KURABO mini480 Nucleic Acid Extraction System
| Brand | KURABO |
|---|---|
| Origin | Japan |
| Manufacturer Status | Authorized Distributor |
| Product Origin | Imported |
| Model | mini480 |
| Price Range | USD 6,800–13,600 (est.) |
| Automation Level | Semi-Automated Assistant |
| Throughput | 1–48 samples per run |
| Sample Volume Range | 20–200 µL |
| Processing Time per Run | 20–40 minutes |
| Membrane Thickness | 80 µm |
| Elution Yield | up to 5 µg DNA from 200 µL whole blood |
| Purity | DNA A260/A280 > 1.7 |
| Workflow Stages | Binding, Washing, Elution (3-pressure-step protocol) |
| Footprint | Benchtop-compatible |
| Optional Modules | 2 mL tube handling (8-channel), high-volume sample processing |
Overview
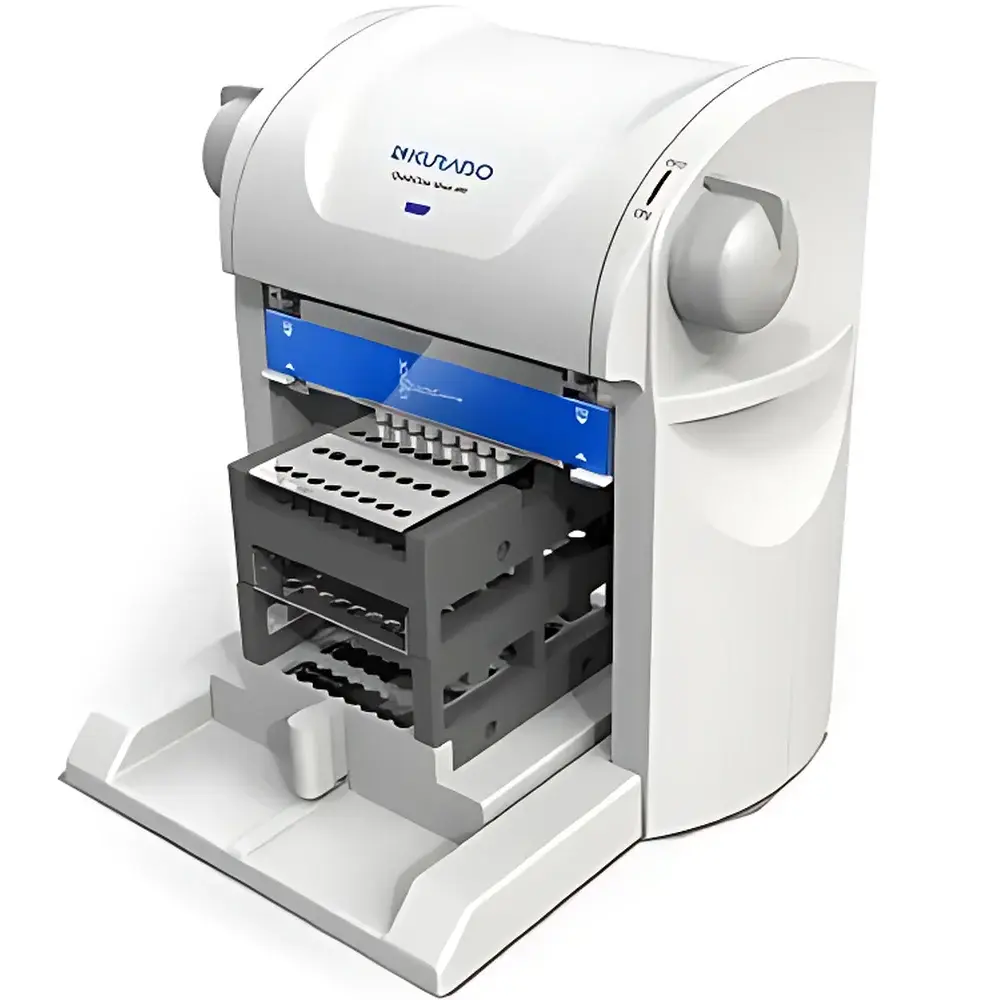
The KURABO mini480 Nucleic Acid Extraction System is a compact, pressure-driven benchtop platform engineered for rapid, centrifuge-free isolation of high-integrity DNA and RNA from diverse biological matrices. Unlike conventional magnetic bead- or column-based systems requiring centrifugation or external vacuum manifolds, the mini480 employs KURABO’s proprietary porous membrane technology—featuring an ultra-thin 80 µm asymmetric polymer membrane—to achieve solid-phase nucleic acid binding under controlled pneumatic pressure. This principle leverages capillary-driven flow and differential affinity under positive pressure gradients, enabling efficient lysis, selective binding, on-cartridge washing, and low-volume elution—all within a sealed, integrated cartridge. Designed specifically for biosafety cabinet integration, the system eliminates aerosol generation risks associated with manual pipetting or centrifuge lid opening, aligning with ISO 15189 and CLIA pre-analytical requirements for molecular diagnostics laboratories.
