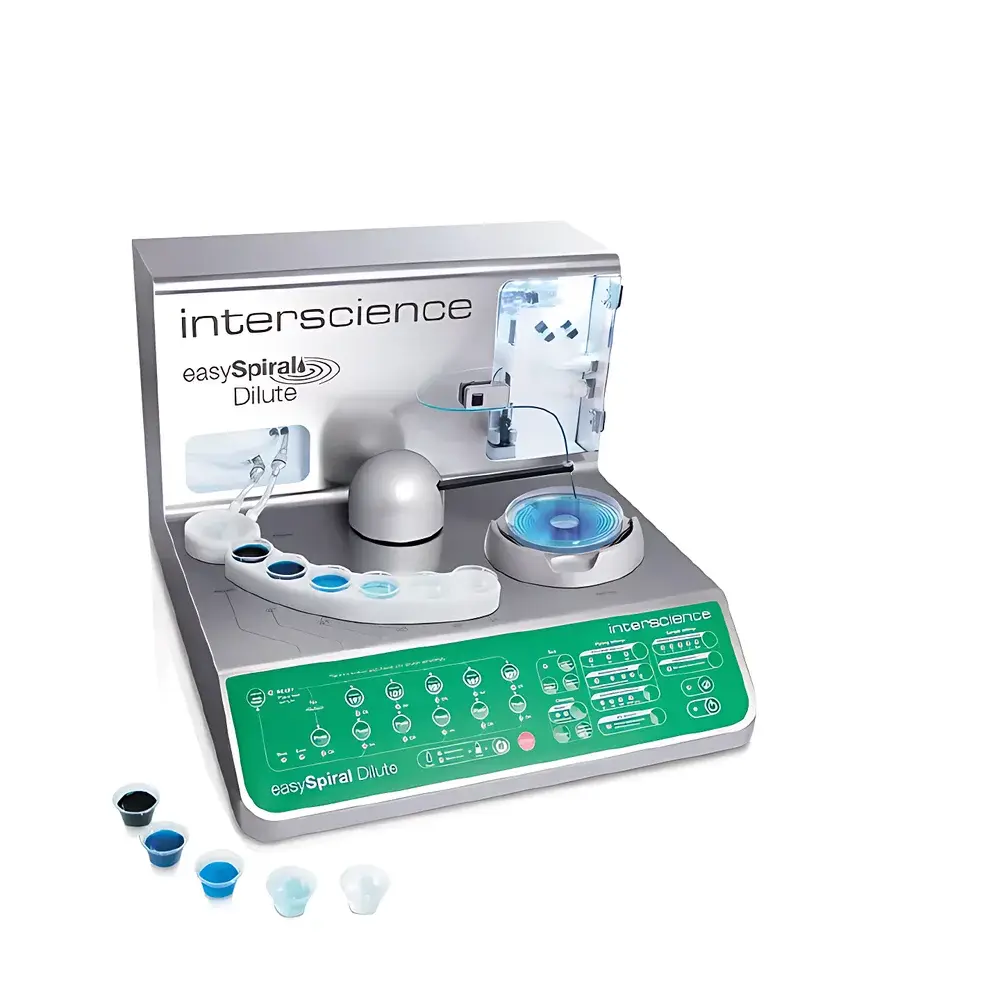
Interscience easySpira Dilutel® Automated Spiral Plater and Dilution System
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | easySpira Dilutel® |
| Syringe Capacity | 1000 µL |
| Preset Inoculation Volumes | 50 µL, 100 µL, 200 µL |
| Counting Range | 30–1×10¹² CFU/mL |
| Cycle Time (1 aspiration + 1 disinfection + 1 plating) | 25 s |
| Cycle Time (1 disinfection + 5 dilutions + 1 plating at one dilution factor) | 134 s |
| Cycle Time (1 disinfection + 5 dilutions + 1 plating per dilution factor) | 234 s |
| Needle Pressure | up to 8 bar |
| Automatic Disinfection Cycles | 1000 cycles (using 2 L bottle) |
| Automatic Dilution Cycles | 63 cycles (using 2 L bottle) |
| Compatible Diluents | Saline, Ringer’s solution |
| Max. Petri Dishes per Sample (50 µL) | 20 |
| Spiral Plating Mode | Optional (Ø90 mm & Ø150 mm dishes) |
| Loop Plating Mode | Optional (3 dilutions on Ø90 mm |
| Uniform Plating Mode | Optional (Ø55 mm, Ø90 mm, Ø150 mm) |
| Pour Plate Mode | Optional (Ø55 mm, Ø90 mm, Ø150 mm) |
| Editable Inoculation Volume via USB | 10–1000 µL |
| Traceability | Excel™ export, LIMS integration, DataLink™ compatibility |
| Sterilizable Components | Two autoclavable bottles (diluent & disinfectant) + connectors |
| Cross-Contamination Prevention | Overflow cleaning technology |
Overview
The Interscience easySpira Dilutel® is a CE-marked, fully automated spiral plater and serial dilution system engineered for high-throughput, regulatory-compliant microbiological enumeration in food safety, pharmaceutical QC, clinical diagnostics, and environmental testing laboratories. It integrates two critical workflows—precision serial dilution and standardized spiral or loop-based surface plating—into a single platform governed by ISO 4833-2:2013, ISO 7218, and AOAC Official Method 990.12 principles. Unlike conventional manual or semi-automated approaches, the easySpira Dilutel® eliminates pipetting variability, tube preparation, and operator-dependent plating geometry by executing reproducible 10-fold dilutions (up to 10⁻⁵ or higher) and controlled inoculum deposition using calibrated positive-displacement syringes and a dynamically balanced rotating arm. Its core architecture follows a closed-fluid-path design with pressure-regulated dispensing (up to 8 bar), ensuring consistent sample delivery across viscous or particulate-laden suspensions without clogging.
