Huan Kai Smartcounter-4K Automated Colony Counter
| Brand | Huan Kai (BHK) |
|---|---|
| Origin | Guangdong, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | Smartcounter-4K |
| Instrument Type | Fully Automated Colony Counter |
| Minimum Detectable Colony Size | 0.2 mm |
| Camera Resolution | 8.29 MP |
| Maximum Petri Dish Diameter | <150 mm |
| Power Supply | AC 100–240 V, 50 Hz |
| Dimensions (W×D×H) | 290 × 260 × 380 mm |
| Software Language | English & Chinese |
Overview
The Huan Kai Smartcounter-4K Automated Colony Counter is an optical image-based microbiological enumeration system engineered for precision, reproducibility, and regulatory compliance in routine and high-throughput microbial testing environments. It operates on the principle of high-resolution digital imaging combined with adaptive thresholding and morphological segmentation algorithms to detect, classify, and quantify microbial colonies on solid agar media. Unlike manual or semi-automated counters reliant on fixed lighting or single-spectrum illumination, the Smartcounter-4K employs dual-axis, spectrally tunable LED illumination (top and bottom) and a distortion-corrected 4K color imaging module to ensure consistent contrast across diverse media types—including chromogenic, selective, and opaque formulations—thereby minimizing false negatives and overcounting artifacts. Designed for laboratories adhering to ISO 4833-1:2013, ISO 7218, AOAC Official Methods of Analysis®, and USP <61>/<62>, the instrument supports quantitative microbiological analysis in food safety, pharmaceutical quality control, clinical diagnostics, cosmetics validation, and environmental monitoring workflows.
Key Features
- True 4K color CMOS imaging sensor (8.29 megapixels) with ultra-low-distortion optics for sub-millimeter colony resolution down to 0.2 mm.
- Dual 360° adjustable LED illumination system: independent top-light intensity control and bottom-light spectral tuning (color temperature + brightness) to optimize contrast on differential media (e.g., MacConkey, CHROMagar™, R2A).
- Open-platform benchtop design with ambient light rejection—no enclosure required; accommodates standard 90 mm, 100 mm, and 150 mm Petri dishes without repositioning.
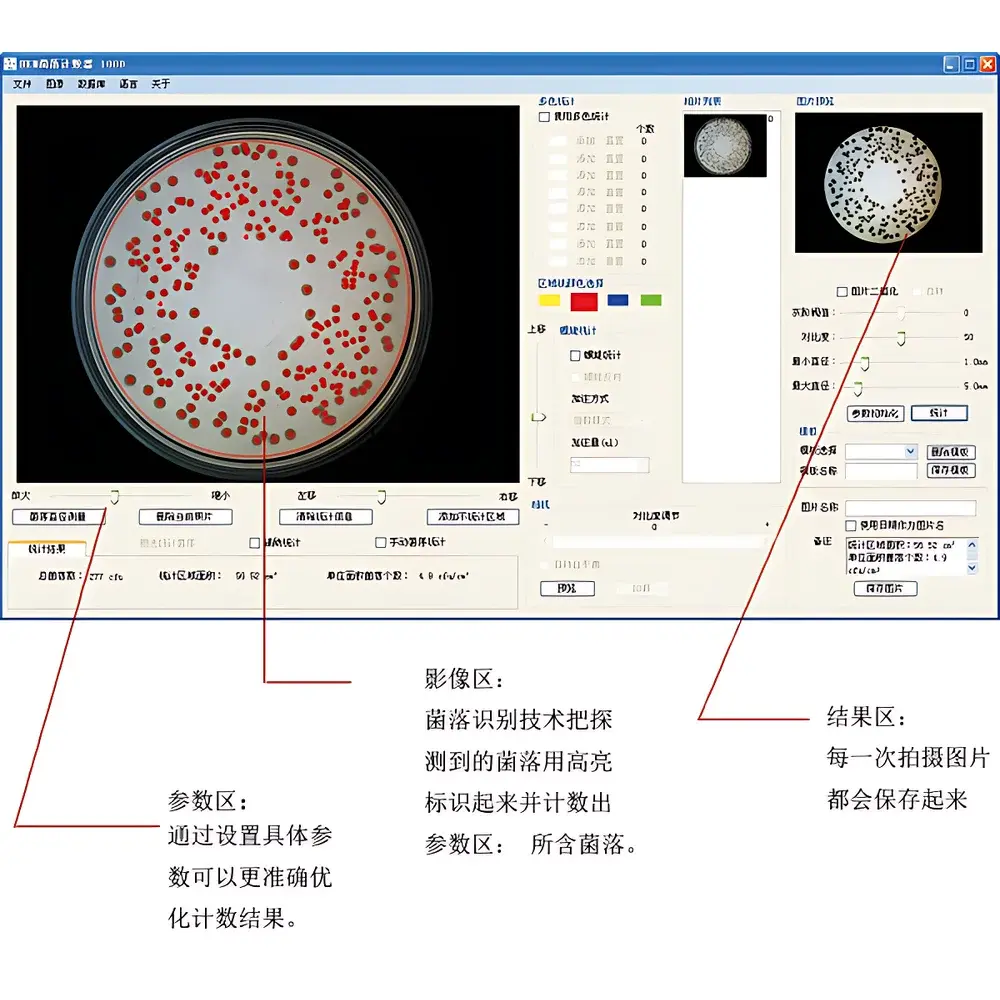
- Real-time parameter adjustment via intuitive GUI: drag-and-drop threshold sliders, dynamic preview of segmentation outcomes, and immediate visual feedback during optimization.
