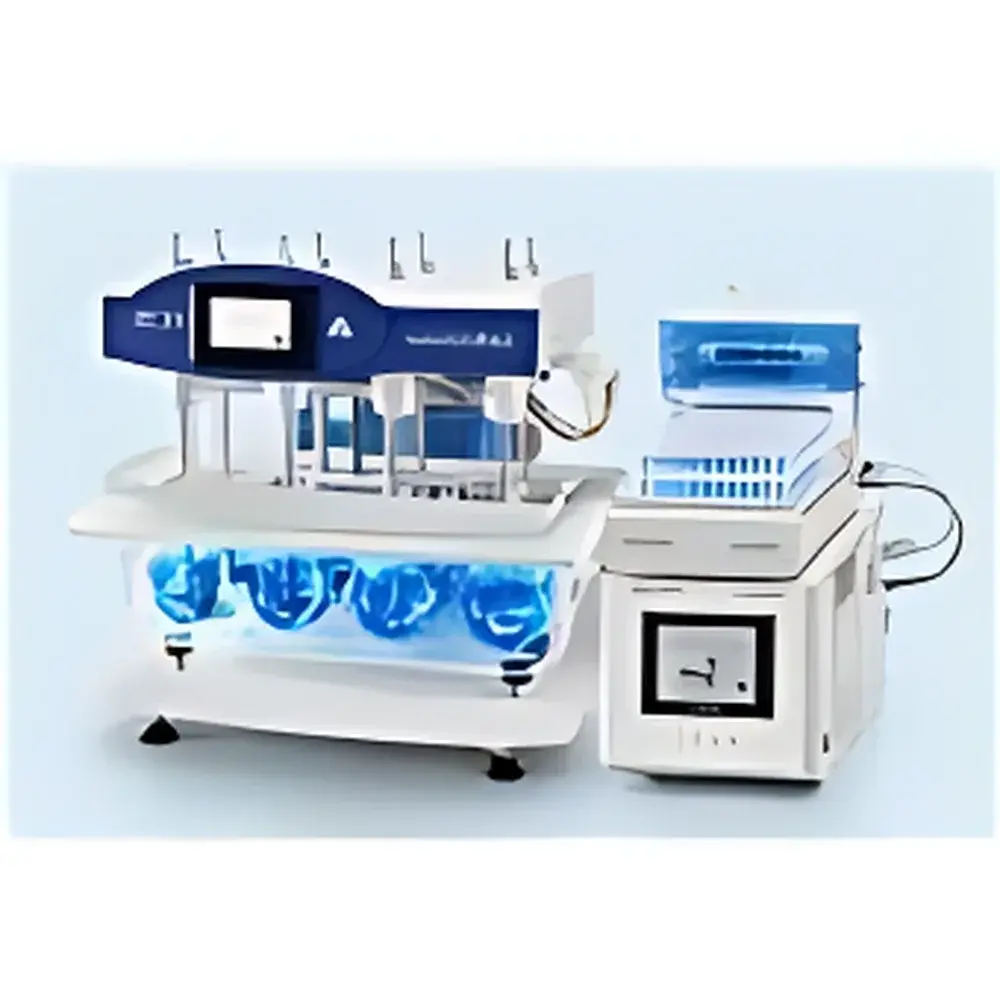
LOGAN SYSTEM 850DLA 8-Position Automated Dissolution Sampling System
| Brand | LOGAN |
|---|---|
| Origin | USA |
| Model | SYSTEM 850DLA |
| Instrument Type | Online Dissolution System |
| Number of Vessels | 8 |
| Temperature Range | 20–60 °C |
| Temperature Accuracy | ±0.1 °C |
| Rotation Speed Range | 25–250 rpm (±1 rpm) |
| Paddle Wobble Amplitude | ≤0.5 mm |
| Basket Wobble Amplitude | ≤1.0 mm |
| Water Bath Temperature Range | Ambient to 45 °C |
| Water Bath Temperature Resolution | 0.1 °C |
| Water Bath Volume | 18 L |
Overview
The LOGAN SYSTEM 850DLA is an online, fully automated 8-position dissolution sampling system engineered for regulatory-compliant pharmaceutical development and quality control. It operates on the principles of USP Apparatus 1 (basket), Apparatus 2 (paddle), and extended methods including mini-vessel, flow-through cell, paddle-over-disc, and rotating cylinder configurations. Designed in strict accordance with FDA guidance for dissolution method development and bioequivalence (BE) studies, the SYSTEM 850DLA integrates precise mechanical actuation, thermally stable fluid handling, and real-time process monitoring to deliver high reproducibility across multiple dissolution vessels simultaneously. Its modular architecture—comprising the UDT-818A dissolution station, SCR-DL sample collector, SYP-8L-10 mL syringe pump, and DSC-800 central controller—enables scalable deployment in both R&D laboratories and GMP-regulated QC environments.
