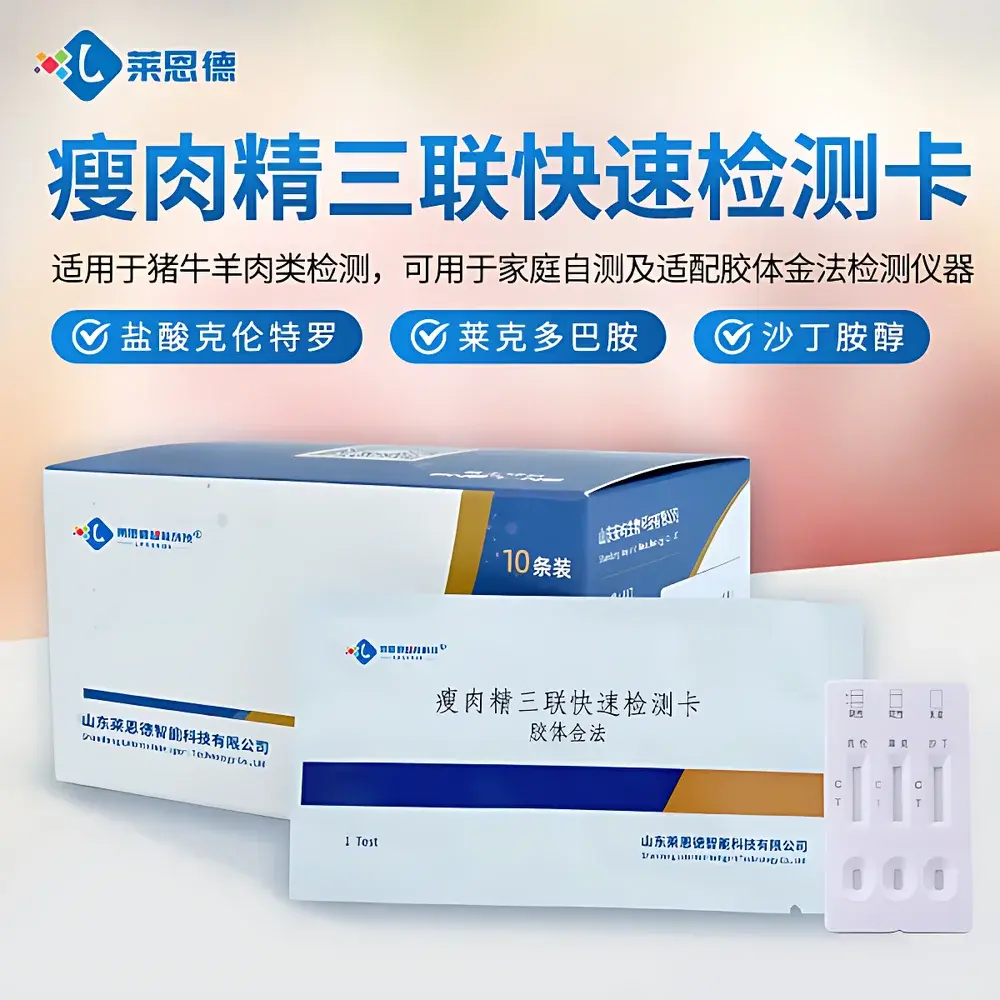
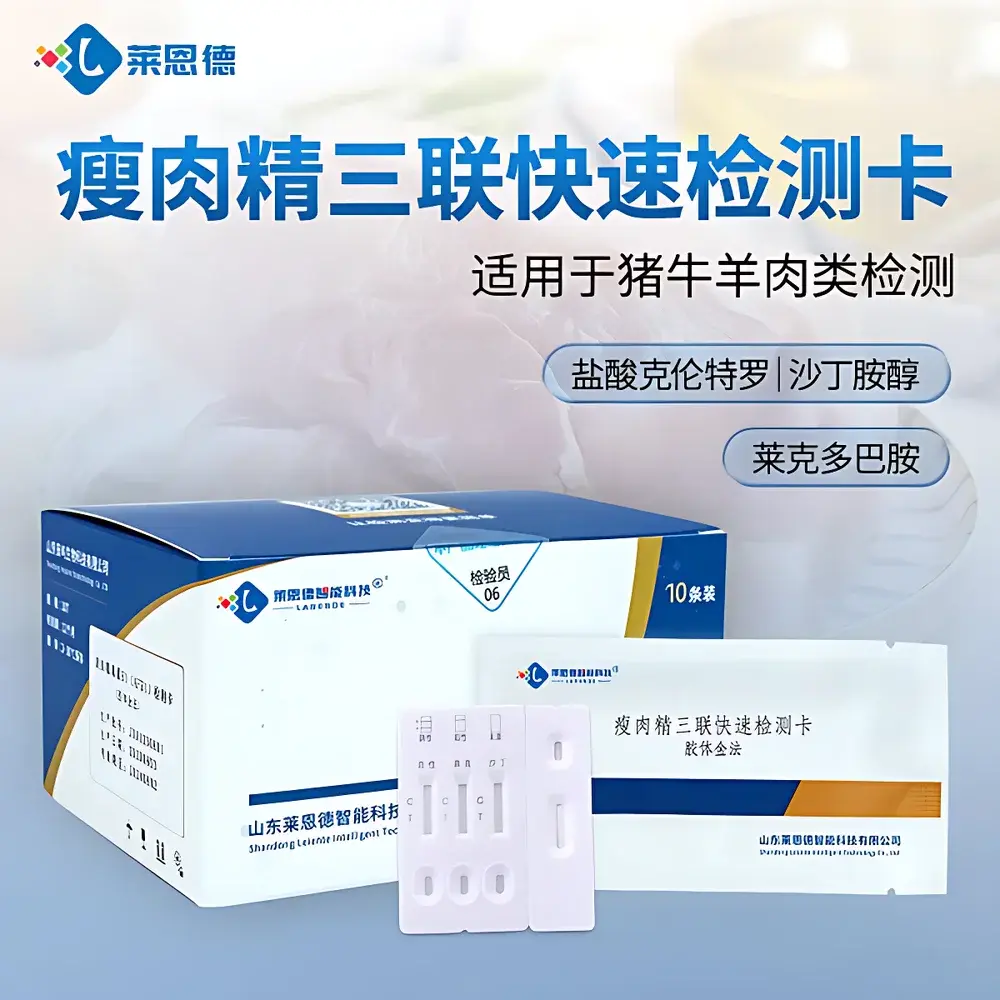
LANENDE LD Triple-Target Rapid Immunoassay Test Strip for Clenbuterol, Ractopamine, and Salbutamol Residues
| Brand | LANENDE Intelligent Technology |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | OEM/ODM Producer |
| Origin Category | Domestic (China) |
| Model | LD |
| Price | USD 28.50 (FOB) |
| Assay Time | ≤5 min per sample |
| Format | Lateral Flow Immunoassay (LFIA) with Colloidal Gold Conjugate |
Overview
The LANENDE LD Triple-Target Rapid Immunoassay Test Strip is a qualitative, on-site screening tool engineered for the simultaneous detection of three β-agonist veterinary drug residues—clenbuterol hydrochloride, ractopamine, and salbutamol—in animal-derived matrices. Designed in accordance with international immunoassay principles, it operates on a competitive inhibition lateral flow immunoassay (LFIA) platform using colloidal gold-labeled monoclonal antibodies. Each test strip integrates three independent assay zones within a single nitrocellulose membrane, enabling parallel visual interpretation without cross-reactivity interference. The device delivers objective, binary (positive/negative) results within 3–5 minutes post-sample application, supporting high-throughput preliminary screening in feed mills, slaughterhouses, regulatory inspection labs, and field-based food safety units. It is not intended for quantitative analysis or regulatory confirmation; positive results require follow-up by LC-MS/MS or GC-MS per ISO 17025-accredited protocols.
Key Features
- Triple-target specificity: Simultaneous detection of clenbuterol, ractopamine, and salbutamol in a single test—no need for separate assays or reagent kits.
- Colloidal gold conjugation: High-affinity monoclonal antibodies conjugated to 40 nm colloidal gold nanoparticles ensure consistent binding kinetics and low background noise.
- Integrated internal control: A built-in C-line (control line) appears in all valid tests, confirming proper capillary flow and reagent integrity.
- No instrumentation required: Visual readout under ambient lighting; no reader, software, or power source needed.
- Robust shelf life: Stable at 2–30 °C for 12 months unopened; desiccant-sealed aluminum pouch prevents humidity-induced antibody degradation.
- Minimal equipment dependency: Compatible with standard lab tools—vortex mixer, centrifuge (4000 × g), analytical balance (0.01 g resolution), and calibrated pipettes.
Sample Compatibility & Compliance
