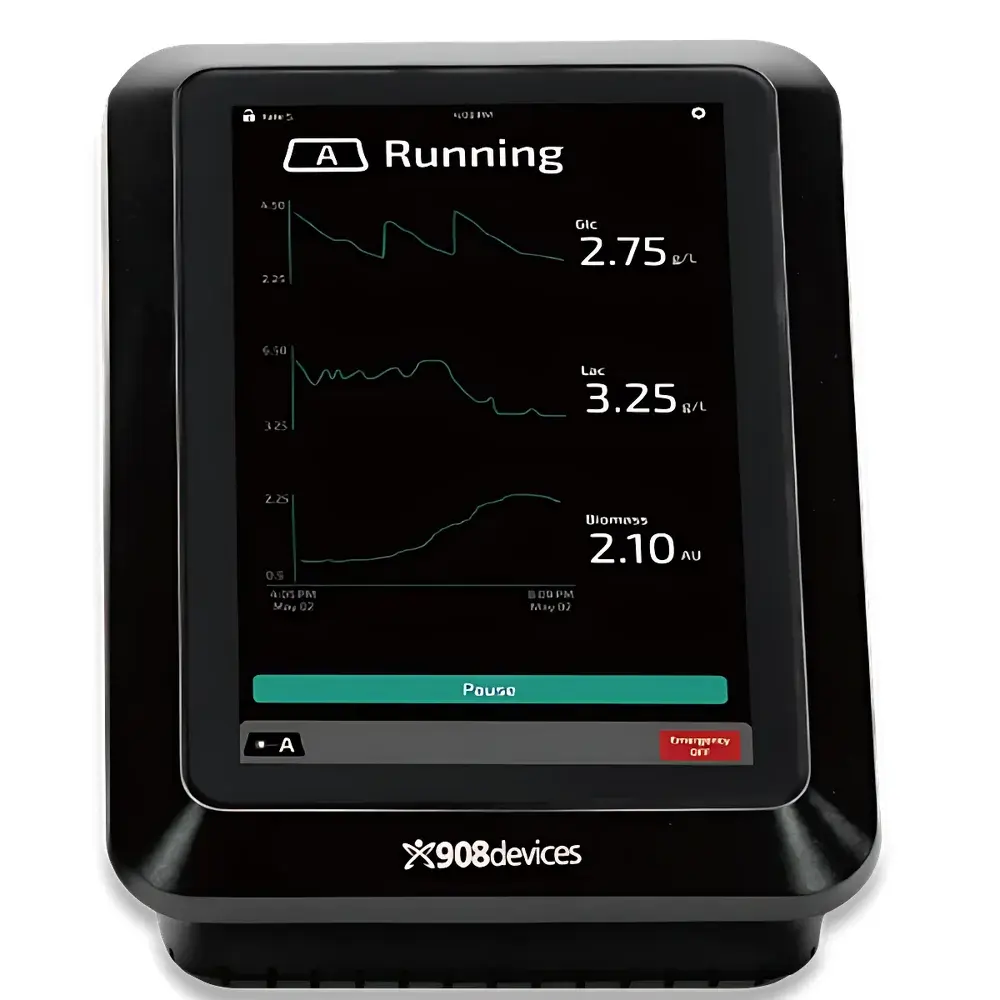
MAVERICK Raman-Based In-Line Bioreactor Process Analytical Technology System by 908 Devices
| Brand | 908 Devices |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | MAVERICK |
| Price Range | USD 1,500 – 150,000 (FOB) |
Overview
The MAVERICK system is an in-line, non-invasive process analytical technology (PAT) platform engineered for real-time, multi-analyte monitoring in mammalian and microbial bioreactors. It leverages high-fidelity Raman spectroscopy coupled with first-principles–driven De Novo modeling to deliver quantitative measurements of critical process parameters—including glucose, lactate, and total viable cell density—without reliance on empirical calibration or reference methods. Unlike conventional chemometric models trained on historical spectral datasets, the De Novo modeling framework embeds fundamental molecular vibrational signatures, stoichiometric relationships, and biophysical constraints directly into the algorithm architecture. This enables robust spectral interpretation across diverse cell lines, media formulations, and operating conditions—eliminating model revalidation cycles typically required during process scale-up or formulation changes.
