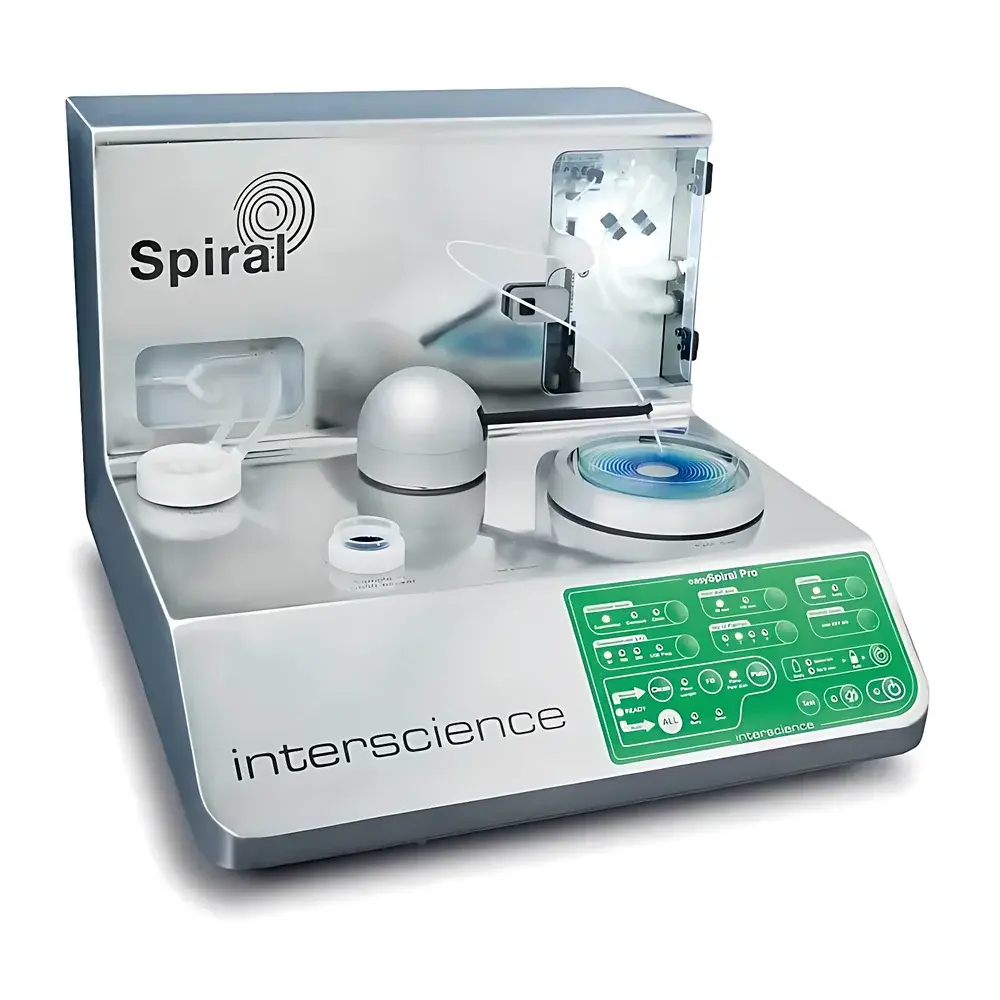
Interscience easySpiral Pro Automated Spiral Plater
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | easySpiral Pro |
| Compliance | AFNOR V08-100, ISO 7218, SN/T 2098–2008 |
| Sample Volume Range | 10–1000 µL (programmable via USB) |
| Plating Time | ≤25 s per plate (including wash, aspirate, and spiral deposition) |
| Detection Range | 30–1.1 × 10⁷ CFU/mL |
| Sterilization | Autoclavable components (GL45 bottle adapters, needle assembly, base plate) |
| Cleaning System | Overflow-based internal/external needle rinse with real-time fluid-level sensing |
| Connectivity | USB interface for method programming, audit trail export, and GLP-compliant data logging |
Overview
The Interscience easySpiral Pro Automated Spiral Plater is a precision-engineered microbiological plating system designed for rapid, reproducible, and traceable quantitative microbial enumeration. Based on the standardized spiral plating principle defined in ISO 7218 and AFNOR V08-100, the instrument deposits a continuous, logarithmically decreasing volume of liquid sample onto the surface of an agar plate using a calibrated spiral trajectory. This eliminates manual serial dilution steps—reducing hands-on time by up to 75%—while maintaining full compliance with regulatory requirements for food, pharmaceutical, environmental, and clinical microbiology laboratories. The system operates on a closed-loop fluidic architecture: sample aspiration, needle priming, overflow cleaning, and controlled deposition are fully automated and optically monitored. Its modular, autoclavable design ensures sterility integrity across high-throughput workflows without cross-contamination risk.
