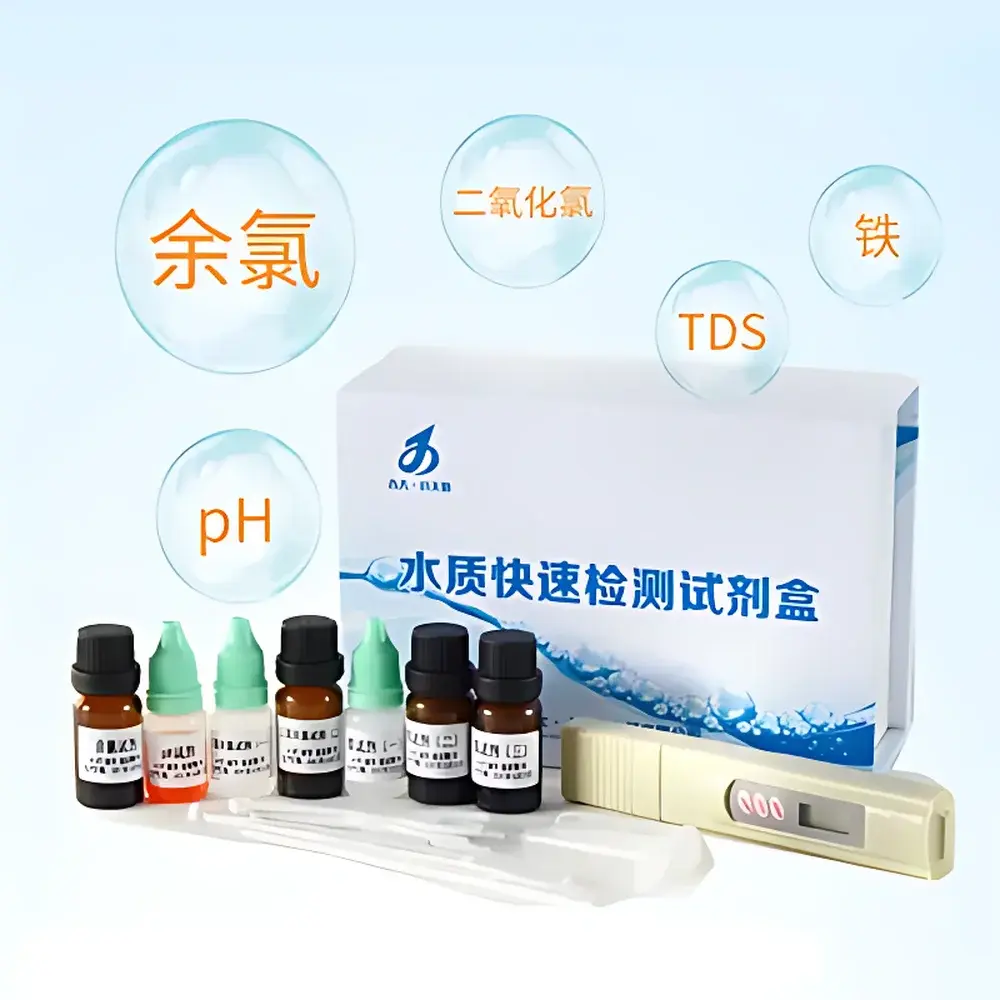
Jilin University Xiaotianshe Water Quality Rapid Test Kit
| Brand | Jilin University Xiaotianshe |
|---|---|
| Origin | Jilin, China |
| Manufacturer Type | Direct Manufacturer |
| Product Category | Domestic |
| Model | Water Quality Rapid Test Kit |
| Price Range | USD 140 – 7,200 (based on configuration) |
| Instrument Type | Portable |
Overview
The Jilin University Xiaotianshe Water Quality Rapid Test Kit is a field-deployable, multi-parameter colorimetric assay system engineered for on-site verification of critical drinking water quality parameters in accordance with China’s national standard GB 5749–2006 (Standard for Drinking Water Quality). Designed for use by municipal water operators, rural water safety inspectors, emergency response teams, and environmental health practitioners, the kit employs standardized N,N-diethyl-p-phenylenediamine (DPD) chemistry for free chlorine and chlorine dioxide, phenanthroline-based spectrophotometry for iron (Fe²⁺/Fe³⁺), pH indicator strips calibrated to NIST-traceable buffer standards, and a digital TDS pen utilizing calibrated conductivity-to-TDS conversion algorithms. All assays are optimized for ambient temperature operation (10–30 °C), require no external power or laboratory infrastructure, and deliver quantitative or semi-quantitative results within 1–10 minutes—enabling real-time decision-making during routine surveillance, post-disaster assessments, or source water vulnerability evaluations.
Key Features
- Five-parameter capability: Simultaneous determination of free residual chlorine, chlorine dioxide, total iron, pH, and TDS—covering all core indicators mandated under GB 5749–2006 for potable water compliance screening.
