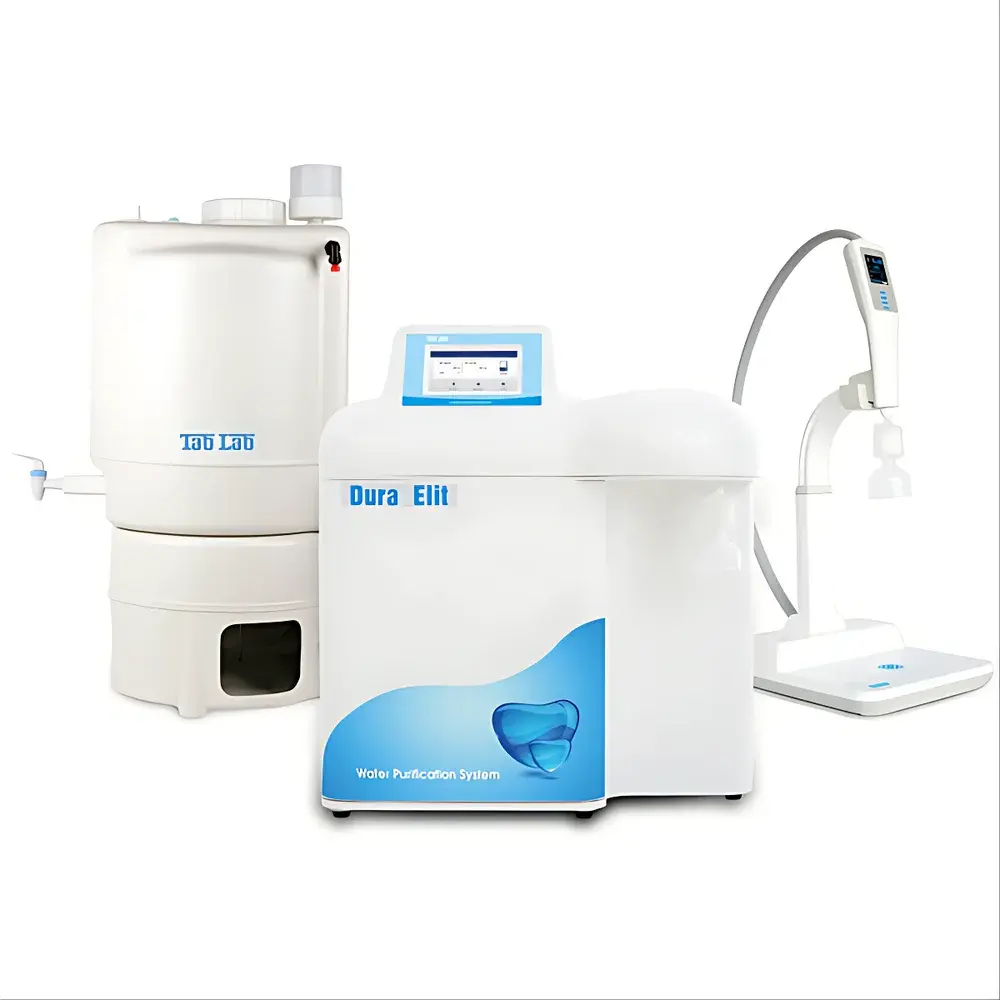
The Lab Dura Elit10 Full-Touchscreen Intelligent Ultra-Pure Water System
| Brand | The Lab |
|---|---|
| Origin | USA |
| Model | Dura Elit10 |
| Purified Water Grade | Type I (ASTM D1193-20, ISO 3696:1987, USP <1231>, EP 2.2.45) |
| Resistivity | 18.2 MΩ·cm @ 25 °C |
| Heavy Metals & Soluble Silica | <0.01 ppb |
| Total Organic Carbon (TOC) | <3 ppb |
| Microbial Count | <0.001 CFU/mL |
| Endotoxins | <0.001 EU/mL |
| Particles (>0.1 µm) | <1 particle/mL |
| Integrated TOC Monitor | 0.5–999.9 ppb, ±0.1 ppb accuracy |
| Daily Production Capacity | Up to 240 L of Type II water (≥5 MΩ·cm, TOC <30 ppb) and 10 L/h of Type I water |
Overview
The Lab Dura Elit10 is an integrated, full-touchscreen ultra-pure water system engineered for laboratories requiring continuous, on-demand production of ASTM Type I ultrapure water and ASTM Type II purified water from municipal tap feed. It employs a rigorously validated dual-stage reverse osmosis (RO) pretreatment train—featuring dual high-efficiency pumps, intermediate storage, and automated anti-fouling membrane flush cycles—followed by Ionpure® electrodeionization (EDI) and a final ultra-purification stage incorporating DuPont nuclear-grade mixed-bed ion exchange resin, dual-wavelength (254 nm / 185 nm) UV irradiation in 316L stainless steel chambers, 5,000 Da molecular weight cutoff (MWCO) ultrafiltration (UF), and dual-layer terminal filtration (0.45 + 0.1 µm PES membranes). This architecture ensures consistent delivery of water meeting or exceeding the most stringent international specifications—including USP , EP 2.2.45, ISO 3696:1987, ASTM D1193-20, CLSI GP32-A5, CAP guidelines, and GB/T 6682–2008 (Class I) and GB/T 33087–2016 (ultra-low TOC). The system is designed for GLP/GMP-compliant environments, with full audit trail capability, user-accessible calibration logs, and traceable operational history.
