JIAHANG JH6100-A Laser Particle Size Analyzer
| Brand | JIAHANG |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Instrument Type | Laboratory Wet-Dispersion Laser Diffraction Particle Size Analyzer |
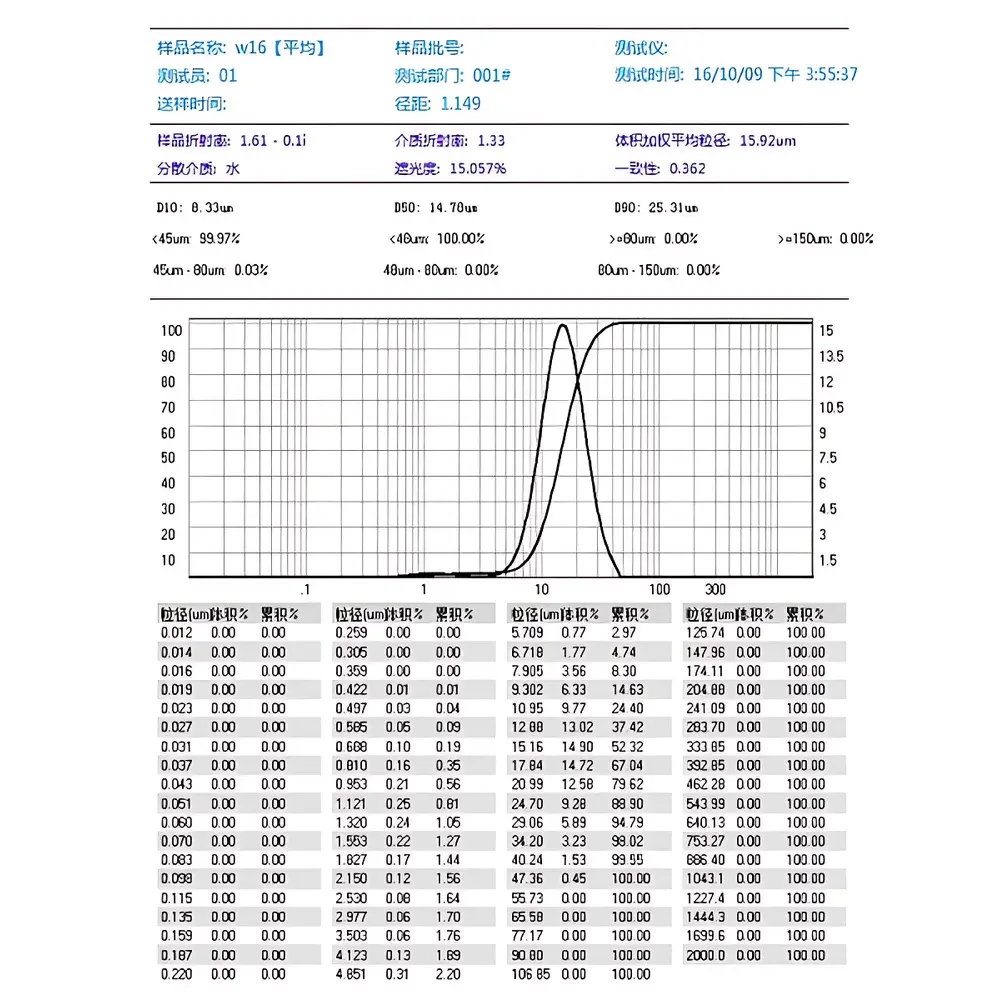
| Measurement Range | 0.01 µm – 2000 µm |
| Repeatability | <1% RSD (D50) |
| Measurement Time | ≤10 seconds per run |
| Dispersing Medium Volume | ≥180 mL (standard), optional 10 mL micro-circulation mode |
| Ultrasonic Power | 100 W with dry-run protection |
| Optical Alignment | Fully automated mechanical–optical dual-axis centering (≤15 s, 0.2 µm step resolution) |
| Detector Channels | 121 |
| Compliance | ISO 13320:2020, GB/T 19077–2016, USP <429>, FDA 21 CFR Part 11 ready (with audit trail, electronic signature, role-based access control) |
Overview
The JIAHANG JH6100-A is a high-precision laboratory laser diffraction particle size analyzer engineered for rigorous physical property characterization of powders, suspensions, emulsions, and colloidal dispersions. It operates on the fundamental principles of Mie scattering theory and convergent-beam Fourier transform optical design—enabling accurate size distribution analysis across an ultra-broad dynamic range from 0.01 µm to 2000 µm. Unlike conventional fixed-focal-length systems, its optimized convergent optical path delivers enhanced angular resolution at short focal distances, significantly improving detection sensitivity for sub-100 nm particles while maintaining robust quantification of coarse fractions up to 2 mm. The instrument integrates a fully sealed, vibration-isolated optical bench with a suspended chassis architecture to minimize environmental interference, ensuring long-term signal stability under routine lab conditions. All optical components—including dual fiber-coupled semiconductor lasers (λ = 635 nm and 650 nm, P > 10 mW)—are housed in a dust-resistant enclosure and managed by an embedded intelligent laser control system that monitors output power in real time, compensates for thermal drift, and achieves operational stability within 3 seconds after activation.
