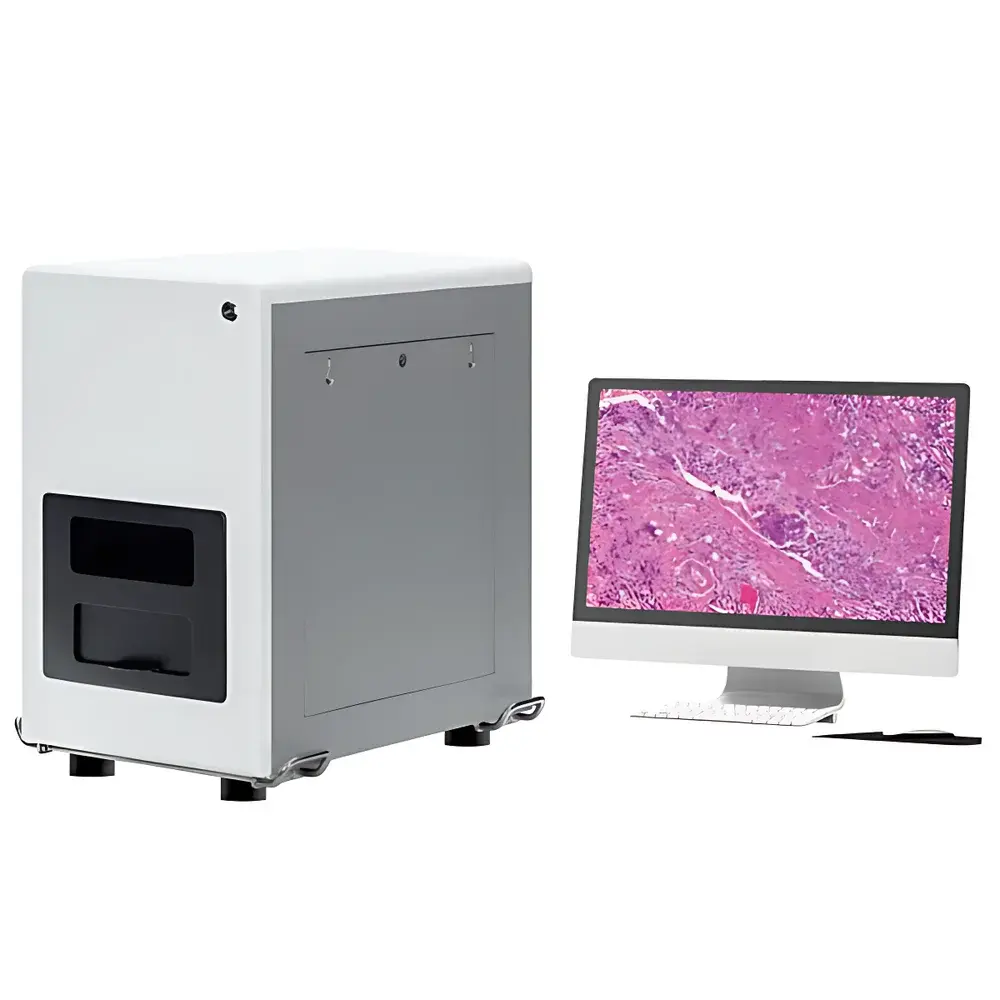
LEI-TECH LK-XWSMY Whole-Slide Digital Pathology Scanner
| Brand | LEI-TECH |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Country of Origin | China |
| Model | LK-XWSMY |
| Price | USD 65,000 (FOB Tianjin) |
Overview
The LEI-TECH LK-XWSMY Whole-Slide Digital Pathology Scanner is a high-precision, dual-mode (brightfield and fluorescence) automated microscopy imaging platform engineered for reproducible, high-fidelity digitization of histological, cytological, microbiological, and hematological specimens. Built upon a robust Köhler-illuminated optical architecture with motorized objective turrets, precision XYZ stage control, and real-time autofocus algorithms, the system captures gigapixel-resolution digital slides compliant with international digital pathology standards—including DICOM-SR, Aperio SVS, and OpenSlide-compatible formats. Its core measurement principle relies on sequential high-magnification tile-based image acquisition, followed by sub-pixel registration and seamless mosaic stitching—enabling quantitative morphometric analysis, remote collaborative diagnosis, and longitudinal archival in GLP/GMP-aligned laboratories.
