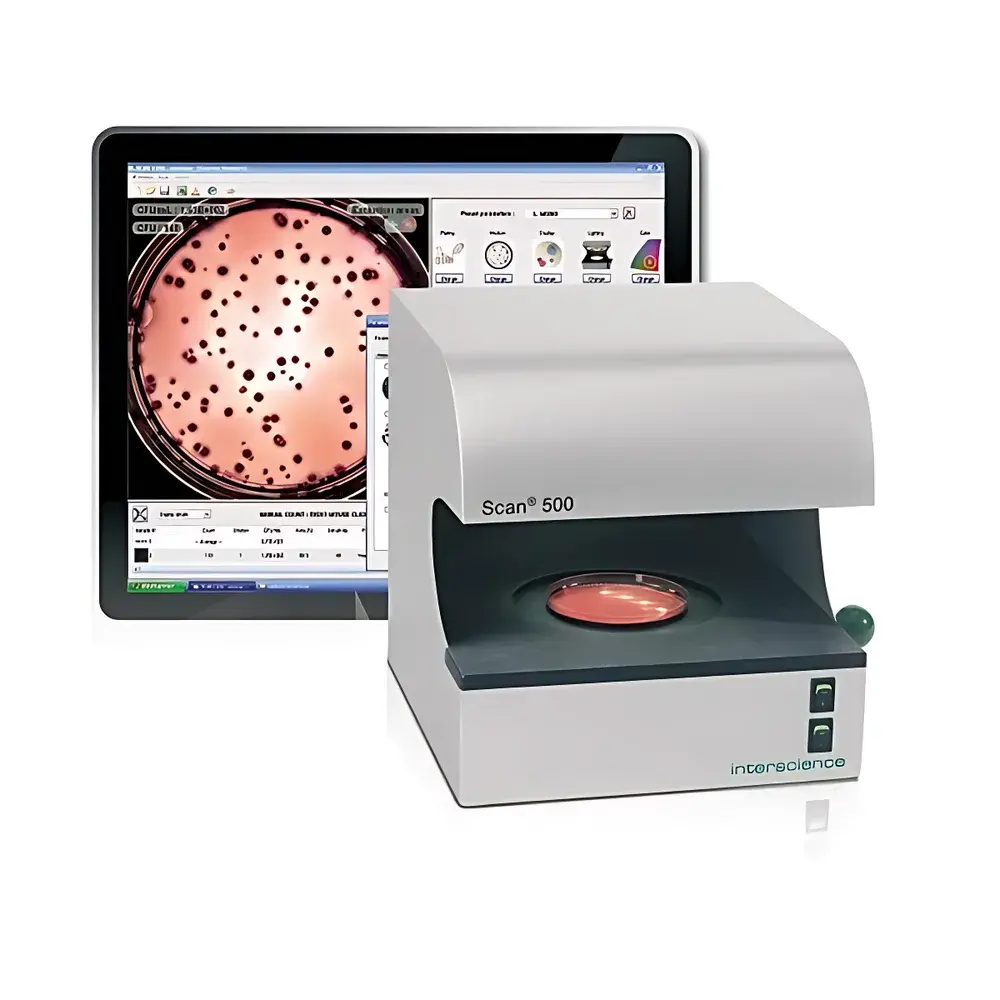
Interscience Scan 500 Automated Colony Counter
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | Scan 500 |
| Camera | 1 MP CMOS color |
| Lens | M12, 28× zoom |
| Illumination | Automatic dark-field system with 6 configurable top/bottom/white/black combinations |
| Petri Dish Compatibility | Ø55–Ø90 mm |
| Minimum Detectable Colony Diameter | 0.1 mm |
| Counting Speed | Up to 1000 colonies/sec |
| Accuracy | ≥98% |
| Inhibition Zone Detection | Yes, with ±0.3 mm measurement precision |
| Standards Compliance | ISO 7218, AOAC 977.27, CLSI, EUCAST, CA-SFM (human & veterinary), custom standard support |
| Software Languages | English, French, German, Spanish, Japanese, Chinese, Russian |
| Connectivity | USB 2.0, LIMS/SIL integration via DataLink® / DataLink Pro® |
| Data Export Formats | CSV, Excel™, PDF, SCA, BIO, JPEG, PNG, BMP |
| Regulatory Compliance | FDA 21 CFR Part 11 (audit trail, electronic signatures, role-based access), GLP/GMP-aligned |
| Power Supply | 100–240 V~, 50–60 Hz, 20 W max |
| Dimensions (W×D×H) | 28.7 × 27.1 × 28.9 cm |
| Weight | 8.38 kg |
| Warranty | 3 years (registration required) |
| Spare Parts Availability | 10 years |
| Software Updates | 3 years (excludes validation documentation) |
Overview
The Interscience Scan 500 Automated Colony Counter is an ISO 7218- and AOAC 977.27-compliant digital imaging system engineered for high-precision, high-throughput microbial enumeration in regulated laboratory environments. It employs advanced dark-field illumination coupled with a calibrated 1 MP CMOS color camera and M12 zoom lens to resolve colonies as small as 0.1 mm in diameter across standard petri dishes (Ø55–Ø90 mm). Unlike manual or basic optical counters, the Scan 500 applies pixel-level morphological segmentation algorithms to distinguish individual colonies—even tightly clustered or partially fused ones—while preserving spatial fidelity and enabling post-acquisition manual correction (addition/deletion, polygonal region definition). Its core measurement principle relies on contrast-enhanced image acquisition under dynamically optimized lighting conditions, followed by threshold-based binary conversion and watershed-based separation logic. This architecture ensures reproducible colony detection independent of operator subjectivity, delivering ≥98% counting accuracy validated against reference plate counts under routine QC conditions.
