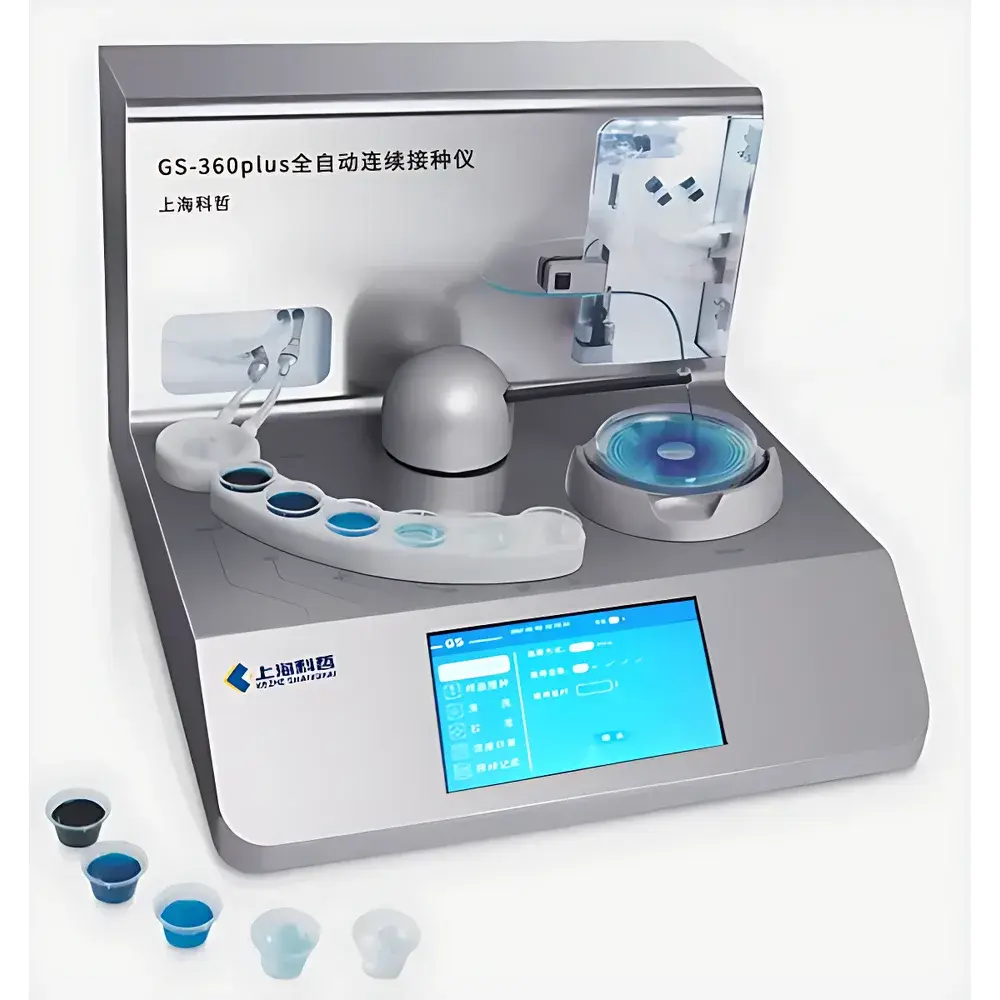
KEZHE SHANGHAI GS-360Plus Fully Automated Continuous Dilution and Inoculation System
| Brand | KEZHE SHANGHAI |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Domestic |
| Model | GS-360Plus |
| Price Range | USD 28,000 – 42,500 |
Overview
The KEZHE SHANGHAI GS-360Plus Fully Automated Continuous Dilution and Inoculation System is an integrated microbiological workstation engineered for precision serial dilution and quantitative surface inoculation onto agar plates. Based on gravimetric and volumetric fluid handling principles, the system performs up to five consecutive 1:10 dilutions—each step executed with calibrated positive-displacement syringes—followed by controlled deposition of defined sample volumes (50–200 µL) onto standard Petri dishes. Designed for compliance-driven laboratory environments, it supports standardized microbial enumeration protocols aligned with ISO 4833-1:2013 (microbiology of food and animal feeding stuffs), AOAC Official Methods of Analysis®, and USP and . Its closed-loop architecture minimizes operator intervention while maintaining traceability across dilution steps, sample identity, and plate positioning—critical for GLP and GMP-regulated quality control laboratories.