Key Features
- Convergent-Beam Fourier Optics: Enables high-resolution detection across the full 0.01–2000 µm range without segmentation or interpolation artifacts; eliminates traditional “gap zones” between detector arrays.
- Dual-Laser, Dual-Path Configuration (Optional High-End Model): Primary and auxiliary beams operate independently with wavelength-specific calibration—improving refractive index compensation accuracy for polydisperse or multi-component samples.
- Fully Automated Optical Alignment: Combines precision stepper motors, micron-grade linear guides, and closed-loop feedback software to achieve sub-micron beam positioning repeatability (<0.2 µm step resolution); alignment completes in ≤15 seconds without manual intervention.
- Micro-Circulation Dispersion System: Optimized wet dispersion loop requires only ≥180 mL dispersant (as low as 10 mL with optional micro-module); features quick-connect fluidic fittings, zero-residue drainage pathways, and anti-sedimentation flow dynamics.
- Intelligent Sample Handling Protocols: Includes dew-point monitoring to prevent window fogging, bubble-free sample introduction, ultrasonic dry-run protection (100 W, 40 kHz), and real-time obscuration feedback for precise sample dosing.
- Robust Mechanical Architecture: Sealed optical chamber, floating base mount, and corrosion-resistant wet-path materials (optional acid/alkali/solvent-compatible configuration available) ensure operational reliability in QC, R&D, and regulated GMP environments.
Sample Compatibility & Compliance
The JH6100-A accommodates a wide spectrum of sample types—including pharmaceutical actives, catalysts, battery cathode materials, ceramic slurries, polymer nanoparticles, and mineral pigments—across aqueous and organic media. Its boiling-type sample cell (190–600 mL capacity) supports both high-shear and low-energy dispersion protocols, while integrated variable-speed stirring (100–33950 rpm) and programmable ultrasonication (0–300 s, adjustable power) allow method optimization per ISO 13320:2020 Annex B guidelines. The system meets key international standards: ISO 13320:2020 (laser diffraction), GB/T 19077–2016 (Chinese national standard), and USP <429> (light scattering for pharmaceutical particle sizing). When configured with validated firmware and audit-trail-enabled software, it satisfies FDA 21 CFR Part 11 requirements for electronic records and signatures, supporting full 3Q qualification (IQ/OQ/PQ) documentation packages for regulated laboratories.
Software & Data Management
The proprietary JH-Particle Analysis Suite is built on a secure, modular architecture compliant with GLP/GMP data integrity principles. It provides role-based user permissions, immutable raw data storage, time-stamped audit trails, and electronic signature workflows—all traceable to individual operators and instrument events. Reporting modules support export to Word, Excel, BMP, and plain-text formats; statistical overlays enable comparative D10/D50/D90 analysis across batches or process steps. Distribution modeling includes free, Rosin-Rammler, log-normal, and user-defined percentile interpolation. Custom report templates can be defined per regulatory need—e.g., volume-weighted vs. number-weighted distributions, cumulative/differential plots, or sieve-equivalent grading tables. Multi-language UI (English, Chinese, customizable) ensures global deployment readiness without code-level modification.
Applications
The JH6100-A serves critical measurement needs across multiple sectors: In pharmaceutical development, it characterizes milled APIs, spray-dried excipients, and nanoemulsion droplets per ICH Q5A and Q5C guidance; in advanced materials R&D, it validates synthesis consistency of quantum dots, metal-organic frameworks (MOFs), and Si/C anode composites for Li-ion batteries; in quality control labs, it monitors batch-to-batch uniformity of pigment dispersions, toner particles, and abrasive slurries against internal SOPs and ASTM D4295. Its ability to resolve sub-100 nm populations with <1% repeatability makes it suitable for nanoparticle toxicology screening and nanomedicine formulation development where primary particle size governs biological interaction.
FAQ
Does the JH6100-A comply with FDA 21 CFR Part 11?
Yes—when deployed with enabled audit trail, electronic signature, and permission-controlled user roles, the system meets all technical and procedural requirements for electronic records and signatures in regulated environments.
Can the instrument measure samples in organic solvents?
Standard configurations support water-based dispersions; optional chemically resistant wet-path kits (PTFE, sapphire windows, solvent-grade seals) are available for use with acetone, isopropanol, xylene, and other aggressive solvents.
What is the minimum sample volume required for testing?
The standard circulation mode requires ≥180 mL dispersant; an optional micro-circulation module enables full-range analysis using as little as 10 mL.
How does the auto-alignment system maintain long-term accuracy?
It performs real-time centroid tracking of the primary laser beam via quadrant photodiode feedback, dynamically adjusting mirror position to maintain beam passage through the exact geometric center of the 121-channel detector array.
Is method validation support available for pharmaceutical applications?
Yes—JIAHANG provides comprehensive 3Q documentation packages, IQ/OQ protocol templates, and assistance with method transfer per ICH Q2(R2) guidelines.