Key Features
- Dual imaging modality: Integrated brightfield and optional fluorescence channels (B/G/UV LED excitation with motorized filter cube turret and optional auto-oiling for 100× oil immersion)
- Modular slide handling: Standard 5-slide cassette (75–76.5 × 25–26.5 × 0.9–1.2 mm), plus optional 2-slide and 1-slide cassettes accommodating custom-format glass slides (e.g., 100 × 75 mm)
- Automated barcode/QR code recognition at load-in, enabling traceable specimen tracking and LIMS integration
- 3-position motorized objective revolver supporting 10×, 20×, 40×, 60×, and 100× objectives (NA up to 1.35); oil-immersion mode fully automated with maintenance-free oil dispenser (rated for >5 million cycles)
- High-sensitivity imaging sensor: 7 MP color CMOS (1.1″ format, 4.5 µm pixel pitch); optional monochrome variant available for enhanced SNR in low-light fluorescence applications
- Real-time terrain-based autofocus and Z-stack acquisition capability for thick-section or multi-layered specimens
- Scan speed: ≤60 seconds per 15 mm × 15 mm field at 20× magnification (brightfield); optimized for throughput without compromising resolution uniformity across full slide area
Sample Compatibility & Compliance
The LK-XWSMY accommodates unstained and conventionally stained (H&E, Gram, Ziehl–Neelsen, Giemsa, Papanicolaou), immunostained (IHC), and multiplex fluorescent (FISH, IF) specimens—both with and without standard coverslips. It supports tissue sections (3–10 µm), cytology smears, blood films, microbial preparations, and fine-needle aspirates. The system complies with ISO 13485:2016 (medical device quality management), meets essential requirements under IEC 61010-1 for laboratory equipment safety, and generates audit-trail-enabled metadata required for FDA 21 CFR Part 11–compliant environments when paired with validated software configurations. All digital slide outputs adhere to CAP and EMA digital pathology validation guidelines for diagnostic use.