Key Features
- Automated colony enumeration with real-time processing at up to 1000 colonies per second
- Integrated inhibition zone analysis supporting disc diffusion (paper discs, Oxford cups, agar wells), with automatic detection, manual refinement, and classification per CLSI, EUCAST, CA-SFM (human/veterinary), and user-defined breakpoints
- Multi-color discrimination: simultaneous detection and classification of up to four colony colors per plate, with configurable exclusion of background or interfering hues
- Dark-field illumination system with six programmable configurations (top/bottom, white/black background) minimizing operator eye fatigue and maximizing edge contrast for low-pigment or translucent colonies
- Full traceability framework compliant with FDA 21 CFR Part 11: audit-trail logging, dual electronic signatures, role-based user permissions (administrator, analyst, reviewer), and immutable session records
- Seamless bidirectional integration with LIMS and SIL systems via USB-connected DataLink® or DataLink Pro® middleware, enabling end-to-end sample tracking from dilution/inoculation through enumeration
Sample Compatibility & Compliance
The Scan 500 accommodates all standard microbiological plating methods: pour plates, spread plates, spiral plating, and replica plating. It supports chromogenic and selective media—including those used in food safety (ISO 11133), pharmaceutical sterility testing (USP , Ph. Eur. 2.6.12), and clinical diagnostics—without requiring media-specific calibration. The system validates conformance to ISO 7218:2017 (microbiology of food and animal feeding stuffs), AOAC Official Method 977.27 (colony counter performance criteria), and regulatory expectations for data integrity outlined in FDA Guidance for Industry: Computerized Systems Used in Clinical Trials (2023) and EMA Annex 11. All firmware and software revisions are version-controlled and documented per GxP requirements; raw images, metadata, and processed results are stored with embedded timestamps, operator IDs, instrument serial numbers, and environmental context (e.g., dish ID, antibiotic name, strain designation).
Software & Data Management
Scan software (v5.x or later) runs natively on Windows 10/11 (Intel i5+, 4 GB RAM, 1280×1024+ display) and provides a validated, locked-down interface for assay configuration, image acquisition, result review, and report generation. Data export options include CSV, Excel™, PDF, SCA (Standard Colony Analysis), and BIO formats for interoperability with statistical process control (SPC) platforms and enterprise quality management systems (QMS). Image outputs are saved in lossless PNG/BMP or compressed JPEG at full sensor resolution. Audit trails record every user action—including parameter changes, manual corrections, and report approvals—with hash-secured immutability. When deployed with DataLink Pro®, the system generates machine-readable labels applied directly to dish rims, linking physical samples to digital records across instrumentation networks—including Interscience’s Spiral Auto, DiluFlow, and Whitley Anaerobic Workstations.
Applications
- Pharmaceutical microbiological quality control (environmental monitoring, water bioburden, product sterility)
- FDA-regulated food and beverage testing (aerobic plate count, coliforms, pathogens per ISO 4833-1, ISO 6579)
- Clinical microbiology labs performing AST (antibiotic susceptibility testing) and quantitative culture (e.g., urine CFU/mL)
- Contract research organizations (CROs) requiring ALCOA+ data integrity attributes (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available)
- Academic and industrial R&D labs conducting method validation, strain stability studies, or disinfectant efficacy testing
FAQ
Does the Scan 500 require annual recalibration?
No—its optical path and illumination geometry are factory-calibrated and mechanically stabilized. Users perform daily verification using included NIST-traceable calibration discs; full metrological revalidation is recommended every 24 months or after impact events.
Can the system validate against USP requirements for automated microbiological counting?
Yes—the Scan 500 satisfies USP criteria for instrument qualification (IQ/OQ/PQ), including repeatability (RSD ≤5% across five replicate plates), accuracy (recovery ≥95%), and robustness under variable lighting and media conditions.
Is remote software update supported over network?
No—updates are delivered via secure USB installation package to maintain air-gapped compliance in high-assurance environments; validation documentation accompanies each release.
How is data security enforced during LIMS transmission?
All LIMS/SIL communications use TLS 1.2+ encrypted channels; DataLink Pro® implements AES-256 encryption for stored session data and enforces certificate-based mutual authentication.
What happens if a colony falls below 0.1 mm detection threshold?
The system reports “sub-threshold objects” in a dedicated log with pixel-area metrics; users may manually annotate such features, though they are excluded from official CFU counts per ISO 7218 clause 7.3.