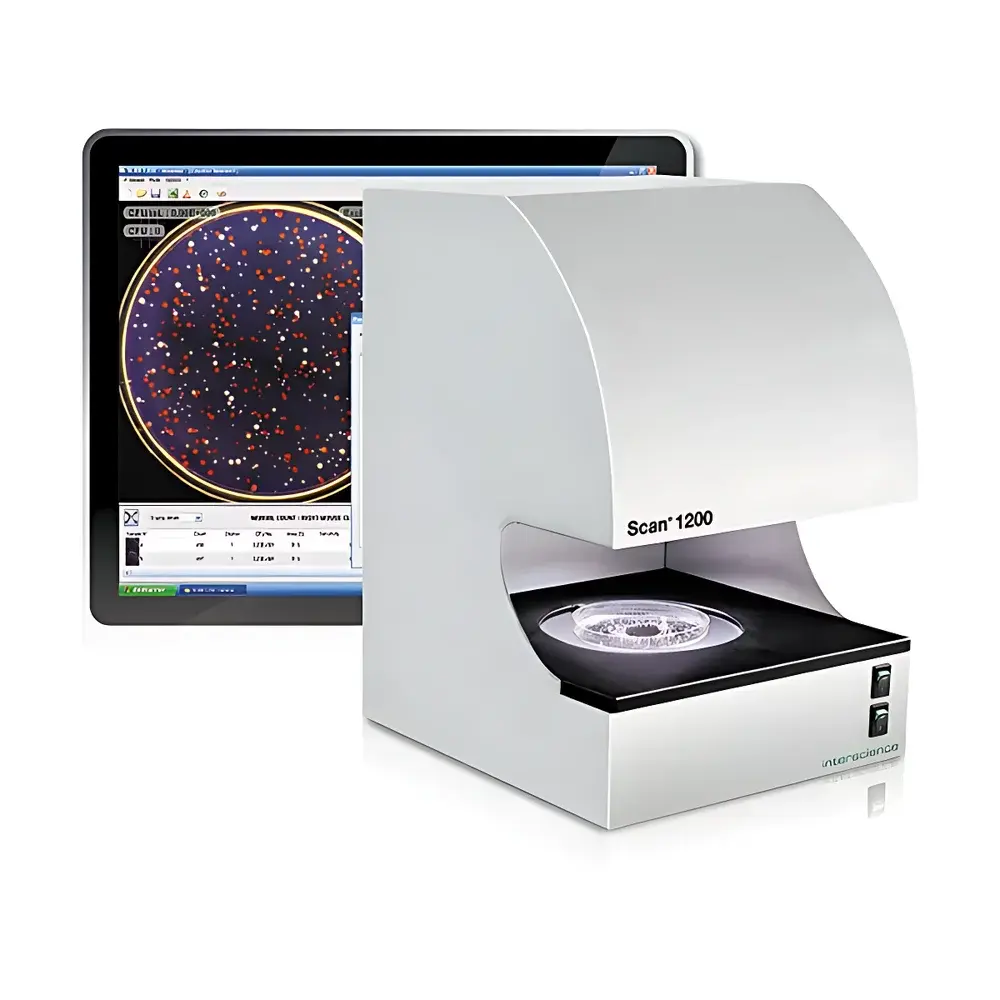
Interscience Scan 1200 HD Automatic Colony Counter and Zone of Inhibition Analyzer
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | Scan 1200 |
| Camera | 1.2 MP HD CCD with 28× zoom lens |
| Illumination | Automated dark-field system (6 configurable top/bottom, white/black combinations) |
| Petri Dish Compatibility | Ø 55–90 mm |
| Minimum Detectable Colony Diameter | 0.05 mm |
| Counting Speed | Up to 1000 colonies/sec |
| Zone Measurement Accuracy | ±0.3 mm |
| Resolution | ±0.1 mm |
| Antibiotic Disc Capacity | Up to 7 per 90 mm plate |
| Analysis Standards | CLSI, EUCAST, CA-SFM (Human & Veterinary), Custom Criteria |
| Software Languages | English, French, German, Spanish, Japanese, Chinese, Russian |
| Compliance | 21 CFR Part 11, ISO 7218, AOAC 977.27 |
| Data Export Formats | CSV, Excel™, PDF, JPEG, PNG, BMP, SCA, BIO |
| Power | 100–240 V~, 50–60 Hz, 20 W |
| Dimensions (W×D×H) | 28.8 × 26.3 × 37.3 cm |
| Weight | 9.4 kg |
| Warranty | 3 years (registration required) |
| Spare Parts Availability | 10 years |
| Software Updates | 3 years (excludes validation) |
Overview
The Interscience Scan 1200 HD Automatic Colony Counter and Zone of Inhibition Analyzer is a CE-marked, regulatory-compliant instrument engineered for precision microbiological enumeration and antimicrobial susceptibility testing in pharmaceutical, food safety, clinical, and contract research laboratory environments. It employs high-resolution digital imaging coupled with adaptive dark-field illumination to achieve robust optical contrast—essential for accurate detection of small, translucent, or low-contrast colonies on diverse solid media. The system operates on the principle of pixel-based morphometric analysis: colonies are identified via thresholded grayscale segmentation, validated through size, shape, and intensity criteria, and resolved using sub-pixel interpolation algorithms. Zones of inhibition are measured by edge-detection algorithms applied to radial intensity gradients around antibiotic discs, wells, or cylinders, with calibration traceable to NIST-traceable standards. Designed for integration into regulated workflows, the Scan 1200 meets the data integrity requirements of FDA 21 CFR Part 11, ISO 7218:2017, and AOAC Official Method 977.27, supporting audit-ready electronic records with full metadata capture.
