LA-ICP-TOFMS Bioimaging Service
| Brand | Chemlab Pro |
|---|---|
| Origin | Shanghai, China |
| Distributor Type | Authorized Regional Distributor |
| Product Origin | Domestic (China) |
| Model | LA-ICP-TOFMS Bioimaging Service |
| Quotation | Upon Request |
| Service Experience | 3–5 Years |
| Sample Types | Animal & Plant Tissue Sections, Fresh/Dried Plant Specimens |
| Turnaround Time | 5–10 Business Days |
| Sample Preparation Requirements | Cryosections or resin-embedded sections (OCT or epoxy resin acceptable) |
| minimum thickness | 10 µm |
| Sample Mounting | Fresh/dried plant samples must be flat-mounted on conductive glass or indium-tin oxide (ITO)-coated slides |
| max. dimensions | 9 cm × 9 cm × 0.8 cm |
| Certified Reference Materials | NIST SRM 1486 Bone Ash, GBW 07605 Spinach Leaves, GBW 07607 Citrus Leaves |
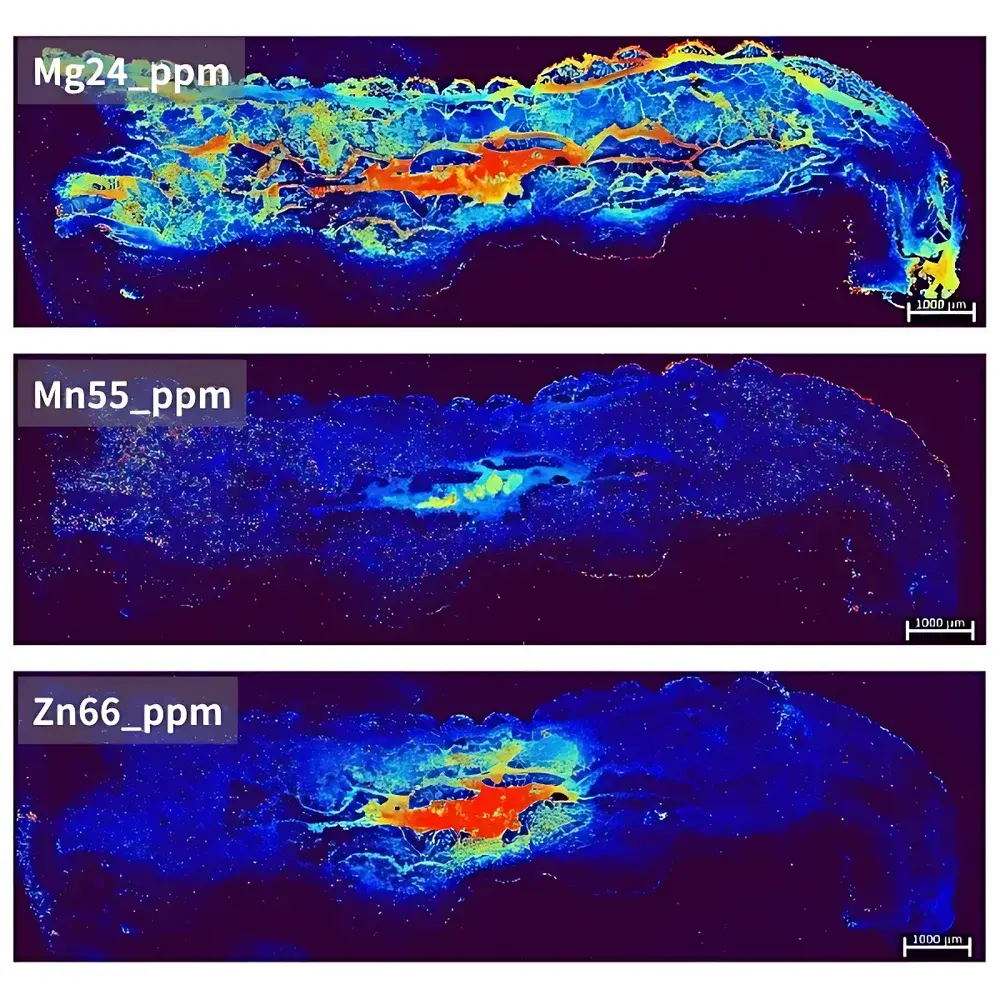
| Target Application | *Ophiocordyceps sinensis* (caterpillar fungus) elemental mapping of insect body |
| Spatial Resolution | 20 µm |
| Pixel Count | 187,500 |
| Imaging Area | 15 mm × 5 mm |
Overview
LA-ICP-TOFMS Bioimaging Service is a high-sensitivity, spatially resolved elemental mapping solution designed for quantitative in situ analysis of biological tissues. This service integrates laser ablation (LA) sampling with time-of-flight inductively coupled plasma mass spectrometry (ICP-TOFMS), enabling simultaneous detection of up to 80+ elements per laser shot with sub-second temporal resolution. Unlike scanning quadrupole-based ICP-MS systems, the TOF detector captures full mass spectra from each ablation event—preserving stoichiometric relationships and minimizing spectral drift during extended imaging sequences. The technique is particularly suited for trace metal distribution studies in heterogeneous biological matrices where preservation of native elemental localization is critical—such as metallomics in neurodegenerative tissue, micronutrient transport in plant vasculature, or metalloprotein co-localization in tumor microenvironments.
