Kingfar ErgoLAB Human-Machine-Environment Synchronization Cloud Platform
| Brand | Kingfar |
|---|---|
| Model | ErgoLAB |
| Type | Cloud-Based Laboratory Information Management System (LIMS) for Human Factors & Ergonomics Research |
| Deployment | SaaS |
| Compliance Framework | Supports GLP/GMP-aligned audit trails, ISO 9241-210 (Human-Centered Design), ASTM F2500 (VR/AR Human Factors), USP <1058> (Analytical Instrument Qualification), FDA 21 CFR Part 11 (Electronic Records & Signatures) |
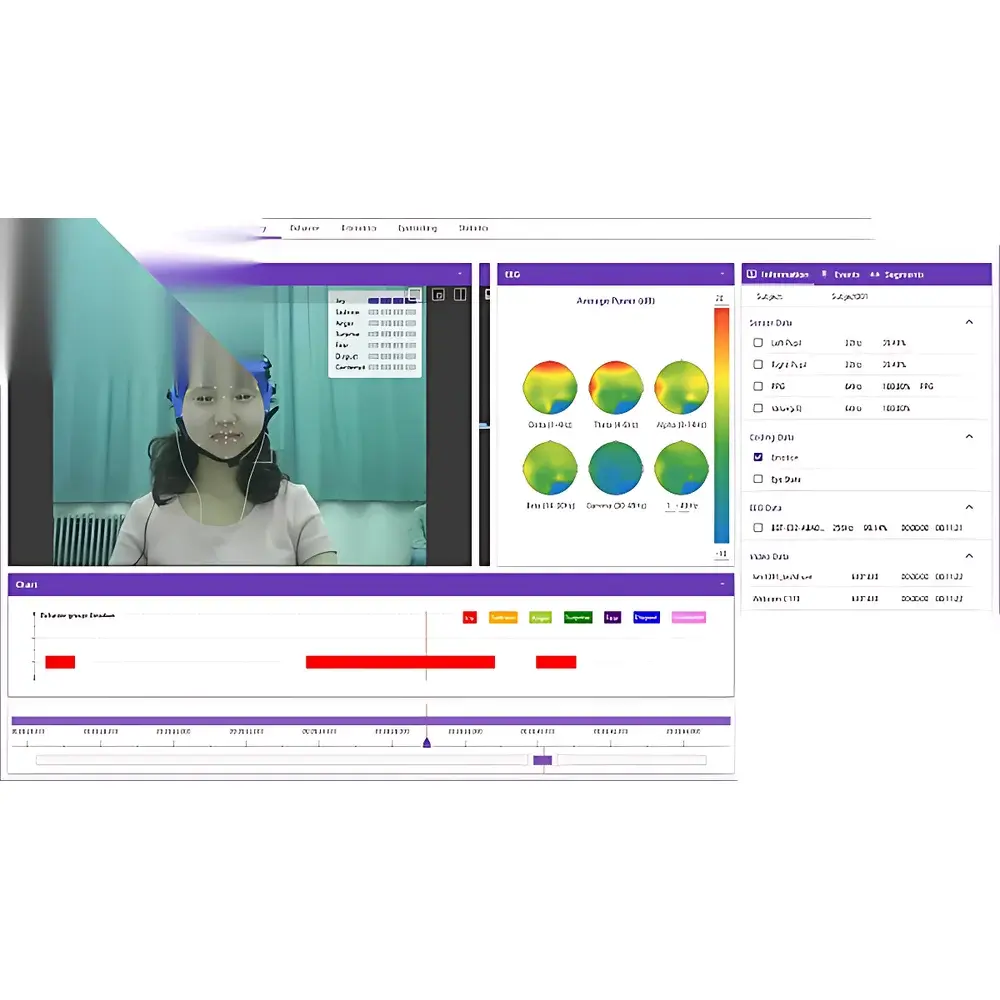
| Data Synchronization Precision | Sub-millisecond (<1 ms) via hardware-triggered TTL and software-based LSL (Lab Streaming Layer) integration |
| Supported Signal Modalities | 35+ concurrent streams including EEG, fNIRS, eye-tracking, EDA/GSR, EMG, ECG, HRV, RESP, PPG, SpO₂, SKT, EOG, facial expression (AU-coded), motion capture, VR/AR interaction logs, web/mobile UI telemetry, environmental sensors (temp, humidity, noise, illuminance, PM2.5), and spatial trajectory data (indoor/outdoor/VR) |
Overview
The Kingfar ErgoLAB Human-Machine-Environment Synchronization Cloud Platform is a purpose-built, cloud-native Laboratory Information Management System (LIMS) engineered for rigorous human factors, ergonomics, and cognitive engineering research. Unlike conventional data acquisition suites or generic lab software, ErgoLAB implements a systems-level architecture grounded in the Human-Cyber-Physical Systems (HCPS) framework. It enables time-synchronized, multi-modal data acquisition across three interdependent domains: the human information system (cognitive load, attention, decision latency), the human physical system (physiology, biomechanics, behavior), and the integrated human-machine-environment interface (interaction logs, spatial navigation, environmental context). Built on a microservices architecture with containerized deployment, ErgoLAB operates as a validated SaaS platform—fully compliant with ISO/IEC 17025 traceability requirements for measurement uncertainty documentation and supporting full ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) for regulatory submissions.
