Loobo LB-QM6 Handheld ATP Bioluminescence Detector
| Brand | Loobo |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Authorized Distributor |
| Country of Manufacture | China |
| Model | LB-QM6 |
| Detection Sensitivity | 1×10⁻¹⁵ mol ATP |
| RLU Range | 0–999,999 RLU |
| Background Noise | ≤2 RLU |
| Detection Limit | 1.0 CFU/mL |
| Assay Time | 10 s |
| Accuracy | ±5% or ±5 RLU |
| Overrange Protection | Tilt-activated interruption |
| Display | 3.5-inch color TFT LCD |
| Data Storage | 10,000 records |
| Connectivity | USB + Bluetooth v5.0 |
| Output Options | Optional Bluetooth thermal printer |
| Power Supply | Rechargeable Li-ion battery (AC travel charger included |
| Battery Life | >10 h |
| Operating Temperature | 5–40 °C |
| Operating Humidity | 20–80% RH |
| Dimensions | 70 × 35 × 190 mm |
| Weight | 300 g |
Overview
The Loobo LB-QM6 Handheld ATP Bioluminescence Detector is an engineered field-deployable instrument designed for rapid, quantitative assessment of surface and liquid microbial contamination based on adenosine triphosphate (ATP) bioluminescence chemistry. It operates on the principle that ATP—present in all metabolically active cells—is quantitatively converted to visible light via a luciferin–luciferase enzymatic reaction. The emitted photon flux is measured in relative light units (RLUs) and linearly correlated with viable microbial load. Unlike culture-based methods requiring 24–72 h, the LB-QM6 delivers results within 10 seconds, enabling real-time hygiene verification in time-critical environments. Its compact form factor (70 × 35 × 190 mm), sub-300 g mass, and >10-hour battery life make it suitable for continuous use across multi-site audits, mobile inspection teams, and resource-constrained field laboratories.
Key Features
- High-sensitivity photomultiplier tube (PMT) detector with optimized optical path design, achieving a detection limit of 1×10⁻¹⁵ mol ATP and background noise ≤2 RLU—meeting ISO 22000 and HACCP prerequisite program requirements for environmental monitoring.
- True 10-second assay cycle: integrated auto-triggered luminometer activation upon swab insertion, eliminating manual timing errors and ensuring inter-operator reproducibility.
- Ruggedized industrial housing rated IP54 for dust and splash resistance, validated for operation across 5–40 °C and 20–80% RH—suitable for cold-chain facilities, steam-intensive kitchens, and outdoor regulatory inspections.
- Calibration traceability: factory-calibrated against NIST-traceable ATP standards; user-accessible calibration verification mode supports routine performance checks per ISO/IEC 17025 internal quality control protocols.
- Dual-mode data capture: stores 10,000 timestamped RLU records with operator ID, location tag, and sampling point metadata—enabling longitudinal trend analysis and audit-ready reporting.
Sample Compatibility & Compliance
The LB-QM6 is validated for use with commercially available ATP swabs (e.g., 3M Clean-Trace™, Hygiena UltraSnap™, and Loobo-certified swabs), supporting standardized sampling of flat surfaces, irregular equipment contours, food contact zones, and aqueous samples (including rinse water and process effluents). It complies with ASTM E2631–22 “Standard Practice for Determining the Presence of Microbial Contamination Using Adenosine Triphosphate (ATP) Bioluminescence” and aligns with FDA Food Code Appendix 8-301.11 for environmental monitoring in retail food establishments. While not a direct colony-forming unit (CFU) counter, its 1.0 CFU/mL equivalent detection limit enables correlation with microbiological culture results under controlled validation studies—supporting GLP-compliant hygiene verification workflows in pharmaceutical cleanrooms and hospital central sterile supply departments (CSSD).
Software & Data Management
Data export is supported via USB mass storage mode or Bluetooth 5.0 pairing with Windows/macOS/iOS/Android platforms. The companion Loobo DataLink desktop application (v3.2+) provides automated RLU-to-CFU conversion tables, statistical process control (SPC) charts (X̄–R, CUSUM), and customizable pass/fail thresholds aligned with sector-specific benchmarks (e.g., <100 RLU for food contact surfaces per ICMSF guidelines; <500 RLU for non-critical healthcare surfaces per CDC/HICPAC recommendations). Audit trails include full change logs, electronic signatures, and exportable PDF reports compliant with FDA 21 CFR Part 11 requirements when used with configured user roles and password-protected access.
Applications
- Food Manufacturing: Verification of cleaning-in-place (CIP) efficacy, post-sanitation validation of conveyor belts and fill nozzles, and supplier hygiene pre-audit screening.
- Foodservice & Retail: Daily operational checks of cutting boards, fryer baskets, refrigeration gaskets, and self-service kiosk touchpoints—integrated into daily sanitation checklists.