Key Features
- Sub-20 µm lateral spatial resolution achieved via high-stability UV excimer laser (193 nm) with optimized spot size and scan synchronization
- Full-spectrum acquisition per ablation pulse (mass range m/z 5–280) supporting retrospective isotope ratio reprocessing without re-ablation
- Quantitative calibration using matrix-matched solid standards: NIST SRM 1486 (bone ash), GBW 07605 (spinach), and GBW 07607 (citrus leaves)
- Integrated signal normalization against 13C or 31P internal references to correct for ablation yield variability across tissue heterogeneity
- Robust vacuum-compatible sample stage accommodating cryo-sections, resin-embedded blocks, and flat-mounted fresh/dried specimens up to 9 cm × 9 cm × 0.8 cm
- Compliance-ready data structure including raw .tdms files, processed .imzML export, and metadata aligned with MSI (Mass Spectrometry Imaging) community standards
Sample Compatibility & Compliance
This service accepts formalin-fixed paraffin-embedded (FFPE) and frozen sections (OCT or LR White resin), provided paraffin is completely removed prior to analysis. Fresh or air-dried plant material must be mechanically stabilized on conductive substrates to prevent charging and thermal distortion during ablation. All sample preparation workflows follow GLP-aligned documentation protocols—including chain-of-custody logs, blank control runs, and daily instrument performance verification using multi-element tuning solutions. Data outputs support audit readiness for ISO/IEC 17025-accredited laboratories and are compatible with regulatory submission frameworks requiring traceable quantification (e.g., FDA Guidance for Elemental Impurities in Drug Products, ICH Q3D).
Software & Data Management
Data acquisition is controlled via proprietary Chemlab Pro LA-ICP-TOFMS Suite v4.x, supporting real-time spectral monitoring, automated focus optimization, and dynamic dwell-time adjustment per mass window. Post-acquisition processing leverages open-format imzML conversion tools compliant with SCiLS Lab and MSiReader ecosystems. Quantitative image reconstruction includes pixel-wise isotope ratio correction, background subtraction using adjacent blank regions, and spatial smoothing via Gaussian kernel convolution (σ = 1.5 pixels). Raw datasets and processed images are archived in encrypted AES-256 storage with immutable timestamps and user-access logs—meeting requirements for 21 CFR Part 11 electronic record integrity.
Applications
- Metallomic profiling of *Ophiocordyceps sinensis* insect body: Zn/Cu/Fe/Mn spatial gradients correlating with sclerotial development stages
- Subcellular metal redistribution in Alzheimer’s disease brain sections (e.g., Aβ plaque-associated Cu/Fe hotspots)
- Zinc and iron translocation dynamics in rice leaf cross-sections under Fe-deficient conditions
- Endogenous platinum distribution mapping in tumor xenograft sections following cisplatin administration
- Calcium and strontium co-localization in bone growth plate histology
FAQ
What sample preparation protocols are required for FFPE tissue sections?
Paraffin must be fully removed using xylene followed by ethanol dehydration series; no residual wax may remain, as it causes carbon deposition and signal suppression.
Can endogenous phosphorus or carbon be used for internal standardization?
Yes—31P is routinely applied for soft-tissue normalization; 13C is preferred for calcified or lipid-rich samples due to higher ion yield stability.
Is imzML export supported for third-party visualization tools?
Yes—all final images are delivered in imzML 1.3 format with embedded metadata, fully compatible with OpenMS, Cardinal, and SCiLS Lab.
How are detection limits determined for each element in a given tissue type?
Method detection limits (MDLs) are empirically established per project using 7-replicate blank measurements (matrix-matched tissue homogenate), calculated as 3.14 × standard deviation per isotope channel.
Do you provide statistical image analysis (e.g., cluster analysis, correlation mapping)?
Yes—advanced multivariate analysis (PCA, k-means clustering, Pearson/Spearman spatial correlation) is available as an optional add-on module with detailed methodology reporting.