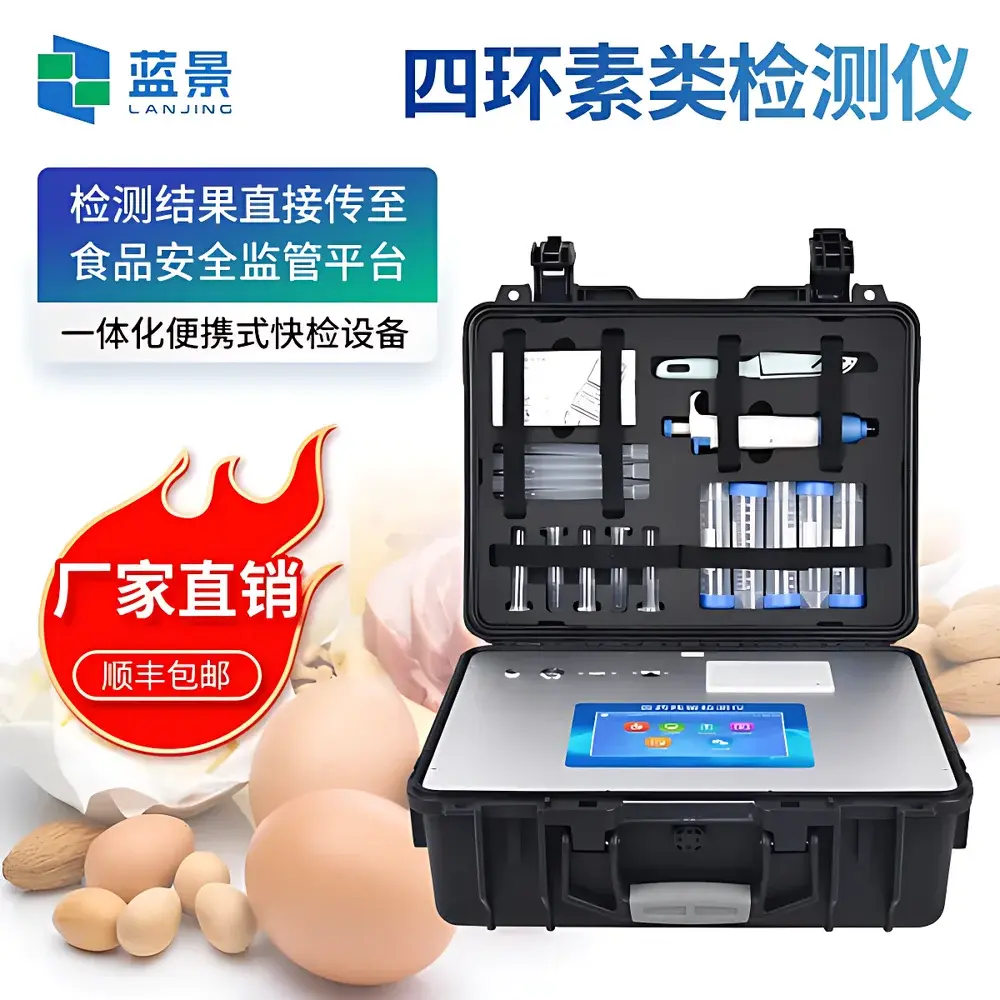
Lanjing LJ-KSS Veterinary Drug Residue Rapid Tester for Antibiotics (Nitrofuran Metabolites, Tetracyclines, and Broad-Spectrum Antibiotics)
| Brand | Lanjing |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | OEM/ODM Producer |
| Country of Origin | China |
| Model | LJ-KSS |
| Price | USD 820 (FOB Qingdao) |
Overview
The Lanjing LJ-KSS Veterinary Drug Residue Rapid Tester is a field-deployable, multi-analyte immunoassay platform engineered for quantitative and semi-quantitative detection of veterinary drug residues in food matrices—particularly meat, dairy, aquaculture products, and animal-derived raw materials. It operates on the principle of colloidal gold immunochromatographic assay (GICA), leveraging antigen–antibody binding kinetics and high-sensitivity optical scanning to deliver rapid, objective, and trace-level residue assessment. Designed for compliance with national food safety monitoring frameworks—including China’s GB 31650–2019 (Maximum Residue Limits for Veterinary Drugs in Foods) and aligned with international reference methodologies (e.g., AOAC Official Method 2014.01 for nitrofuran metabolites), the system supports regulatory screening at point-of-collection without requiring centralized laboratory infrastructure.
Key Features
- Integrated triple-module architecture: simultaneous operation of food safety residue detection, multi-channel pesticide residue analysis, and colloidal gold immunochromatography—controlled via unified Android-based firmware.