Key Features
- 5.0-inch full-color capacitive touchscreen interface with animated navigation menus, real-time system status visualization, and intuitive finger-driven operation
- Dual-stage RO system with optimized pressure balancing, intermediate buffer tank, and programmable automatic RO membrane flush (interval and duration configurable)
- Ionpure® EDI module enabling continuous, chemical-free generation of ≥5 MΩ·cm Type II water (TOC <30 ppb) without DI cartridge regeneration or softener salt addition
- Integrated MiniTOC online analyzer compliant with USP and EP 2.2.44 system suitability requirements; measurement range 0.5–999.9 ppb, accuracy ±0.1 ppb
- 50 L conical-bottom PE storage tank with CO₂-absorbing air filter, optional 254 nm UV sterilization module, and level-sensing feedback control
- R-DIS remote independent dispenser with 2.0-inch color graphical display, height-adjustable arm (up to 45 cm), detachable ergonomic handle, and 3 m cable reach (0.9 m handle extension)
- Three-point real-time resistivity monitoring (RO, DI, UP) and dual high-precision dispensing modes: volumetric (10–999,999 mL) and quality-triggered (0–18.25 MΩ·cm)
- Comprehensive consumables management: lifetime tracking and predictive replacement alerts for PP (5 µm), KDF, coconut-shell activated carbon, RO membranes, DI/UP cartridges, UV lamps, UF, and TF filters
- NSF-certified fluid path components (tubing, fittings, valves); low-TOC leaching polymer construction; fully enclosed engineering-grade plastic chassis resistant to corrosion and microbial adhesion
Sample Compatibility & Compliance
The Dura Elit10 delivers water suitable for highly sensitive analytical and biological applications where ionic, organic, particulate, microbial, and endotoxin contamination must be minimized. Its output meets the purity thresholds required for HPLC mobile phase preparation, ICP-MS calibration, cell culture media formulation, IVF procedures, ELISA assay buffers, and semiconductor wafer rinsing. All wetted materials comply with NSF/ANSI Standard 61 and FDA 21 CFR Part 11–ready data logging architecture supports electronic signatures and secure user authentication (dual-level password protection). System firmware maintains full traceability per GLP Annex 11 and GMP Annex 11 requirements, including time-stamped event logs, alarm history, consumables usage records, and USB-exportable datasets covering ≥12 months of continuous operation.
Software & Data Management
Embedded firmware provides centralized control over all operational parameters—including system clock (Y/M/D/H/M), scheduled standby (0–60 min), timed shutdown (0–24 h), and manual disinfection protocols (tank refill, loop circulation, point-of-use sanitization, drain activation). All water quality events—resistivity excursions, TOC deviations, microbial threshold breaches—are logged with timestamps and severity flags. Internal microSD card stores raw sensor outputs, dispensing logs, alarm histories, and maintenance records. Data export via USB port supports CSV-formatted reports compatible with LIMS integration and regulatory submission workflows. Audit trail integrity is preserved through immutable timestamping, user ID tagging, and write-protection of historical entries.
Applications
- Preparation of HPLC, UHPLC, GC, and IC mobile phases and standards
- Calibration and rinse solutions for ICP-OES, ICP-MS, AAS, and TOC analyzers
- Formulation of cell culture media, transfection reagents, and monoclonal antibody purification buffers
- Production of low-endotoxin water for in vitro fertilization (IVF), stem cell expansion, and primary neuronal cultures
- Final rinse water for photomask cleaning, microfluidic device fabrication, and nanomaterial synthesis
- Reference water for clinical chemistry analyzers, hematology systems, and molecular diagnostics platforms
FAQ
What standards does the Dura Elit10 meet for ultrapure water quality?
It complies with ASTM Type I and Type II specifications, USP , EP 2.2.45, ISO 3696:1987 Class 1, CLSI GP32-A5, CAP laboratory accreditation criteria, and Chinese national standards GB/T 6682–2008 and GB/T 33087–2016.
How is TOC continuously monitored and verified?
An integrated MiniTOC analyzer performs real-time, non-destructive UV-persulfate oxidation with photometric detection; results are traceable to NIST-traceable standards and satisfy USP system suitability tests.
Can the system operate under GLP or GMP conditions?
Yes—its firmware supports 21 CFR Part 11–compliant electronic records, dual-password access control, immutable audit trails, and exportable validation-ready datasets.
What is the expected service life of the RO membrane and EDI stack?
With proper pretreatment and scheduled anti-fouling flushes, RO membranes typically exceed 3 years; Ionpure® EDI modules demonstrate >5 years of stable operation under typical lab load profiles.
Is remote monitoring or network integration supported?
While the base configuration uses local USB and touchscreen interfaces, optional Ethernet or RS-485 communication modules enable integration into building management systems (BMS) or centralized lab infrastructure dashboards.