- Healthcare Facilities: Routine monitoring of high-touch surfaces in operating rooms, ICU bed rails, endoscope reprocessing sinks, and staff hand hygiene compliance tracking.
- Environmental Health: Rapid assessment of wastewater treatment influent/effluent, ballast water compliance, and public pool filtration system integrity.
- Regulatory Enforcement: On-the-spot evidence collection during health code inspections, including documentation for legal proceedings where chain-of-custody metadata is retained.
FAQ
Does the LB-QM6 require annual recalibration?
No—calibration is stable for 12 months under normal use. However, users must perform quarterly verification using Loobo ATP Standard Solution (Cat. No. QM-CAL-01) per ISO/IEC 17025 Section 6.5.2.
Can the device distinguish between bacterial and fungal ATP?
No. ATP bioluminescence detects total viable biomass regardless of taxonomic origin. Species identification requires complementary methods such as PCR or MALDI-TOF MS.
Is Bluetooth printing supported out-of-the-box?
Yes—the optional Loobo LP-200 thermal printer connects automatically via Bluetooth; printed reports include QR-coded RLU values and batch-swab IDs for traceability.
What swab types are compatible?
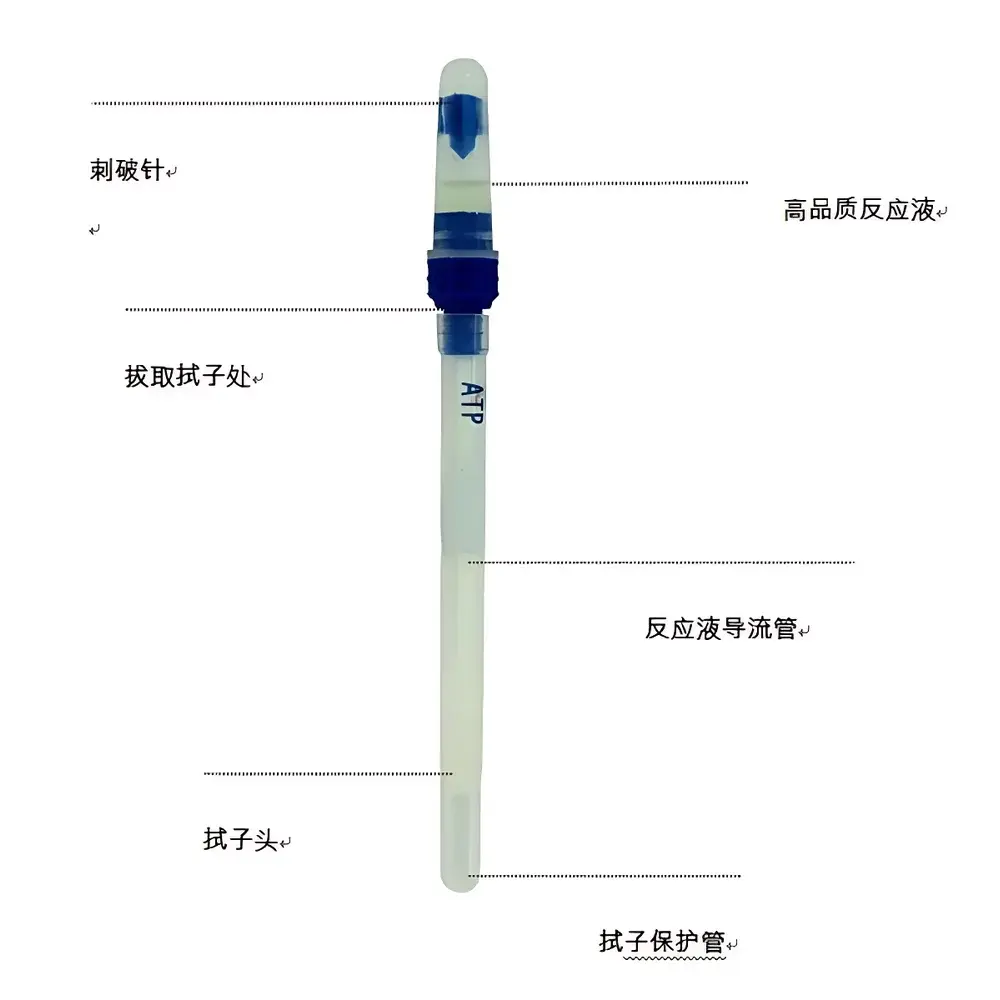
All industry-standard dry and wet swabs with ≥95% ATP recovery efficiency (validated per ASTM E2871–13); Loobo recommends swabs with lyophilized luciferase for ambient-temperature stability.
How is firmware updated?
Via USB-connected Loobo DataLink software; updates include security patches, RLU algorithm refinements, and new regulatory template libraries.