Key Features
- PTFE-coated inoculation needle ensures zero carryover between samples and eliminates biofilm adhesion during high-throughput runs.
- Dynamic agar-thickness compensation mechanism automatically adjusts needle height and contact force in real time, accommodating standard 90 mm and optional 150 mm Petri dishes with agar depths ranging from 3 mm to 8 mm—preventing surface scratching or penetration.
- High-precision glass syringe drive system delivers ≤0.5% volumetric accuracy per aspiration and dispensing cycle, validated per ISO 8655-5 standards.
- Five-step programmable serial dilution module integrates seamlessly with inoculation sequencing—no manual tube transfer required.
- Four configurable inoculation patterns: Spiral (logarithmic spiral deposition per ISO 7218), Uniform (even radial distribution), Circular (concentric ring deposition), and Pour (edge-to-center coverage mimicking manual pour-plate technique).
- Integrated wash cycle uses dual-solvent path (deionized water + ethanol or validated disinfectant) with pressure-controlled rinse and dry phases—ensuring cross-contamination risk below 1 CFU per 10⁴ operations.
- Embedded industrial-grade microprocessor with real-time event logging, onboard non-volatile memory (≥10,000 operation records), and USB/RS-232 interfaces for external LIMS integration.
Sample Compatibility & Compliance
The GS-360Plus accommodates liquid suspensions of bacteria, yeasts, molds, and spores—including viscous matrices such as milk homogenates, soil extracts, and fermented broth. It supports both aerobic and anaerobic culture media (e.g., Tryptic Soy Agar, Plate Count Agar, Sabouraud Dextrose Agar) without modification. All wetted components comply with USP Class VI biocompatibility requirements. The system meets electromagnetic compatibility (EMC) per IEC 61326-1 and electrical safety per IEC 61010-1. Audit trails include timestamped operator ID, sample ID, dilution factor, inoculation volume, pattern selection, and cleaning cycle execution—fully compliant with FDA 21 CFR Part 11 when paired with validated third-party LIMS or ELN platforms.
Software & Data Management
The GS-360Plus ships with two dedicated software modules: (1) Dilution Sequence Manager v3.2, enabling user-defined gradient schemes (e.g., 10⁻¹ to 10⁻⁵), error-checking logic for missing dilution steps, and automatic calibration log export; and (2) Inoculation Pattern Studio v4.1, supporting custom spiral pitch definition, zone-based colony density mapping, and overlay visualization of theoretical vs. actual colony distribution. Data output formats include CSV, PDF report (with embedded QC charts), and XML for direct import into laboratory information management systems (LIMS). All software binaries are digitally signed and version-controlled; firmware updates require dual-password authentication and generate immutable update logs.
Applications
- Quantitative microbial load testing in pharmaceutical raw materials, excipients, and finished dosage forms per USP .
- Environmental monitoring in cleanrooms and aseptic processing suites (ISO 14644-1 Class 5–8).
- Food safety validation including shelf-life studies, challenge testing, and HACCP verification.
- Water and wastewater analysis per ISO 9308-1 and EPA Method 1603.
- Antimicrobial efficacy testing (e.g., ISO 22196, JIS Z 2801) requiring reproducible inoculum density.
- Research applications involving strain isolation, mutant screening, and bioburden correlation studies.
FAQ
Does the GS-360Plus support regulatory audit requirements for electronic records?
Yes—when operated with validated software configuration and enabled audit trail settings, it satisfies data integrity criteria under ALCOA+ principles and provides full traceability for FDA, EMA, and PMDA inspections.
Can the system handle viscous or particulate-laden samples without clogging?
The PTFE needle bore (inner diameter ≥0.8 mm) and pulse-free syringe actuation minimize shear-induced aggregation; optional pre-filtration adapters (0.45 µm) are available for highly particulate suspensions.
Is method validation support provided by KEZHE?
KEZHE offers IQ/OQ documentation templates, performance qualification (PQ) test protocols, and on-site installation qualification services upon request—aligned with ISO/IEC 17025 and ASTM E2500 guidelines.
What maintenance intervals are recommended for long-term reliability?
Daily: Needle rinse and visual inspection; Quarterly: Syringe seal replacement and gravimetric accuracy verification; Annually: Full optical encoder recalibration and mechanical alignment certification.