Key Features
- Automated colony enumeration across pour-plated, spread-plated, spiral-plated, and loop-serially inoculated agar surfaces
- Multi-color colony discrimination: simultaneous detection and classification of up to four colony colors per plate, with user-defined exclusion of background or interfering hues
- Advanced cluster separation algorithm for resolving fused or overlapping colonies without manual splitting artifacts
- Interactive correction interface: real-time addition, deletion, or reclassification of colonies with timestamped operator annotation
- Configurable polygonal region-of-interest (ROI) definition for selective counting within irregular zones (e.g., membrane filters, selective agar sectors)
- Zone of inhibition analysis supporting paper discs (all major brands), agar wells, Oxford cups (stainless steel and plastic), and custom geometries
- Preloaded interpretive criteria libraries: CLSI M02/M100, EUCAST v14.0, CA-SFM Human & Veterinary, plus customizable breakpoint tables
- Integrated dark-field illumination system with six programmable lighting configurations—top/bottom, white/black, single/dual source—to maximize contrast for challenging media (e.g., chromogenic, blood, or MacConkey agars)
- HD 1.2 MP color CCD sensor with Japanese-engineered 28× optical zoom lens, delivering consistent resolution across Ø 55–90 mm Petri dishes
Sample Compatibility & Compliance
The Scan 1200 accommodates standard and non-standard microbiological substrates including conventional agar plates, Petrifilm™, Compact Dry™, MC-Media Pads™, EasyPlate™, and membrane filters. Its optical architecture ensures reliable detection on chromogenic, selective, and differential media—even under low-contrast conditions. All analytical outputs include embedded metadata: sample ID, operator, timestamp, plate dimensions, medium type, incubation parameters (if imported), antibiotic identity, measured zone diameters, and interpretive category (S/I/R). The system supports ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) through immutable audit trails, dual electronic signatures, and role-based access control (RBAC) with password-protected administrator, analyst, and reviewer accounts. Validation documentation—including IQ/OQ templates, risk assessments, and traceability matrices—is provided to support GxP compliance (GLP, GMP Annex 11, ISO/IEC 17025).
Software & Data Management
Scan software (v5.x) runs natively on Windows 10/11 (Intel i5+, 4 GB RAM, USB 3.0, 1280×1024 display) and features a modular architecture compatible with LIMS/SIL interfaces via HL7, ASTM E1384, or direct SQL/ODBC connectivity. DataLink Pro integration enables bidirectional traceability from dilution and plating steps through final enumeration and AST interpretation. All sessions are stored as encrypted, version-controlled projects with checksum-verified backups. Export options include CSV (for statistical analysis), Excel™ (with formatted reports), PDF (signed and stamped), and image formats (JPEG, PNG, BMP) with embedded EXIF metadata. Audit logs record every action—including parameter changes, corrections, report generation, and user logins—with SHA-256 hashing and tamper-evident timestamps. Software updates are delivered via secure HTTPS channel; post-warranty validation services are available under separate agreement.
Applications
The Scan 1200 serves critical quality control and R&D functions across regulated industries: environmental monitoring in sterile manufacturing facilities (ISO 14644-1); microbial limit testing per USP / and Ph. Eur. 2.6.12; antimicrobial efficacy testing (AOAC 966.02, EN 13624); raw material bioburden assessment; stability-indicating assay development; and clinical microbiology lab accreditation (CAP, COLA, UKAS). Its reproducibility (CV <2% across repeated counts of identical plates) eliminates inter-operator variability inherent in manual counting, while its throughput—processing 100 plates per hour with full traceability—reduces turnaround time for release testing. In food safety labs, it supports ISO 4833-1 enumeration and ISO 20776-1 disk diffusion testing, with automated reporting aligned to EFSA and FDA Food Code requirements.
FAQ
Does the Scan 1200 comply with 21 CFR Part 11 requirements for electronic records and signatures?
Yes. It implements role-based authentication, audit trails with immutable timestamps, electronic signatures with dual approval, and system-generated records that meet ALCOA+ data integrity principles.
Can the instrument validate zone measurements against CLSI M02 reference standards?
Yes. Built-in calibration routines use NIST-traceable reference discs and agar plates; measurement uncertainty is characterized per ISO/IEC 17025 and documented in the validation package.
Is offline operation supported, and how is data synchronized when reconnecting to LIMS?
Yes. Local session storage preserves all metadata and images during network outages; upon reconnection, DataLink Pro performs conflict-free, timestamp-ordered synchronization with full change logging.
What is the expected service life of the optical components and camera module?
The HD CCD sensor and Japanese lens assembly are rated for >50,000 operational cycles; Interscience guarantees spare parts availability for 10 years from date of manufacture.
How does the system handle plates with condensation or uneven agar surfaces?
Dark-field illumination minimizes specular reflection; adaptive histogram equalization and multi-threshold segmentation reduce false positives from surface artifacts without compromising sensitivity to microcolonies ≥0.05 mm.