- DPD-based methodology: Free chlorine detection via DPD colorimetry (detection range: 0.02–5.0 mg/L Cl₂; LOD: 0.02 mg/L); chlorine dioxide quantification using chlorophenol red–DPD sequential oxidation (detection range: 0.01–2.0 mg/L ClO₂; LOD: 0.01 mg/L).
- Iron analysis: Ferrous/ferric ion detection via 1,10-phenanthroline complexation yielding an orange-red chromophore (detection range: 0.05–3.0 mg/L Fe; LOD: 0.05 mg/L), validated against ISO 6597:2000.
- pH assessment: Dual-range indicator strips (pH 4.5–9.0, ±0.2 unit accuracy) with visual color-matching reference chart, traceable to NIST SRM 186c buffer solutions.
- TDS measurement: Digital handheld TDS meter (0–9990 ppm range, ±2% full scale), temperature-compensated (25 °C), calibrated with KCl standard solution (1413 µS/cm).
- Portability & ruggedness: All reagents supplied in sealed, light-protected vials with desiccant; test cards and cuvettes pre-packaged in waterproof, impact-resistant carry case compliant with IP65 ingress protection.
Sample Compatibility & Compliance
The kit is validated for use with tap water, groundwater, surface water (after filtration if turbid >5 NTU), and treated effluent meeting Class I–III criteria per GB 3838–2002. It excludes seawater, highly colored industrial wastewater, or samples containing strong oxidants (e.g., permanganate >0.5 mg/L) or reducing agents (e.g., sulfite >1 mg/L) without pretreatment. All analytical procedures align with methodological principles referenced in ISO 7393-2 (chlorine), ISO 11258 (chlorine dioxide), ISO 6597 (iron), and ASTM D1293 (pH). While not certified for regulatory reporting under GLP or EPA methods, it satisfies WHO Guidelines for Drinking-water Quality (4th ed.) Tier 1 field screening requirements and supports internal QA/QC protocols aligned with ISO/IEC 17025:2017 clause 7.2.2 (method validation).
Software & Data Management
This is a non-instrumental, manual assay system—no embedded firmware, Bluetooth, or cloud connectivity. However, optional companion tools include a downloadable Excel-based data logging template (compatible with Windows/macOS) supporting batch ID, sampling location (GPS coordinates manually entered), date/time stamp, analyst ID, and pass/fail flagging per GB 5749–2006 limits. Audit trails are maintained through physical record retention: each test card includes a unique lot number and expiration date; reagent vials feature batch-specific calibration certificates traceable to CNAS-accredited reference laboratories. For organizations operating under GMP or ISO 9001 frameworks, the kit supports documented procedure adherence via SOP templates provided upon request (e.g., “SOP-WQ-RTK-01: Field Use of Rapid Test Kit”).
Applications
- Routine distribution system monitoring: Verification of disinfectant residual at entry points, storage tanks, and endpoint taps to ensure maintenance of minimum CT values.
- Rural water safety programs: Low-cost, operator-independent verification of iron-induced staining risk and pH-driven corrosion potential in aging pipe networks.
- Emergency response: Rapid triage of flood-affected wells or temporary water supplies following natural disasters or infrastructure failure.
- Public health outreach: Visual demonstration kits for community education on chlorine decay kinetics, iron precipitation thresholds, and TDS-related taste acceptability.
- Pre-commissioning verification: On-site validation of treatment plant output prior to formal certification testing per GB/T 5750 series.
FAQ
What is the shelf life of the reagents?
Unopened reagent vials retain stability for 18 months when stored at 2–8 °C in darkness. After opening, DPD powder must be used within 30 days; liquid reagents remain stable for 90 days under refrigeration.
Can this kit be used for wastewater or industrial effluent?
No. The assay chemistries are optimized for low-turbidity, low-organic-load potable water matrices. High COD (>50 mg/L), suspended solids (>10 mg/L), or interfering ions (e.g., bromide >0.5 mg/L) will cause positive bias in chlorine measurements and false negatives in iron assays.
Is calibration required before each use?
The TDS pen requires daily two-point calibration (0 ppm and 1413 µS/cm KCl) per manufacturer instructions. Colorimetric tests do not require instrument calibration but rely on strict adherence to reaction time, mixing protocol, and ambient lighting conditions per included SOP.
Does the kit meet international regulatory requirements such as US EPA or EU Directive 98/83/EC?
It is designed and validated specifically for compliance with GB 5749–2006. While measurement principles are harmonized with ISO standards, formal recognition under US EPA Methods 334.0 or EN ISO 7890-3 requires additional third-party validation per local regulatory authority protocols.
How are results documented for audit purposes?
Each test generates a physical record: filled-out result sheet with analyst signature, sample ID, and timestamp; photo documentation of color development (optional); and retained used test cards stored with batch logs for minimum 2 years—sufficient for internal audits and GB 5749 implementation reviews.