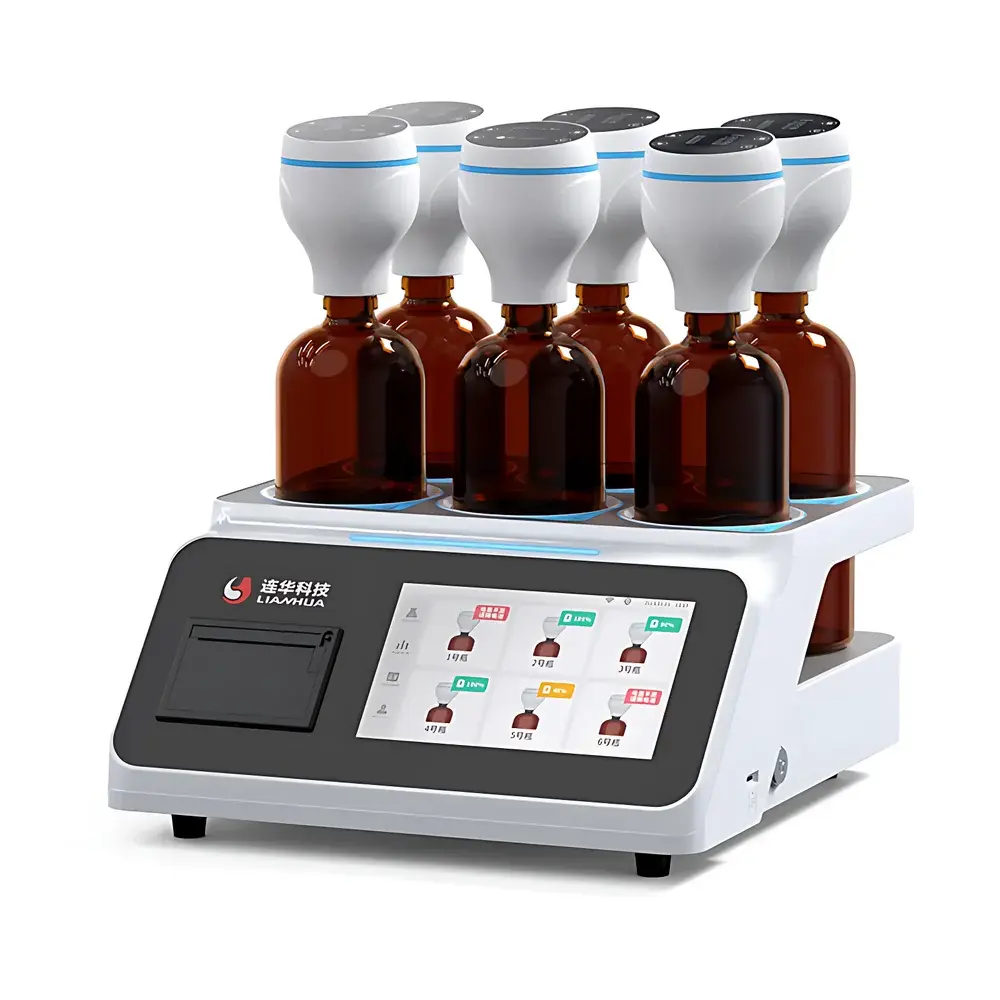
Lianhua LH-BOD606 BOD5 Analyzer
| Brand | Lianhua |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Regional Classification | Domestic (China) |
| Model | LH-BOD606 |
| Instrument Type | Laboratory BOD Analyzer |
| Measurement Principle | Manometric (Pressure-Difference) Method |
| Measurement Range | 0–4000 mg/L |
| Detection Limit | 0.1 mg/L |
| Accuracy | ±0.1 mg/L for values <10 mg/L |
| Incubation Duration | Selectable from 1 to 30 days |
| Repeatability | ≤±10% RSD |
Overview
The Lianhua LH-BOD606 BOD5 Analyzer is a fully automated, manometric (pressure-difference) based laboratory instrument engineered for precise and compliant determination of five-day biochemical oxygen demand (BOD5) in water and wastewater samples. It operates on the fundamental principle of measuring the pressure differential generated by microbial oxygen consumption within a sealed sample chamber—eliminating mercury-based manometers and associated environmental and safety hazards. Designed in strict accordance with HJ 505–2009 (“Determination of Five-Day Biochemical Oxygen Demand (BOD5) in Water Quality — Dilution and Inoculation Method”) and aligned with ISO 9408:1999 (“Water Quality — Determination of Biochemical Oxygen Demand after N Days — BODN”), the LH-BOD606 delivers traceable, auditable, and method-validated results suitable for regulatory reporting, environmental compliance monitoring, and wastewater treatment process control.