Key Features
- Full automation cycle completed in ≤25 seconds—including needle sterilization, sample aspiration, and spiral deposition—enabling throughput of >120 plates per hour.
- Overflow cleaning system simultaneously rinses both internal lumen and external surface of the stainless-steel inoculation needle using pre-sterilized rinse solution; fluid level sensors trigger alerts when reservoirs fall below operational thresholds.
- USB-programmable volume control (10–1000 µL) with customizable spiral pitch, start radius, and rotation speed—supporting method validation and SOP alignment across multiple sample matrices.
- All critical wetted parts—including GL45 bottle adapters, needle assembly, and base plate—are validated for steam sterilization at 121°C for 20 minutes (EN 285), ensuring compatibility with GMP/GLP environments.
- Real-time visual confirmation of all fluid paths: transparent tubing, illuminated reservoirs, and LED status indicators provide immediate verification of system readiness and process continuity.
Sample Compatibility & Compliance
The easySpiral Pro accommodates standard Petri dishes (90 mm and 150 mm diameters) and accepts samples in sterile cups (DB50 or 1000-pack disposable cups), bottles with GL45 threads, or direct syringe coupling. It supports viscous suspensions (e.g., homogenized food extracts, wastewater concentrates) and low-biomass samples (e.g., purified water, cleanroom swabs) without clogging or flow deviation. Regulatory alignment includes full adherence to ISO 7218:2018 (Microbiology of food and animal feeding stuffs — General requirements and guidance for microbiological examinations), AFNOR V08-100 (Qualitative and quantitative enumeration of microorganisms by spiral plating), and SN/T 2098–2008 (Inspection and quarantine industry standard for microbial detection in imported/exported goods). Optional barcode scanning integration enables sample ID traceability aligned with FDA 21 CFR Part 11 requirements for electronic records and signatures.
Software & Data Management
The included easySpiral Pro software—delivered on optical media and installable on Windows-based laboratory PCs—provides method configuration, run logging, and audit-trail generation. Each plating event records timestamp, operator ID (if network-authenticated), sample ID (manual or scanned), volume setting, rinse cycle count, and error flags. Export formats include CSV and PDF reports compliant with ISO/IEC 17025 documentation standards. USB-based firmware updates ensure long-term platform support, while encrypted local storage preserves data integrity during power interruption or unplanned shutdown. Software validation packages (IQ/OQ documentation templates) are available upon request for regulated facilities requiring formal system qualification.
Applications
This instrument is routinely deployed in accredited quality control laboratories for total viable count (TVC), coliforms, Enterobacteriaceae, yeasts/molds, and pathogen enumeration (e.g., Salmonella, Listeria monocytogenes) in food and beverage manufacturing. It is also used in pharmaceutical water system monitoring (USP , EP 2.6.12), environmental bioburden assessment of cleanrooms (ISO 14644-1), and clinical research involving antibiotic susceptibility testing preparation. Its accuracy in low-concentration ranges (30–10² CFU/mL) makes it suitable for verifying disinfectant efficacy (EN 13697) and validating sterilization cycles where precise inoculum quantification is required.
FAQ
What regulatory standards does the easySpiral Pro comply with?
It meets ISO 7218:2018, AFNOR V08-100, and SN/T 2098–2008. Optional configurations support 21 CFR Part 11 compliance via software audit trails and electronic signature modules.
Can the instrument handle viscous or particulate-laden samples?
Yes—the positive-displacement piston pump and large-diameter fluid path minimize clogging; optional filtration adapters are available for highly turbid suspensions.
Is routine maintenance required beyond autoclaving consumables?
No scheduled mechanical maintenance is needed. All fluidic operations are self-monitoring; only periodic visual inspection of tubing and sensor calibration (annually recommended) are advised.
How is cross-contamination prevented between runs?
Each cycle begins with an overflow rinse that flushes both interior and exterior needle surfaces with sterile rinse solution; residual carryover is physically impossible due to fixed-volume aspiration and air-gap separation.
Does the system support integration into LIMS or ELN platforms?
Raw data exports (CSV) and structured logs enable seamless ingestion into common LIMS/ELN systems; API access is available under enterprise licensing agreements.