Software & Data Management
The bundled LEI-SCAN Suite provides end-to-end workflow automation: auto-focus mapping, region-of-interest (ROI) selection via panoramic preview, batch scanning with pause/resume, multi-channel fluorescence fusion, and lossless TIFF/BMP/JPEG export. Local or network-attached storage deployment is supported; DICOM-SR export enables PACS integration. Annotation tools include ROI markup, linear/angular measurement, cell counting overlays, and customizable reporting templates. Software logs all user actions, parameter changes, and calibration events with timestamped, immutable records—fully satisfying GLP audit requirements. Remote access is enabled via secure HTTPS tunneling; no cloud-hosted data processing occurs unless explicitly configured by the institution.
Applications
- Microbiology: Digitization of Gram-stained smears (e.g., E. coli identification), acid-fast bacilli detection (Ziehl–Neelsen), and auramine-O fluorescence imaging—enabling AI-assisted pattern recognition and cross-site reference comparison
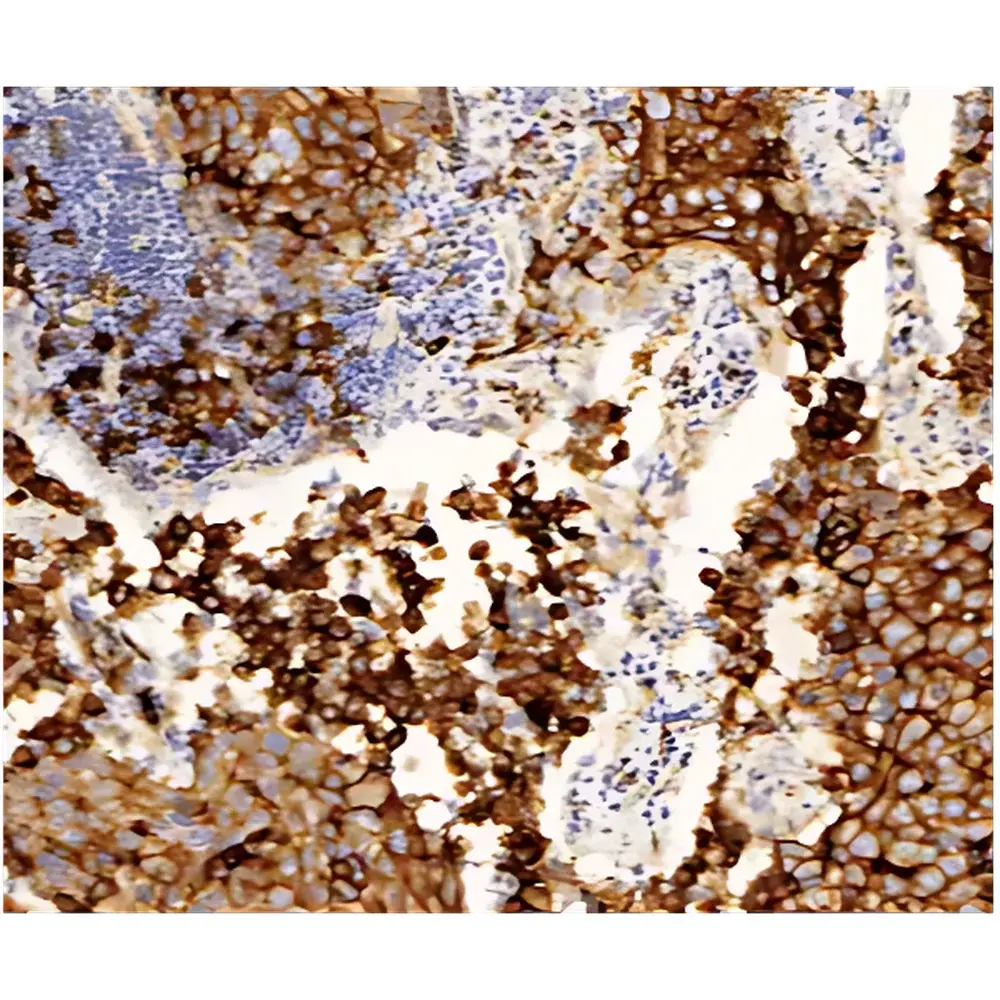
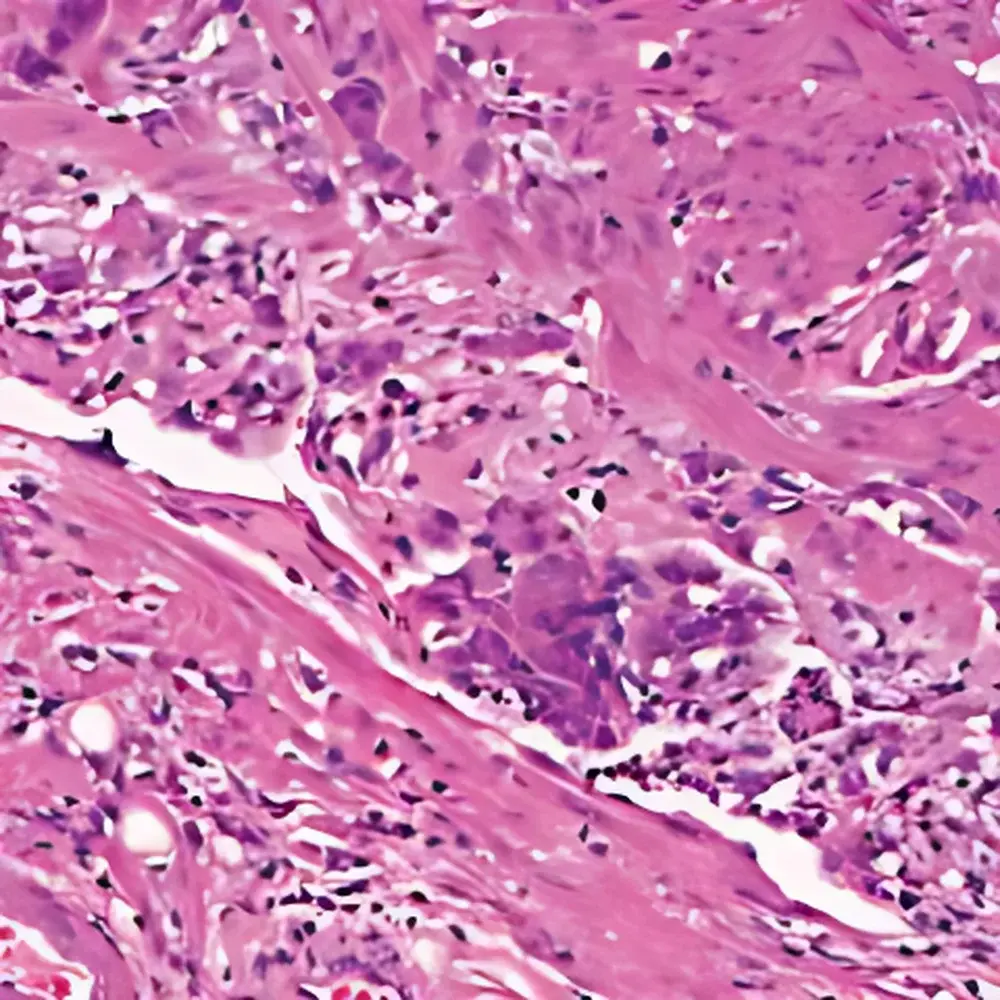
- Anatomic & Clinical Pathology: High-fidelity scanning of H&E, trichrome, and special stains for primary diagnosis, second-opinion consultation, and tumor board review; optimized for nuclear detail resolution at 40× and 100× oil
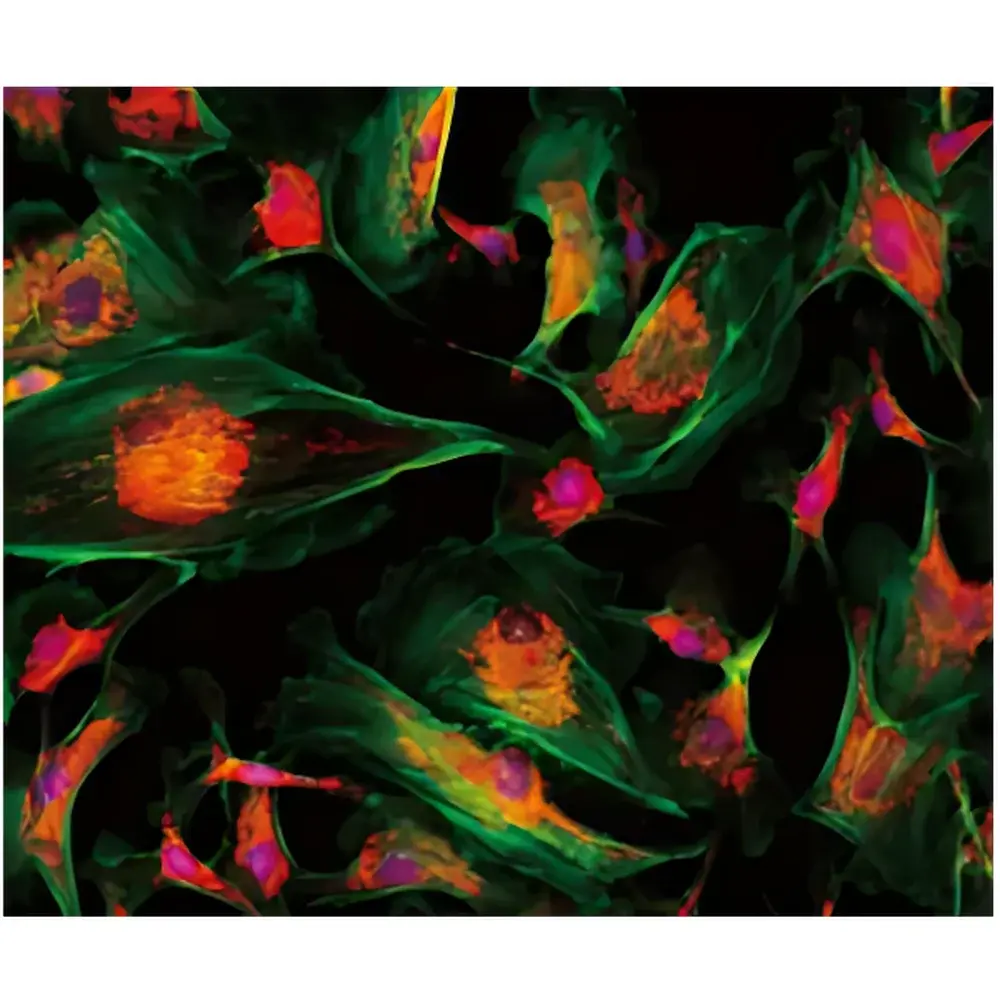