Key Features
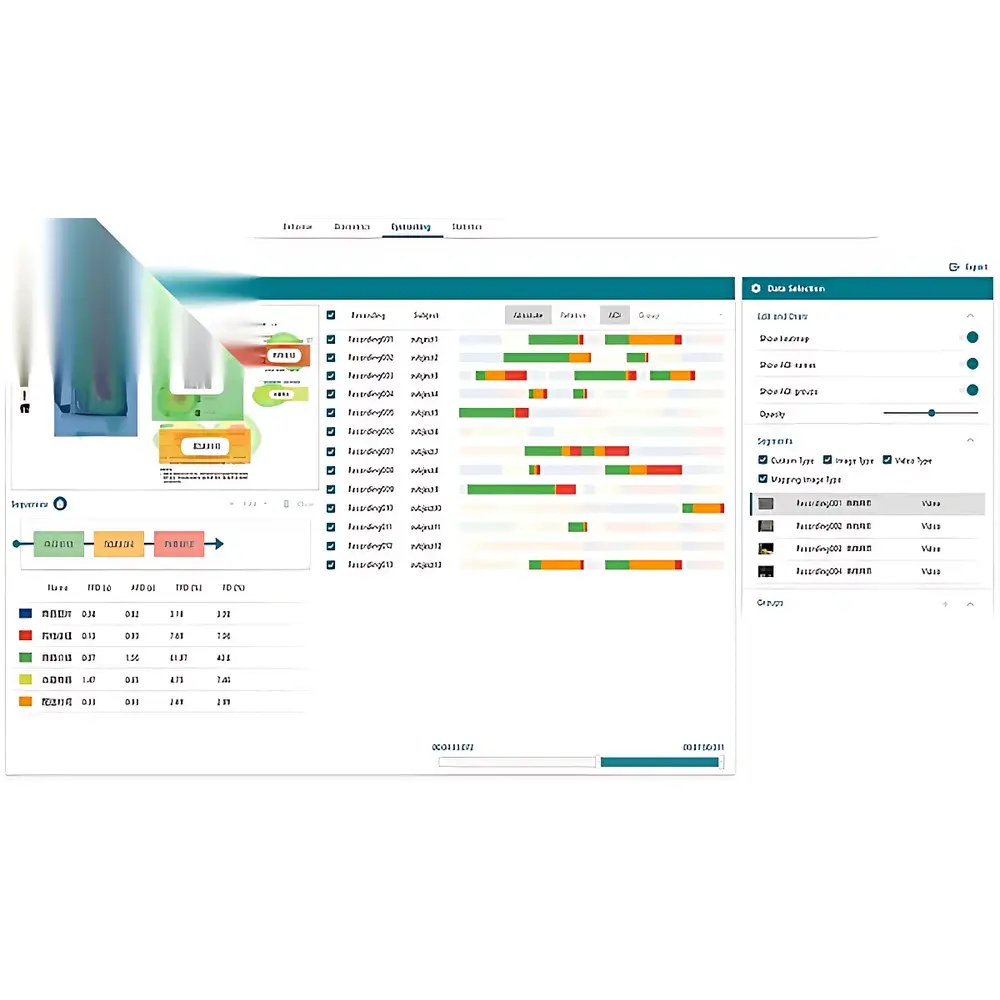
- End-to-End Cloud Workflow Integration: Unified management of project lifecycle—from cloud-hosted experimental protocol design and participant scheduling to real-time synchronized data ingestion, signal processing, statistical modeling, and automated report generation.