Key Features
- Modular, field-upgradable platform supporting USP Apparatus 1, 2, 3, 4, 5, and 6 via interchangeable hardware kits
- Independent temperature monitoring with eight embedded Pt100 probes—one per vessel—ensuring vessel-specific thermal validation and compliance with USP & Ph. Eur. requirements
- Dual-stage filtration (pre- and post-sampling) integrated into the sampling path to minimize particulate carryover and protect downstream analytical instrumentation (e.g., HPLC, UV-Vis)
- High-precision 10 mL syringe pump (SYP-8L) delivering volumetric accuracy ≤±0.5% CV at standard sampling volumes, eliminating operator-induced variability
- Hermetic dual-lid vessel sealing system reducing solvent evaporation to <0.5 mL/h per vessel under continuous 60 °C operation
- Integrated LED illumination module enabling visual assessment of tablet disintegration and particle dispersion kinetics during dissolution
- Automated media replenishment and temperature-matched replacement fluid delivery to maintain sink conditions without manual intervention
- FDA 21 CFR Part 11–compliant audit trail with electronic signatures, time-stamped event logging, and role-based three-tier access control (Administrator, Supervisor, Analyst)
Sample Compatibility & Compliance
The SYSTEM 850DLA accommodates solid oral dosage forms—including immediate-release and modified-release tablets, capsules, suspensions, transdermal patches, and hydrogels—across all major pharmacopeial dissolution methods. Each vessel supports standardized 1000 mL or mini-vessel (100–250 mL) configurations. The system meets ISO/IEC 17025 calibration traceability requirements when paired with NIST-traceable temperature and rotational verification tools. All firmware and software modules are validated per ICH Q2(R2) and aligned with GMP Annex 11 for computerized system validation (CSV). Full documentation packages—including IQ/OQ/PQ protocols, risk assessments (FMEA), and change control records—are available upon request to support regulatory submissions (IND, ANDA, MAA).
Software & Data Management
Controlled by the DSC-800 system software, the SYSTEM 850DLA provides a secure, Windows-based interface with configurable run templates, dynamic method sequencing, and real-time graphing of temperature, rotation speed, and sampling events. Data export is native to CSV, PDF, and XML formats compatible with LIMS integration. The embedded database enforces immutable storage of raw acquisition data, metadata, and user actions. Remote monitoring capability—accessible via authenticated HTTPS connection—allows supervisory review of active runs through a dedicated mobile application (iOS/Android), with push notifications for critical deviations (e.g., temperature excursion >±0.3 °C, pump stall, lid open event). All electronic records comply with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available).
Applications
- Comparative dissolution profiling for generic product development and Quality-by-Design (QbD) initiatives
- In vitro–in vivo correlation (IVIVC) modeling using multi-timepoint sampling and kinetic deconvolution
- Biopharmaceutics Classification System (BCS) and Biopharmaceutics Drug Disposition Classification System (BDDCS) characterization
- Dissolution-permeation coupling studies via integration with parallel artificial membrane permeability assay (PAMPA) or Caco-2 systems
- Stability-indicating dissolution testing under stressed conditions (pH, surfactant, enzymatic media)
- Method transfer between development labs and commercial manufacturing sites
- Support for FDA’s Draft Guidance on Dissolution Testing of Immediate-Release Solid Oral Dosage Forms (2023)
FAQ
Does the SYSTEM 850DLA support USP Apparatus 4 (flow-through cell)?
Yes—when configured with the optional FT-850 flow-through cell module and peristaltic pump assembly, the system complies with USP & JP specifications for Apparatus 4 operation.
Can the system be validated for GMP production release testing?
Yes—the platform is designed for GMP use, with full CSV documentation, electronic signature support, and audit trail retention exceeding 15 years as configured.
Is temperature uniformity verified across all eight vessels?
Yes—each vessel is equipped with an independent Pt100 sensor calibrated to ±0.05 °C; system-level qualification includes mapping per USP & ASTM E2911-22.
What analytical instruments can be directly interfaced?
UV-Vis spectrophotometers (via TTL trigger), HPLC autosamplers (RS-232/485), fiber-optic UV probes, dilution workstations (e.g., Gilson GX-271), and mass spectrometry interfaces via custom API integration.
How is data integrity maintained during power interruption?
The DSC-800 controller features non-volatile flash memory and automatic resume-on-reboot functionality; all pending samples and timestamps are preserved without loss.