- FISH & Multiplex IF: Simultaneous acquisition of ≥3 fluorophores with spectral unmixing support; registered overlay of DNA probes and protein markers on single digital slides for biomarker co-localization studies
- Hematology: Standardized digitization of peripheral blood smears and bone marrow aspirates, facilitating morphology review, differential counts, and educational archiving
- Research & Education: Integration with open-source analysis frameworks (QuPath, HALO, ImageJ) via OME-TIFF export; scalable for biobank-scale slide digitization projects
FAQ
Does the LK-XWSMY support FDA-cleared diagnostic use?
Yes—when deployed with validated software configuration, documented IQ/OQ/PQ protocols, and integrated into a 21 CFR Part 11–compliant IT infrastructure, the system meets regulatory expectations for primary digital diagnosis in clinical pathology labs.
Can the scanner process non-standard slide sizes?
Yes—via optional cassettes accommodating 100 × 75 mm and 75 × 51 mm slides; custom cassette engineering is available under OEM agreement.
What is the optical resolution limit at 100× oil immersion?
0.067 µm/pixel (theoretical Nyquist-limited resolution ~0.13 µm), consistent with diffraction-limited performance of UPLXAPO 100×/1.35 NA optics.
Is remote technical support available 24/7?
Yes—LEI-TECH operates global service hubs with 7×24 remote diagnostics, on-site engineer dispatch within 48 hours (major markets), and two-year comprehensive warranty covering parts, labor, and software updates.
How is data security managed during networked operation?
All communications use TLS 1.2+ encryption; local storage remains on-premise unless explicitly routed through institutional-approved cloud gateways; no telemetry or automatic data upload occurs without administrator consent.