- Industrial-grade ABS housing with shock-absorbing design, IP54-rated enclosure, and ergonomic carry handle—validated for mobile deployment across slaughterhouses, cold-chain logistics hubs, border inspection posts, and rural veterinary clinics.
- ARM Cortex-A7 RK3288 quad-core processor (1.88 GHz), 2 GB RAM, and 16 GB internal storage ensure deterministic response during concurrent multi-channel scanning and data upload.
- 7-inch capacitive touchscreen with localized English UI; supports on-device sample annotation, customizable test protocols, and real-time pass/fail threshold adjustment per analyte.
- Auto-calibrating optical sensor with dynamic LED brightness control and thermal drift compensation—maintains photometric stability over 8+ hours of continuous operation.
- Dual-power capability: 12 V DC input (compatible with vehicle power systems) + optional 6 Ah Li-ion battery pack enabling ≥6 h field autonomy.
- Embedded regulatory database: preloaded with Chinese MRLs (GB 31650–2019), EU Commission Regulation (EU) No 37/2010, and FDA CVM guidance thresholds—updatable via secure OTA firmware patches.
- Regulatory-grade data governance: full audit trail (user login, timestamped result generation, calibration logs), 200,000-result local storage, USB export to Excel (.xlsx), and encrypted cloud sync via HTTPS/Wi-Fi/4G LTE.
Sample Compatibility & Compliance
The LJ-KSS accepts lateral flow immunochromatographic test strips from third-party manufacturers certified to ISO 13485 and CE-IVD standards—including those targeting furazolidone metabolites (AOZ), tetracyclines (TC, OTC, CTC, DC), chloramphenicol, enrofloxacin, sulfonamides, and nitrofuran derivatives (AMOZ, SEM, AHD, MYA). Sample types include muscle tissue homogenates, milk supernatants, egg yolk extracts, and aquaculture pond water filtrates—processed using standardized extraction kits compliant with GB/T 22338–2008 and SN/T 1751.2–2007. Instrument output meets GLP documentation requirements and supports 21 CFR Part 11–compliant electronic signatures when integrated with validated LIMS platforms.
Software & Data Management
The embedded Android OS (v8.1) hosts a purpose-built application supporting batch testing, auto-generated QR-coded reports, and configurable result templates. All measurements are time-stamped, geotagged (via connected GPS module), and assigned unique sample IDs with auto-increment logic. Data export options include direct USB mass-storage mode, Wi-Fi SMB file sharing, MQTT-based telemetry to centralized dashboards (e.g., provincial food safety surveillance networks), and RESTful API integration for enterprise-level traceability systems. Firmware updates are digitally signed and verified; configuration backups support factory reset recovery without loss of calibration history or user-defined MRL libraries.
Applications
Field laboratories of municipal market supervision bureaus perform routine surveillance of pork, beef, and poultry at wholesale markets. Veterinary health authorities deploy units during outbreak investigations involving nitrofuran misuse in aquaculture farms. Dairy cooperatives screen raw milk at collection centers for tetracycline carryover from dry-cow therapy. Import inspection agencies verify compliance of imported seafood shipments against destination-country MRLs prior to customs clearance. The system also serves as a primary triage tool in GMP-certified feed mills verifying absence of cross-contamination from antibiotic-laden premixes.
FAQ
Does the LJ-KSS require consumables other than commercial colloidal gold test strips?
No—the instrument is strip-agnostic and compatible with any CE-marked or NMPA-registered GICA strip meeting standard dimensions (e.g., 6 mm × 80 mm) and T/C line spacing specifications.
Can detection thresholds be adjusted to match non-Chinese regulatory limits?
Yes—users may import custom MRL tables in CSV format and assign them to specific analytes; the software applies linear interpolation for interpolated cutoff values.
Is remote diagnostic support available for firmware or hardware troubleshooting?
Yes—enabled via secure SSH tunneling and remote desktop (TeamViewer QuickSupport embedded); log files include sensor diagnostics, thermal profiles, and flash memory wear metrics.
What validation documentation accompanies the device for regulatory audits?
Each unit ships with a Factory Calibration Certificate (traceable to NIM SI standards), IQ/OQ protocol templates, and a Declaration of Conformity referencing GB/T 19001–2016 (ISO 9001) and YY/T 0287–2017 (ISO 13485).
How frequently must optical calibration be performed?
Automatic daily zero-point calibration occurs at power-on; manual full-range calibration is recommended every 30 days or after 500 scans—guided by on-screen prompts and logged in the audit trail.