- Multi-Modal Synchronization Engine: Sub-millisecond temporal alignment across 35+ heterogeneous data sources via hybrid triggering: hardware TTL pulses for EEG/fNIRS/eye-tracking, LSL-compliant streaming for VR/AR/game engines (Unity, Unreal), and RESTful API hooks for web/mobile applications and third-party stimulus delivery tools (E-Prime, SuperLab, Tobii Pro Lab).
- Modular Experimental Design Studio: Visual, timeline-based authoring environment supporting nested randomization, counterbalancing, adaptive branching, and multi-channel stimulus presentation—including text, audio, static images, 360° video, live websites, native mobile apps, and interactive prototypes. Pre-integrated normative datasets and validated paradigms (NASA-TLX, PANAS, Stroop, MOT, attentional blink) accelerate protocol development.
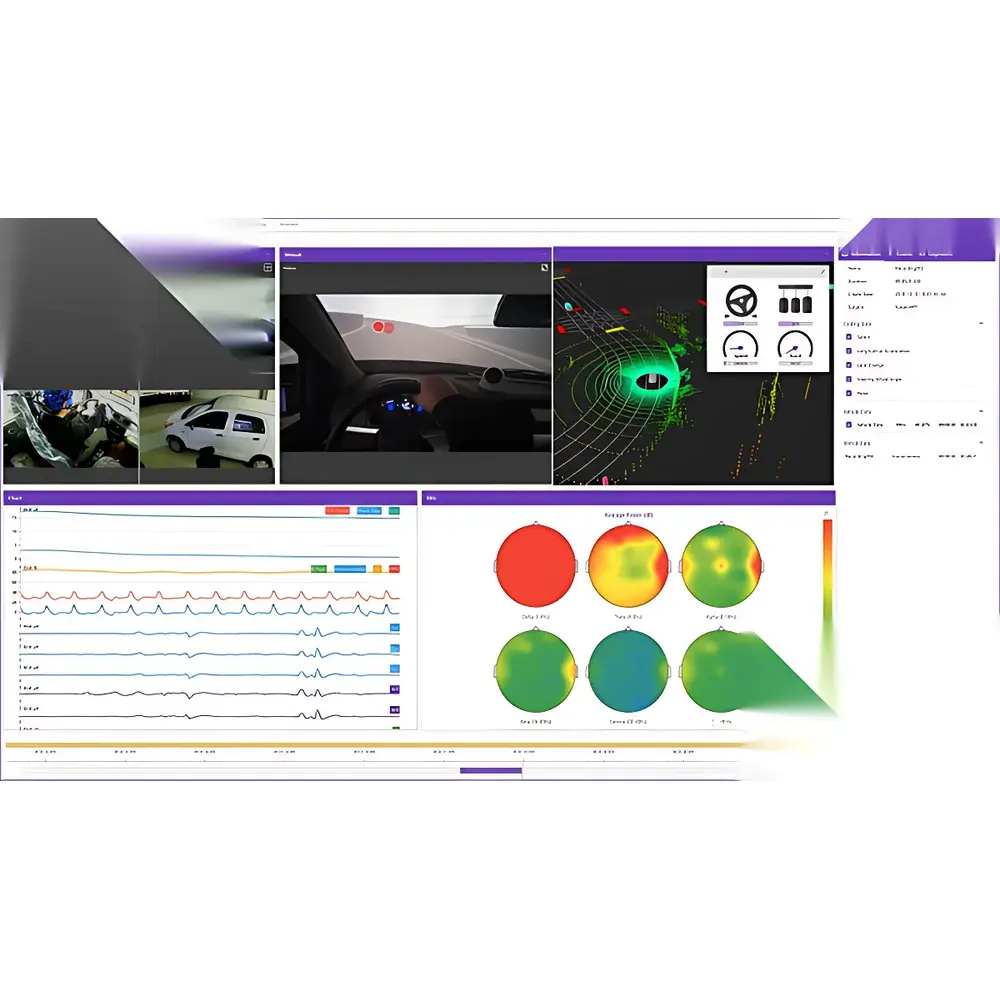
- Domain-Specific Real-Time Sync Modules: Dedicated, pre-validated synchronization bridges for ErgoVR (immersive virtual reality), ErgoAI (high-fidelity driving simulation), and ErgoSIM (controlled environmental chamber studies), each delivering domain-optimized metadata tagging, event ontology mapping, and compliance-ready calibration logs.