Key Features
- Integrated dilution and plating: Performs up to five sequential 10-fold dilutions followed by simultaneous or sequential plating across multiple dilution factors—eliminating manual transfer steps and associated contamination risks.
- Four selectable plating modes: Spiral (ISO-compliant logarithmic distribution on Ø90 mm and Ø150 mm dishes), Loop (discrete zone plating for multi-dilution analysis on Ø90 mm/Ø150 mm), Uniform (even coverage for colony isolation or inhibition assays on Ø55/90/150 mm), and Pour Plate (pre-poured agar compatibility).
- Autoclavable fluid handling: Includes two Class VI-certified, pressure-rated polypropylene bottles (diluent and disinfectant) and quick-connect sterilizable tubing—validated for ≥121°C, 15 psi, 20 min autoclave cycles per ISO 17665-1.
- Overflow-based disinfection: Patented rinse-and-drain protocol ensures complete needle and syringe tip decontamination between samples, validated per EN 13697 for non-porous surface efficacy.
- High-speed kinematics: Robotic arm achieves full cycle completion—sample aspiration, disinfection, 5-step dilution, and 6-point plating—in ≤234 seconds, supporting >200 samples per 8-hour shift.
- User-configurable volume control: Inoculation volume programmable from 10 µL to 1000 µL via USB interface, enabling method adaptation for low-biomass samples or high-titer suspensions without hardware modification.
Sample Compatibility & Compliance
The easySpira Dilutel® accommodates liquid, homogenized, and membrane-filter eluate samples compatible with standard microbiological media (e.g., PCA, TSA, VRBA, mFC). It supports saline and Ringer’s solution as primary diluents, meeting USP and Ph. Eur. 2.6.12 requirements for neutralizing agents. All firmware operations log timestamped metadata—including user ID, sample ID, dilution factor, plating mode, volume, and cycle duration—enabling full audit trail generation compliant with FDA 21 CFR Part 11 (electronic signatures, audit logs, data integrity controls) when deployed with Interscience DataLink™ software. The system is designed for GLP/GMP environments and has been verified for repeatability (RSD ≤3.2% across n=6 replicates, 10⁴–10⁸ CFU/mL range) per ISO 5725-2.
Software & Data Management
Controlled via Interscience’s Windows-based Dilutel® Software v4.x, the system supports method creation, parameter locking, and role-based access control (administrator/operator). Raw plating coordinates, dilution history, and instrument status are exported in native .csv or Excel™-formatted reports. LIMS integration is achieved through ASTM E1482-compliant HL7 or direct SQL database push. DataLink™ enables real-time synchronization with enterprise quality management systems (QMS), including traceability to original sample receipt, analyst assignment, and final report sign-off—fully satisfying ISO/IEC 17025 clause 7.5.2 on result reporting and record retention.
Applications
- Quantitative microbial enumeration in ready-to-eat foods, dairy products, and beverages per ISO 4833-1 and NF V08-034.
- Environmental monitoring of cleanrooms and aseptic processing areas (EU GMP Annex 1, ISO 14644-1).
- Pharmaceutical water system validation (USP , EP 2.2.47) and bioburden testing of non-sterile products (USP ).
- Antimicrobial susceptibility assay preparation requiring precise inoculum density (CLSI M07-A11).
- Research applications involving strain isolation, phage titering, and biofilm dispersion quantification where reproducible surface distribution is essential.
FAQ
Does the easySpira Dilutel® require calibration certificates for ISO/IEC 17025 accreditation?
Yes—Interscience provides optional UKAS-accredited calibration services for syringe volume accuracy (±0.8% tolerance at 100 µL–1000 µL), plating geometry verification (spiral pitch deviation ≤±0.3 mm), and timer traceability (NIST-traceable stopwatch validation).
Can the system handle viscous samples such as yogurt or sauces?
Yes—the 8-bar dispensing pressure and large-bore stainless-steel needle (ID 0.8 mm) enable reliable aspiration and ejection of suspensions up to 10,000 cP, provided samples are pre-homogenized per ISO 6887-1.
Is remote monitoring or troubleshooting supported?
The system includes Ethernet and optional Wi-Fi connectivity; Interscience Remote Assist allows secure, encrypted technician access for diagnostic logging and firmware updates under GDPR-compliant data handling protocols.
What maintenance intervals are recommended for routine operation?
Daily: Visual inspection of syringe seals and overflow drain path. Weekly: Autoclaving of bottles and connectors; performance verification using certified reference suspension (CRM 8401). Annually: Full metrological recalibration and mechanical alignment check by authorized service engineer.