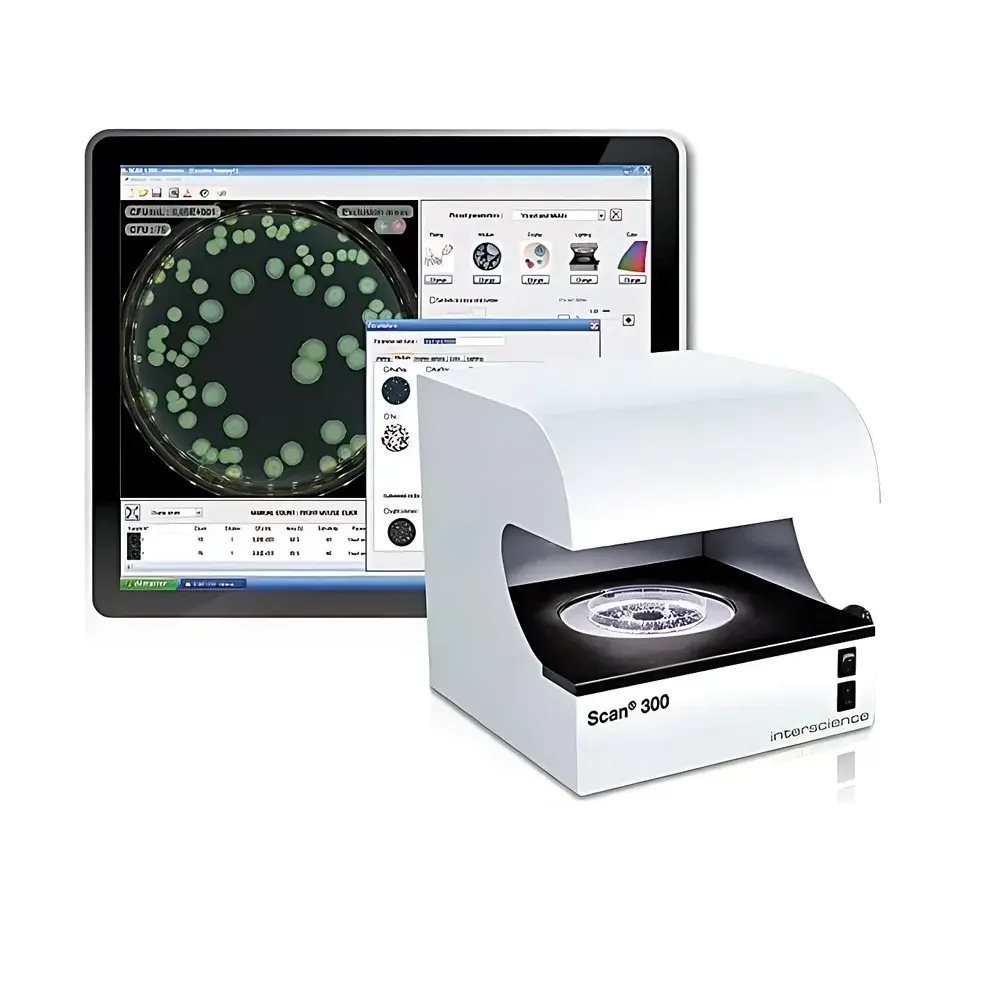
Interscience Scan 300 Automated Colony Counter
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | Scan 300 |
| Camera | 1 MP CMOS color sensor with M12 lens (28× zoom) |
| Illumination | Automatic dark-field system with 6 configurable combinations (top/bottom, white/black background) |
| Petri Dish Compatibility | Ø 55–90 mm |
| Minimum Detectable Colony Diameter | 0.1 mm |
| Counting Speed | Up to 1000 colonies/sec |
| Counting Accuracy | ≥98% |
| Software Languages | English, French, German, Spanish, Japanese, Chinese, Russian |
| Compliance | ISO 7218, AOAC 977.27, FDA 21 CFR Part 11 (audit trail, electronic signatures, role-based access), GLP/GMP-aligned |
| Data Export Formats | CSV, Excel™, PDF, JPEG, PNG, BMP, SCA, BIO |
| Interface | USB 2.0 |
| Resolution Support | ≥1280 × 1024 px |
| Power Supply | 100–240 V~, 50–60 Hz, 20 W max |
| Dimensions (W×D×H) | 28.7 × 27.1 × 28.9 cm |
| Net Weight | 8.42 kg |
| Warranty | 3 years (registration required) |
| Spare Parts Availability | 10 years |
| Software Updates | 3 years (excluding validation support) |
Overview
The Interscience Scan 300 Automated Colony Counter is an ISO 7218- and AOAC 977.27-compliant digital imaging system engineered for precise, high-throughput enumeration of microbial colonies on standard agar plates. It employs advanced dark-field illumination combined with a high-sensitivity 1-megapixel CMOS color camera and motorized 28× zoom optics to resolve colonies as small as 0.1 mm in diameter—critical for early-stage growth detection in environmental, food, pharmaceutical, and clinical microbiology workflows. Unlike conventional manual or semi-automated counters, the Scan 300 implements pixel-level contrast analysis and adaptive threshold segmentation to distinguish individual colonies—even under conditions of partial merging or low-contrast morphology—without reliance on user-defined templates or fixed algorithms. Its stainless-steel enclosure ensures durability in regulated laboratory environments, while its compact footprint (28.7 × 27.1 × 28.9 cm) supports integration into biosafety cabinets or QC benches without compromising ergonomics or workflow continuity.