Key Features
- Benchtop footprint (W × D × H: 290 × 330 × 280 mm) and lightweight construction (<12 kg) enable flexible placement inside Class II biosafety cabinets or laminar flow hoods without compromising workflow ergonomics.
- Semi-automated operation: users load samples and cartridges, select pre-validated protocols via intuitive touchscreen interface, then initiate fully automated binding-wash-elution cycles—no manual aspiration, centrifugation, or magnet handling required.
- Flexible throughput: accommodates 1–48 samples per run; optimized for small-batch clinical testing, research validation, or urgent pathogen screening where rapid turnaround is critical.
- Three-stage pressure-controlled protocol ensures consistent fluid dynamics across all channels, minimizing inter-sample variability and supporting reproducible yields (up to 5 µg DNA from 200 µL whole blood).
- Modular design supports optional 2 mL tube adapters (8-channel configuration) for larger-volume extractions (e.g., plasma, CSF, or environmental water concentrates), extending utility beyond standard swab/blood workflows.
Sample Compatibility & Compliance
The mini480 processes a broad spectrum of input materials without preprocessing: nasopharyngeal/oropharyngeal swabs in viral transport media, whole blood (EDTA/K2EDTA), serum/plasma, cultured cells, fresh/frozen animal tissues, bacterial pellets, fungal mycelia, and environmental microbial lysates. All extraction protocols are validated per ISO 20387:2018 (biobanking) and support GLP-compliant documentation when paired with optional audit-trail-enabled software (see Software & Data Management). The system meets IEC 61010-1 safety standards for laboratory equipment and operates within ambient lab conditions (15–30°C, 30–80% RH non-condensing). Cartridges are sterile, single-use, and certified DNase/RNase-free—eliminating carryover risk and fulfilling USP and FDA 21 CFR Part 11 data integrity prerequisites when used with compliant LIMS integration.
Software & Data Management
The embedded control software provides protocol selection, real-time pressure monitoring, cycle progress visualization, and error logging with timestamped event records. Optional PC-based KURABO ExtractLink™ software enables remote scheduling, batch report generation (PDF/CSV), electronic signature capture, and full audit trail functionality—including user login history, parameter changes, and run completion status—for compliance with FDA 21 CFR Part 11 and EU Annex 11. Raw run logs are exportable for QC review or internal SOP verification, and firmware updates are delivered via secure USB authentication to maintain regulatory alignment.
Applications
- Clinical molecular labs performing rapid SARS-CoV-2, influenza, RSV, or HPV nucleic acid testing under CLIA/CAP-accredited workflows.
- Research institutions isolating high-purity DNA/RNA for qPCR, digital PCR, NGS library preparation, or methylation analysis—particularly where inhibitor-sensitive downstream assays demand A260/A280 > 1.7 and A260/A230 > 2.0.
- Biosafety Level 2/3 facilities requiring closed-system nucleic acid purification to minimize operator exposure during high-risk sample handling.
- Field-deployable or satellite lab settings where space, power constraints, or lack of centrifuge infrastructure preclude traditional extraction platforms.
- Biobanks validating extraction consistency across heterogeneous donor cohorts using standardized, pressure-based membrane chemistry.
FAQ
Does the mini480 require centrifugation or magnetic separation equipment?
No—the system uses only regulated air pressure to drive lysis, binding, washing, and elution through integrated membranes; no external centrifuge, magnets, or vacuum pumps are needed.
Are reagents and consumables supplied by KURABO only?
Yes—proprietary extraction cartridges and chemistries are designed exclusively for the mini480 platform to ensure membrane compatibility, pressure tolerance, and yield reproducibility; third-party kits are not supported.
Can the mini480 be integrated into automated liquid handling workflows?
While not natively robotic-arm compatible, its standardized cartridge dimensions and API-accessible status reporting (via optional RS-232/USB interface) allow integration with middleware systems for semi-automated sample-to-result pipelines.
What is the shelf life and storage condition for unopened cartridges?
Cartridges retain full performance for 18 months when stored at 4–25°C in original packaging; no refrigeration is required prior to use.
Is technical support available outside Japan?
Yes—authorized distributors provide local service contracts, application training, and ISO 17025-accredited calibration services aligned with regional regulatory expectations (e.g., UKAS, DAkkS, ANAB).