Key Features
- Modular, wireless test-cap architecture: Each test cap functions as an autonomous micro-analyzer—equipped with a 1.5-inch OLED display, internal rechargeable battery (supporting uninterrupted operation during brief power outages), onboard data logging (100 entries per cap, auto-overwrite), and independent parameter configuration (incubation duration: 1–30 days; thermal equilibration delay: 1–10 h).
- Zigbee 2.4 GHz bidirectional mesh networking: Enables real-time, low-latency synchronization between up to six test caps and the central host unit—ensuring consistent command execution, status updates, and fault detection without communication lag or packet loss.
- 7-inch capacitive touchscreen host interface: Provides live visualization of cap battery levels, incubation progress, BOD concentration trends, sample naming, and time-series curve overlays—optimized for multi-user lab environments.
- Integrated Android-derived LHOS operating system: Delivers intuitive UI navigation, responsive touch interaction, and robust firmware extensibility—including over-the-air (OTA) updates, custom assay protocol deployment, and audit-trail-enabling system logging.
- Zero-mercury, pressure-difference transduction: Utilizes high-stability piezoresistive sensors calibrated against NIST-traceable references—ensuring long-term drift stability and eliminating hazardous waste disposal requirements.
- Full experimental workflow support: Includes pre-qualified reagents (nutrient buffer, nitrification inhibitor, inoculum), consumables (sealing gaskets, fermentation bottles), and embedded digital tutorials compliant with GLP training documentation standards.
Sample Compatibility & Compliance
The LH-BOD606 accommodates standard 60 mL or 250 mL BOD bottles (ASTM D5210, ISO 5815-1) and supports undiluted or serially diluted samples across the full 0–4000 mg/L range. Its pressure-compensated algorithm automatically corrects for barometric fluctuations and temperature-induced gas expansion, satisfying HJ 505–2009’s requirement for atmospheric pressure compensation. The system meets ISO/IEC 17025:2017 general requirements for competence of testing laboratories and supports 21 CFR Part 11-compliant electronic records when paired with validated LHOS firmware versions (audit trail, user authentication, electronic signatures). All measurement outputs are directly traceable to SI units (mg O2/L) without manual conversion factors.
Software & Data Management
Data handling is implemented via a three-tier architecture: local cap storage → synchronized host database → cloud-accessible mobile dashboard (via official Lianhua小程序). The host system enables temporal, keyword-, and sample ID–based filtering; side-by-side comparison of historical BOD curves; statistical summary (mean, SD, CV%); and export to CSV, PDF, or XML formats compatible with LIMS integration. Audit logs record all user actions—including method changes, calibration events, and result modifications—with immutable timestamps and operator IDs. Firmware-level encryption (AES-128) secures stored data at rest and in transit.
Applications
- Regulatory compliance testing for municipal and industrial wastewater discharge permits (e.g., EPA NPDES, EU Urban Wastewater Treatment Directive)
- Influent/effluent monitoring in activated sludge, MBR, and oxidation ditch treatment plants
- Environmental impact assessments (EIAs) and baseline water quality characterization
- Research on biodegradability kinetics of emerging contaminants (pharmaceuticals, microplastics, PFAS)
- Quality assurance in drinking water source protection programs
- Educational laboratories requiring GLP-aligned, low-maintenance BOD instruction platforms
FAQ
Does the LH-BOD606 require daily manual intervention during incubation?
No—once configured and sealed, each test cap operates autonomously. The host provides remote status monitoring, but no manual sampling, titration, or sensor recalibration is required.
Can the system validate BOD5 results against reference materials?
Yes—the instrument supports certified reference materials (CRMs) such as potassium hydrogen phthalate (KHP) solutions. Built-in QC protocols calculate recovery rates and flag deviations exceeding ±15% per HJ 505–2009 Annex C.
Is method validation documentation available for regulatory submissions?
Lianhua provides a comprehensive IQ/OQ/PQ protocol package, including installation qualification checklists, operational verification reports, and performance verification data against ISO 9408:1999 interlaboratory study benchmarks.
How is data integrity ensured under GLP/GMP conditions?
LHOS firmware implements role-based access control, electronic signature capture for critical actions, and tamper-evident audit trails compliant with OECD Principles of GLP and FDA 21 CFR Part 11 Subpart B.
What maintenance is required for long-term reliability?
Annual sensor calibration using certified pressure standards and quarterly cleaning of cap sealing interfaces are recommended. No routine fluid replacement or mercury handling is involved.