Key Features
- Real-time, continuous in-situ measurement via fiber-optic Raman probe integrated into standard bioreactor ports (e.g., 1/4″ sanitary fittings)
- Simultaneous quantification of glucose (0.1–25 g/L), lactate (0.1–15 g/L), and total viable cell density (0.1–30 × 10⁶ cells/mL) with typical root-mean-square error (RMSE) < 5% relative to offline HPLC and viability assays
- De Novo modeling engine—fully physics-informed, requiring no user-generated calibration spectra or batch-specific training
- Self-diagnosing optical path integrity with automated background correction and laser power stabilization
- IP65-rated industrial enclosure with CE, UL 61010-1, and IEC 60601-1 compliance for integration into GMP manufacturing environments
- Plug-and-play compatibility with DeltaV, DCS, and SCADA systems via OPC UA and Modbus TCP protocols
Sample Compatibility & Compliance
The MAVERICK system is validated for use with common suspension and adherent mammalian cultures (CHO, HEK293, CAP-T), microbial systems (E. coli, P. pastoris), and perfusion and fed-batch modes. It operates without consumables, reagents, or sample extraction—thereby eliminating cross-contamination risk and supporting closed-system processing per USP <1043> and ISO 13408-1. All firmware and data handling comply with FDA 21 CFR Part 11 requirements, including electronic signature support, audit trail logging, and role-based access control. The system supports full traceability from raw spectrum acquisition to final parameter output, satisfying ALCOA+ principles for GxP-regulated biomanufacturing.
Software & Data Management
MAVERICK is operated through the cloud-connected 908 Devices Control Suite—a validated desktop application supporting method configuration, real-time dashboarding, alarm management (with configurable thresholds and hysteresis), and export to CSV, HDF5, or SQL databases. Raw Raman spectra (785 nm excitation, 5–3200 cm⁻¹ range) are archived with metadata (timestamp, reactor ID, operator, versioned model ID). Software updates follow a formal change control process aligned with ICH Q5A and Q9. Audit trails record all user actions—including parameter edits, model deployments, and report generation—with immutable timestamps and digital signatures. Integration with LIMS and MES platforms is supported via RESTful APIs and secure SFTP transfer.
Applications
- Glucose-lactate metabolic flux monitoring during peak production phases to inform feed strategy optimization
- Early detection of culture stress or contamination events via spectral deviation scoring
- Reducing reliance on offline sampling—cutting manual assay burden by >70% and minimizing bioreactor breach frequency
- Supporting Quality-by-Design (QbD) initiatives through continuous multivariate process understanding
- Accelerating tech transfer by eliminating model portability barriers between development, pilot, and commercial-scale reactors
- Enabling adaptive control loops when interfaced with advanced process controllers (e.g., model-predictive control)
FAQ
Does MAVERICK require periodic recalibration using reference standards?
No. The De Novo model is inherently stable and does not rely on empirical calibration; routine verification is performed using built-in NIST-traceable polystyrene reference peaks.
Can MAVERICK be deployed in single-use bioreactors?
Yes—compatible with industry-standard single-use bags equipped with Raman-accessible optical windows (e.g., Sartorius BIOSTAT STR, Thermo Fisher HyPerforma).
Is the system qualified for use in commercial GMP manufacturing?
Yes—validated IQ/OQ documentation packages are provided, and the platform has been installed in multiple FDA-inspected commercial facilities for Phase III and commercial supply.
How is data security ensured during cloud-based software updates?
All cloud communications use TLS 1.3 encryption; firmware updates are cryptographically signed and verified prior to installation.
What level of technical support is included post-purchase?
Standard coverage includes 24/7 remote diagnostics, annual performance verification, and priority access to application scientists for method troubleshooting and regulatory documentation support.