- Open Analytics Architecture: Native support for export of raw time-series data (CSV, MATLAB .mat, HDF5) and processed feature vectors; direct integration with Python (MNE, SciPy, scikit-learn), MATLAB (EEGLAB, FieldTrip), and R (eegUtils, eyetrackingR); extensible via REST APIs and WebSocket endpoints for custom ML pipeline integration.
Sample Compatibility & Compliance
ErgoLAB is designed for use in academic laboratories, clinical trial units, automotive R&D centers, aerospace human-systems integration labs, and industrial safety departments conducting GLP-, GCP-, or ISO 13485-regulated studies. The platform natively supports data ingestion from FDA-cleared and CE-marked instrumentation—including medical-grade EEG systems (e.g., g.tec, Brain Products), Class II physiological monitors (BIOPAC, ADInstruments), ISO 15008-compliant eye-trackers (Tobii, SMI), and calibrated environmental sensor networks (Onset HOBO, Vaisala). All data handling workflows conform to ISO/IEC 27001 security controls, with role-based access, encrypted at-rest and in-transit storage (AES-256), and immutable audit logs meeting FDA 21 CFR Part 11 requirements for electronic signatures and record retention. Validation documentation (IQ/OQ/PQ protocols, risk assessments per ISO 14971) is available upon request for regulated deployments.
Software & Data Management
ErgoLAB’s cloud infrastructure provides centralized, version-controlled experiment templates, participant metadata management, and granular permission hierarchies (principal investigator, analyst, technician, external collaborator). Each dataset is assigned a persistent DOI and FAIR-compliant metadata schema (based on BIDS extensions for multimodal human neuroscience). The embedded signal processing engine includes artifact rejection (ICA, wavelet denoising), spectral analysis (Welch, Morlet wavelets), time-frequency decomposition, and event-related potential (ERP) averaging—all configurable via GUI or scriptable pipelines. Integrated visualization modules generate publication-ready plots: heatmaps for gaze distribution, trajectory overlays for spatial behavior, coherence spectra for cross-modal coupling, and multivariate radar charts for holistic workload assessment. All reports are exportable as PDF, HTML, or interactive Dash/Plotly dashboards, with optional integration into institutional data repositories (e.g., Figshare, OSF).
Applications
- Automotive Human Factors: Objective evaluation of driver distraction, HMI usability, and ADAS trust calibration using synchronized vehicle CAN bus, eye-tracking, EEG, and driving simulator telemetry.
- Aviation & Defense: Cognitive workload assessment during mission-critical tasks under variable G-load, vibration, and auditory masking—correlating neurophysiological markers with operational performance metrics.
- Healthcare Interface Design: Usability validation of EHR interfaces, surgical navigation systems, and telehealth platforms using gaze-behavior alignment, error rate analysis, and stress biomarker trajectories (EDA, HRV).
- Consumer Product Development: Immersive testing of AR retail experiences, smart home ecosystems, or wearable device interactions—capturing implicit preference (facial AUs, pupillometry), explicit feedback (embedded questionnaires), and behavioral engagement (dwell time, path efficiency).
- Academic Cognitive Science: Reproducible, multi-lab studies leveraging standardized paradigms and shared normative baselines for cross-population comparisons in attention, memory, and emotion regulation research.
FAQ
Does ErgoLAB require on-premise server installation?
No. ErgoLAB is delivered as a fully managed SaaS platform hosted on ISO 27001-certified cloud infrastructure (AWS GovCloud or Azure Government options available for classified research). Local edge devices (sensors, VR headsets) connect via secure TLS tunnels.
Can I integrate proprietary algorithms or machine learning models?
Yes. ErgoLAB provides documented REST APIs, WebSocket feeds for real-time stream access, and a Python SDK for embedding custom preprocessing, feature extraction, or classification logic directly into the analysis pipeline.
How is data privacy and participant consent managed?
The platform includes built-in GDPR/CCPA-compliant consent management: dynamic digital consent forms with version history, biometric data anonymization options (k-anonymity, differential privacy presets), and granular data export controls aligned with IRB/ethics board requirements.
Is validation support provided for regulated environments?
Yes. Kingfar supplies comprehensive validation packages—including URS, FRS, IQ/OQ/PQ test scripts, traceability matrices, and change control documentation—alongside on-site or remote qualification support for GxP compliance.
What training and technical support options are available?
Comprehensive onboarding includes hands-on workshops for experimental design, data synchronization troubleshooting, and statistical interpretation. 24/7 enterprise support SLAs, dedicated customer success managers, and quarterly platform update briefings are included in annual subscription tiers.