The LD test strip is validated for use with bovine/pork ovine urine, skeletal muscle (fat-free), liver homogenates, and compound feed samples. Sample preparation follows harmonized extraction protocols aligned with GB/T 22286–2008 (Chinese national standard for β-agonists in animal tissues) and AOAC Official Method 2011.05 (for ractopamine in feed). While not certified to ISO/IEC 17025 or FDA 21 CFR Part 11, the assay supports GLP-aligned workflows when used as a tier-1 screening step prior to confirmatory testing. Cross-reactivity studies confirm <5% interference from structurally related β-agonists (e.g., terbutaline, fenoterol), ensuring analytical specificity per IUPAC guidelines.
Software & Data Management
This is a non-instrumented, analog diagnostic format. No embedded firmware, Bluetooth, or digital output is present. Result documentation must be performed manually via lab notebook entries or LIMS-integrated image capture (using standardized lighting and scale reference). For audit readiness, users are advised to record batch number, expiration date, operator ID, sample ID, and result interpretation timestamp. Digital archiving of photographed test strips complies with basic traceability requirements under China’s Food Safety Law (Article 42) and EU Regulation (EC) No 854/2004 for official controls.
Applications
- Pre-slaughter screening at abattoirs to identify suspect animals prior to processing.
- Routine surveillance in feed manufacturing facilities to verify raw material compliance.
- Field deployment by municipal food safety inspectors during market inspections.
- QC/QA verification in meat processing plants prior to product release.
- Educational use in food science laboratories for immunoassay methodology training.
FAQ
What is the limit of detection (LOD) for each analyte?
The LOD varies by matrix: 0.5 µg/L in urine, 0.2 µg/kg in muscle tissue, and 1.0 µg/kg in feed—per validation data submitted to CNAS-accredited third-party labs.
Can this test be used for milk or blood samples?
No. The assay has not been validated for milk, serum, or plasma. Matrix effects from casein or hemoglobin may cause false negatives or invalid C-lines.
Is centrifugation mandatory for all sample types?
Yes—for turbid urine, tissue extracts, and feed supernatants. Clarification ensures uniform flow and prevents membrane clogging or non-specific binding.
What does an invalid result indicate?
Absence of the C-line indicates either insufficient sample volume, damaged membrane, expired reagents, or improper storage (e.g., exposure to >30 °C or humidity >60% RH).
Does the test comply with EU MRLs?
It aligns with EU Commission Regulation (EU) No 37/2010 MRLs for clenbuterol (0.01 µg/kg in muscle) and ractopamine (10 µg/kg in liver), but serves only as a pass/fail screen—not a compliance certificate.