Key Features
- Automated counting across four standard inoculation methods: pour plate, spread plate, spiral plating, and loop dilution—each with method-specific calibration and validation protocols.
- Real-time manual correction interface: operators can add, delete, or reclassify colonies directly on the live image; merged colonies are automatically separated using morphological deconvolution based on edge gradient and convex hull analysis.
- Polygonal region-of-interest (ROI) definition for selective counting within irregularly shaped or segmented plates—essential for membrane filtration or multi-zone assays.
- Configurable dark-field illumination engine with six preprogrammed lighting modes (top/bottom, white/black background combinations), optimized to maximize colony contrast while minimizing operator eye fatigue during extended sessions.
- Full 21 CFR Part 11 compliance: built-in audit trail, time-stamped electronic signatures (dual sign-off for review/approval), role-based user permissions (administrator, analyst, reviewer), and immutable session logs stored locally and exportable in encrypted format.
- Integrated traceability architecture via DataLink® Pro connectivity—enabling bidirectional synchronization with LIMS/SIL systems and automated propagation of sample IDs, dilution factors, incubation parameters, and instrument metadata from preparatory steps (e.g., Interscience DiluFlow or Spiral System).
Sample Compatibility & Compliance
The Scan 300 accepts standard Petri dishes ranging from Ø 55 mm to Ø 90 mm—including polystyrene, polypropylene, and glass variants—with automatic dish-centering and focus calibration. Its optical path accommodates both opaque and translucent media (e.g., PCA, TSA, VRBA, mFC, Sabouraud Dextrose Agar) without recalibration. Regulatory alignment extends beyond ISO 7218: the system supports full GxP documentation requirements through structured data capture (sample ID, operator, date/time, plate batch, incubation conditions), tamper-evident file hashing for exported reports, and optional IQ/OQ documentation packages aligned with pharmaceutical validation standards (Annex 11, USP , EU GMP Annex 15). All firmware and software revisions undergo version-controlled release testing per ICH Q9 principles.
Software & Data Management
Scan Software v5.x (Windows 10/11, Intel i5+ CPU, 4 GB RAM minimum) provides a modular, intuitive interface with three operational tiers: acquisition, analysis, and reporting. Raw images and processed count matrices are stored in a local SQLite database with SHA-256 checksums; exports include CSV (for statistical analysis in R or Python), Excel™ (with embedded formulas for log₁₀ CFU/mL calculation), PDF (signed, watermarked, with embedded image thumbnails), and lossless BMP/PNG/JPEG formats for archival or regulatory submission. Audit trails record every user action—including parameter changes, ROI modifications, and signature events—with granular timestamps and IP address logging where networked. Integration with LIMS occurs via HL7-compatible REST API or ODBC drivers, supporting automated result ingestion and status flagging (e.g., “count out of specification”, “plate rejected”).
Applications
- Food & beverage safety testing: rapid enumeration of total viable counts, coliforms, yeasts/molds per ISO 4833-1, ISO 21528-2, and FDA BAM protocols.
- Pharmaceutical microbiological quality control: environmental monitoring (EM) plate enumeration, sterility test support, and bioburden assessment compliant with USP , , and EP 2.6.12.
- Clinical and public health laboratories: quantification of pathogens (e.g., E. coli, S. aureus) from urine, wound swabs, or water samples per CLSI M07 and ISO 11133.
- Research microbiology: kinetic colony growth studies, antimicrobial susceptibility screening (e.g., zone-of-inhibition integration), and biofilm assay quantification on agar-embedded membranes.
- Contract testing laboratories: auditable, repeatable reporting for ISO/IEC 17025 accreditation—particularly for turnaround-critical projects requiring same-day data delivery to clients.
FAQ
Does the Scan 300 require annual calibration or certification?
No routine calibration is required—the system uses factory-traceable optical and geometric references embedded in each calibration disc. However, users must perform daily verification using the supplied dual-diameter calibration discs (Ø 0.5 mm and Ø 2.0 mm) and document results per internal SOP or ISO 17025 clause 6.4.3.
Can the Scan 300 process plates with condensation or surface moisture?
Yes—its dark-field illumination minimizes specular reflection artifacts, and the software includes moisture artifact suppression filters trained on >10,000 real-world plate images. For optimal accuracy, plates should be equilibrated to room temperature prior to scanning.
Is validation support available for regulated environments?
Interscience provides IQ/OQ protocol templates, URS mapping documents, and on-site installation qualification services (fee-based). Full PQ support requires collaboration with the end-user’s QA team to define acceptance criteria per product-specific risk assessment.
What happens to data if the USB connection is interrupted during acquisition?
All image capture and counting operations occur locally on the connected PC; the device functions as a smart camera peripheral. Interrupted transfers do not corrupt existing datasets—session files are written incrementally with atomic commit logic to prevent partial writes.
How does the system handle overlapping or chain-forming colonies (e.g., streptococci)?
The algorithm applies iterative watershed segmentation informed by colony circularity, solidity, and intensity gradient profiles. Users may adjust separation aggressiveness via a continuous “merge tolerance” slider (0–100%), validated against reference slides prepared per ISO 7218 Annex B.