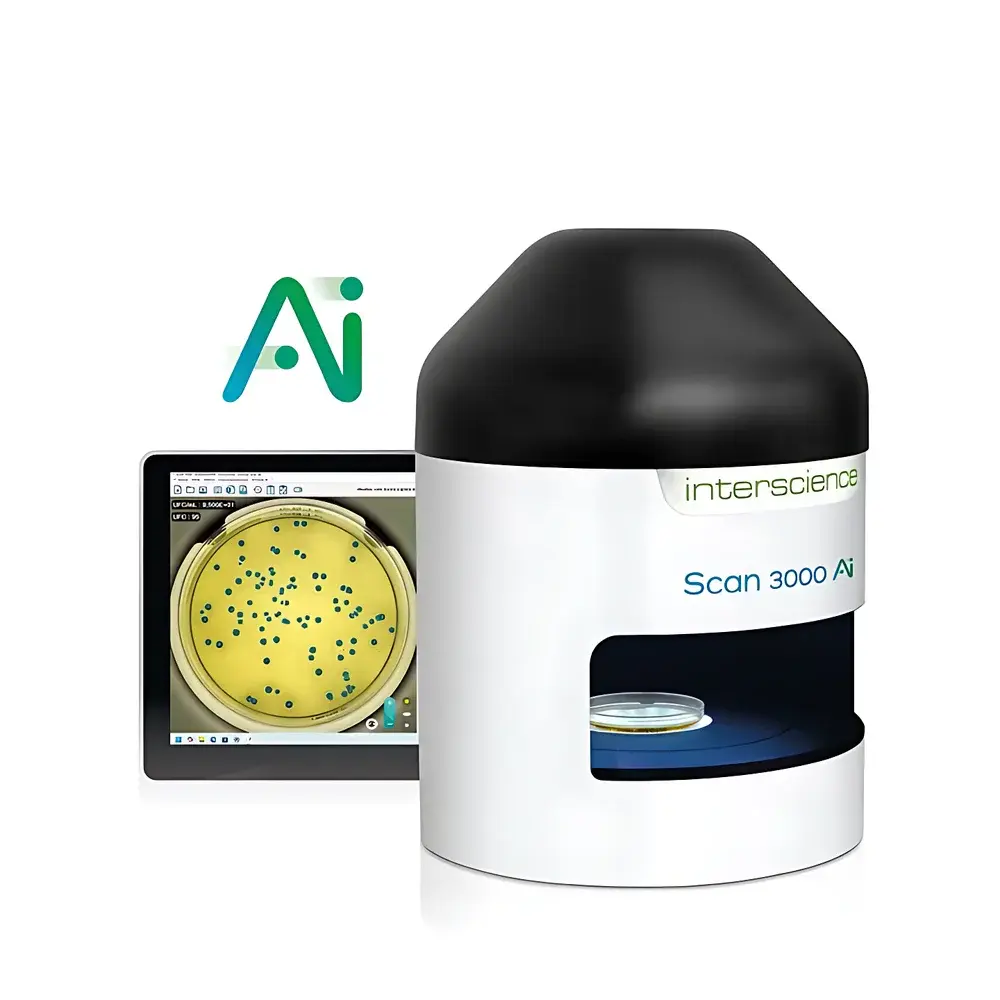
Interscience Scan 3000 Ai Automated AI-Powered Colony Counter and Zone of Inhibition Analyzer
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | Scan 3000 Ai |
| Camera | 69 MP ultra-HD CMOS color sensor with Japanese HD zoom lens |
| Illumination | Auto-switching 7-mode LED dome lighting (top/bottom, white/black background) |
| Petri Dish Compatibility | Ø55–Ø90 mm |
| Colony Detection Limit | ≥0.03 mm diameter |
| Counting Speed | Up to 1,000 colonies/sec |
| Zone Measurement Accuracy | ±0.2 mm |
| Resolution | ±0.1 mm |
| Antibiotic Disc Capacity | Up to 7 per Ø90 mm plate |
| Analysis Time per Plate | 1–3 sec (7 zones) |
| Color Discrimination | 4 target colors + 2 exclusion colors |
| Software Languages | English, French, German, Spanish, Japanese, Chinese, Russian |
| Compliance | 21 CFR Part 11, ISO 7218, AOAC 977.27 |
| Data Export Formats | CSV, Excel™, PDF, JPEG, PNG, BMP, SCA, BIO |
| Connectivity | USB 3.0, LIMS/SIL via DataLink®/DataLink Pro® |
| Power | 100–240 V~, 50–60 Hz, 70 W max |
| Dimensions (W×D×H) | 32 × 32 × 44 cm |
| Net Weight | 11.15 kg |
| Warranty | 3 years (registration required) |
| Spare Parts Availability | 10 years |
Overview
The Interscience Scan 3000 Ai is a fully automated, AI-driven colony counter and zone-of-inhibition (ZOI) analyzer engineered for high-throughput, regulatory-compliant microbiological quality control in pharmaceutical, food safety, clinical diagnostics, and academic research laboratories. It operates on a dual-core imaging and machine learning architecture: high-resolution color morphometric analysis combined with a deep convolutional neural network (CNN) trained on over one million expert-annotated colony images across diverse media types, incubation conditions, and microbial morphologies. Unlike rule-based algorithms, the Scan 3000 Ai’s AI model dynamically distinguishes true microbial colonies from artifacts—including condensation droplets, agar imperfections, pen markings, and debris—without requiring manual threshold tuning. Its optical path integrates a 69-megapixel CMOS sensor, precision Japanese HD zoom lens, and adaptive 7-mode LED dome illumination system that eliminates specular reflection and shadow artifacts, enabling consistent, reproducible quantification across all standard culture formats including pour plates, spread plates, spiral plating, membrane filters, and commercial rapid methods (Petrifilm™, Compact Dry™, MC-Media Pads™, EasyPlate™). The system delivers full-dish coverage analysis at sub-30 µm spatial resolution, supporting accurate enumeration of bacteria, yeasts, and molds in mixed-culture scenarios.