- Multi-mode detection engine supporting standard pour plates, spread plates, spiral plating (ISO 7218 Annex B), membrane filtration grids (including Millipore® and Sartorius® grid membranes), and inhibition zone analysis (for antimicrobial susceptibility screening).
- Built-in anti-pen-mark interference algorithm—automatically excludes hand-drawn annotations without user-defined exclusion zones.
- Intelligent colony splitting logic for clustered or confluent growth using watershed-based separation and shape-based boundary refinement.
- Multi-color classification capability: distinguishes up to eight discrete colony hues simultaneously to support chromogenic assay interpretation (e.g., E. coli vs. Enterococcus on CHROMagar Orientation).
Sample Compatibility & Compliance
The Smartcounter-4K accepts all standard non-fluorescent, non-bioluminescent agar-based culture media used in regulatory-compliant microbiology labs. It has been validated for use with ISO-standardized methods including ISO 4833-1 (aerobic colony count), ISO 6579 (Salmonella), ISO 16649-2 (E. coli), and ISO 11133 (culture medium performance testing). Its audit trail functionality—comprising timestamped operator logins, parameter change history, image metadata embedding (EXIF + custom tags), and immutable result archiving—meets GLP and GMP documentation requirements under FDA 21 CFR Part 11 when configured with optional role-based access control (RBAC). All stored data retain full traceability to original plate images, acquisition settings, and analyst identity—ensuring defensible data integrity for internal audits or regulatory inspections.
Software & Data Management
The embedded Windows-native software requires no driver installation or administrative privileges; it runs portably from USB storage or network share across Windows 10/11 (32- and 64-bit). Data export formats include CSV (with per-colony coordinates, size, circularity, and color index), PDF reports (with embedded thumbnails and statistical summaries), and DICOM-compatible TIFF stacks for long-term archival. The system maintains a local SQLite database with encrypted audit logs tracking every action: login/logout events, parameter modifications, manual corrections, and report generation. Optional RBAC enables three-tier permission levels (Administrator, Supervisor, Technician) to enforce procedural segregation—e.g., restricting method editing to QA personnel while allowing QC analysts to execute only pre-approved protocols.
Applications
This instrument is routinely deployed in accredited food testing laboratories for total viable counts (TVC), coliforms, yeasts/molds, and pathogen confirmation assays; in pharmaceutical cleanroom monitoring programs per EU GMP Annex 1 and USP <1116>; in hospital infection control units for environmental surface swab enumeration; and in cosmetic challenge testing per ISO 11930. Its ability to process spiral plates at calibrated dilution factors makes it suitable for ISO 7218 Annex B-compliant enumeration without serial dilution steps. In research settings, its high-fidelity image capture and batch-processing capability support longitudinal studies requiring colony morphology trend analysis across time-series incubations.
FAQ
Does the Smartcounter-4K comply with FDA 21 CFR Part 11 for electronic records?
Yes—when deployed with optional RBAC and enabled audit trail logging, it satisfies electronic signature and record retention requirements for regulated environments.
Can it process membrane filter grids from different manufacturers?
Yes—the system recognizes standardized grid patterns (e.g., 25-, 47-, and 50-mm diameter filters) and supports user-defined grid templates for proprietary formats.
Is calibration required before each use?
No routine hardware calibration is needed; however, daily verification using NIST-traceable colony size reference standards (e.g., polystyrene microsphere arrays) is recommended per ISO/IEC 17025.
What file formats are supported for data export?
CSV (tabular colony metrics), PDF (formatted reports), TIFF (lossless image archives), and XML (structured metadata for LIMS integration).
How does the system handle overlapping or irregularly shaped colonies?
It applies multi-scale edge detection followed by seeded region growing and Voronoi-based partitioning to resolve adjacency ambiguities while preserving morphological fidelity.