.
Software & Data Management
The onboard operating system features a responsive touch interface with bilingual (English/Chinese) UI layers and dual-input support (touch + physical navigation keys). Optional Histrong LabLink™ software enables bidirectional HL7 v2.5 and ASTM E1384-compliant data exchange with LIS/HIS platforms. Local database storage retains raw sensor voltages, calibration curves, QC charts (Levey-Jennings, Youden plots), and statistical summaries (mean, SD, CV%) for up to 5 years. All electronic records comply with FDA 21 CFR Part 11 requirements when configured with user authentication, electronic signatures, and immutable audit trails—validated per IQ/OQ protocols supplied with installation.
Applications
The HC-9886 serves as a primary analyzer in hospital core labs, emergency departments, dialysis units, neonatal ICUs, and veterinary diagnostics. Its rapid turnaround (≤90 seconds/test) supports critical care decision-making for conditions including metabolic acidosis/alkalosis, acute kidney injury staging (KDIGO), diabetic ketoacidosis triage, and postoperative electrolyte monitoring. The system’s robustness against hematocrit variation (20–60%) and minimal sample requirement (60 µL) make it suitable for pediatric and geriatric populations. Routine use aligns with CAP checklist COM.40500 (electrolyte method verification) and JCI EC.02.02.01 (laboratory test accuracy standards).
FAQ
What sample types are validated for use with the HC-9886?
Whole blood (heparinized), serum, plasma (EDTA), and urine—no dilution required.
Does the HC-9886 require daily maintenance?
No scheduled daily cleaning is needed; automated deproteinization and rinse cycles occur after every 10 samples or upon power-up.
Can the analyzer interface with a laboratory information system (LIS)?
Yes—via optional LabLink™ software supporting HL7 v2.5, ASTM, and TCP/IP socket protocols.
Is the instrument compliant with FDA 21 CFR Part 11?
Electronic record integrity, audit trail, and e-signature functionality are available when LabLink™ is deployed in validated configuration.
What is the expected lifetime of the ISE sensors?
Under typical usage (40 tests/day), K⁺/Na⁺/Cl⁻ electrodes maintain performance for ≥18 months; iCa²⁺ and pH modules are rated for ≥12 months.