Key Features
- AI-powered colony detection and classification trained on >1,000,000 annotated images across >120 species and 35+ media formulations
- Full-dish scanning with 100% surface coverage and automatic separation of clustered or overlapping colonies using morphological segmentation
- Real-time ZOI detection and measurement for up to seven antibiotic agents per Ø90 mm plate, compliant with CLSI, EUCAST, CA-SFM (human & veterinary), and custom interpretive criteria
- Multi-color discrimination engine capable of identifying and quantifying up to four distinct colony chromogenic phenotypes simultaneously while excluding two user-defined background colors
- Configurable polygonal ROI definition for selective counting in irregularly shaped or partially inoculated dishes
- Integrated traceability architecture: electronic audit trail, dual electronic signature, and immutable session logs meeting FDA 21 CFR Part 11 requirements
- USB 3.0 interface with native compatibility for Interscience DataLink® and DataLink Pro® systems, enabling bidirectional integration with LIMS, SIL, and automated diluters
- Regulatory-ready software platform with role-based access control (RBAC), password policy enforcement, and configurable user permissions aligned with GLP/GMP Annex 11 principles
Sample Compatibility & Compliance
The Scan 3000 Ai supports all standard microbiological culture formats used in ISO 7218:2017–compliant testing workflows. It accommodates Petri dishes ranging from Ø55 mm to Ø90 mm—including deep-dish variants—and processes plates prepared by pour, spread, spiral, loop, or membrane filtration techniques. The system is validated for use with chromogenic and selective media (e.g., CHROMagar™, Oxoid Brilliance™, Hardy Diagnostics), as well as commercially licensed rapid test platforms such as 3M Petrifilm™, Hygiena Compact Dry™, NeoGen MC-Media Pads™, and BioControl EasyPlate™. All analytical outputs—including raw image data, processed counts, ZOI diameters, classification metadata, and operator annotations—are stored with full time-stamped provenance and cryptographic integrity. The instrument meets AOAC Official Method 977.27 for automated colony enumeration and is designed to support validation under USP , , and , as well as EU GMP Annex 1 and ICH Q5C guidelines for microbial assay qualification.
Software & Data Management
Scan 3000 Ai runs on Interscience’s proprietary ScanSoft v5.x platform—a Windows-based application certified for operation on Windows 10/11 (x64) with Intel i7/i9 or Xeon Gold processors and NVIDIA RTX 3060/4050 GPUs. The software provides granular session management: each analysis generates a tamper-evident .SCA file containing unprocessed TIFF images, pixel-level mask overlays, count history, correction logs, and operator metadata. Export options include CSV and Excel™ for statistical analysis; PDF reports with embedded images and compliance footers; and lossless JPEG/PNG/BMP exports for archival or external review. When connected to DataLink Pro®, the system auto-populates sample IDs, dilution factors, strain identifiers, and antibiotic names directly from upstream instruments, eliminating transcription errors. Audit trails record every action—including manual corrections, zone additions/removals, and parameter changes—with timestamps, user IDs, and IP addresses. All data fields are digitally signed and cryptographically hashed to ensure non-repudiation during regulatory inspections.
Applications
- Pharmaceutical QC: Enumeration of bioburden in non-sterile products, environmental monitoring (EM) isolates, and sterility test confirmations per USP and Ph. Eur. 2.6.12
- FDA-regulated food and beverage testing: Total viable count (TVC), coliforms, E. coli, Staphylococcus aureus, and Listeria spp. per Bacteriological Analytical Manual (BAM) and ISO standards
- Clinical microbiology labs: AST interpretation according to CLSI M100 and EUCAST breakpoints; rapid screening of multidrug-resistant organisms (MDROs)
- Academic and contract research organizations (CROs): High-fidelity replication of colony morphology studies, antifungal susceptibility testing, and biofilm quantification assays
- Biotechnology process development: Monitoring microbial contamination in cell culture reagents, fermentation broths, and purified protein batches
FAQ
Does the Scan 3000 Ai require routine recalibration?
No—its optical calibration is factory-performed and maintained via built-in reference discs included in the standard kit. Users perform periodic verification using the three supplied NIST-traceable calibration plates.
Can the AI model be retrained with lab-specific strains or media?
Yes. Interscience offers optional AI model fine-tuning services under controlled validation protocols, allowing customization for proprietary growth substrates or atypical morphologies while preserving regulatory compliance.
Is remote software update supported?
Yes. Firmware and ScanSoft updates are delivered via secure HTTPS channels with digital signature verification and rollback capability.
What happens if an antibiotic disc is misaligned or partially obscured?
The ZOI detection algorithm applies edge-enhanced contour mapping and iterative Hough transform refinement to reconstruct incomplete circles, ensuring robustness against minor positional variance or light scattering artifacts.
How does the system handle mixed-species colonies with overlapping colors?
Using multi-channel spectral deconvolution and texture-aware clustering, the software assigns probabilistic class labels (bacteria/yeast/mold) per colony based on size, shape, edge gradient, and RGB histogram distribution—validated against ATCC reference strains.