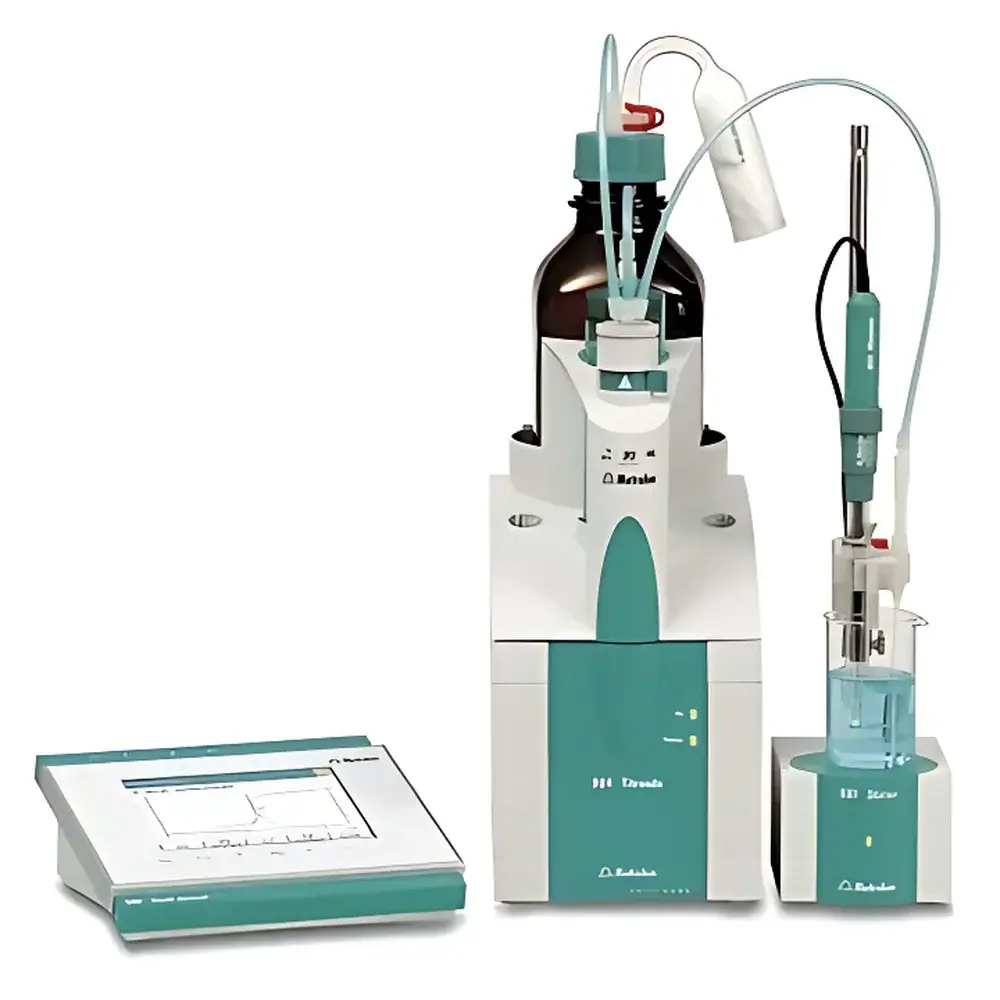
Metrohm 904 Titrando Automatic Potentiometric Titrator
| Brand | Metrohm |
|---|---|
| Origin | Switzerland |
| Manufacturer | Metrohm AG |
| Type | Universal Automatic Potentiometric Titrator |
| Model | 904 Titrando |
| Measurement Range | ±1200 mV, ±120 µA |
| Electrode Readout Accuracy | 0.15% (±30 µL at 20 mL) |
| Resolution | 0.001 pH, 0.1 mV, 0.1 °C |
| Titration Accuracy | 0.15% (±30 µL at 20 mL) |
| Compliance | FDA 21 CFR Part 11, GLP/GMP-ready |
| Interface | 4 × MSB ports, USB, PCMCIA, optional Bluetooth |
Overview
The Metrohm 904 Titrando is a high-precision, fully automated potentiometric titrator engineered for reproducible quantitative chemical analysis in regulated and research-intensive laboratory environments. Based on the core principle of potentiometric endpoint detection—measuring potential difference between a reference and indicator electrode as a function of titrant addition—the system delivers traceable, thermodynamically grounded results across acid-base, redox, precipitation, and complexometric reactions. Its architecture integrates Metrohm’s proprietary *e-Titration* intelligence platform, embedding microprocessors within critical components—including the 806 Dosino dispensing unit, smart electrodes, and digital measurement cables—to enable real-time calibration validation, automatic component recognition, and self-documenting operation. Designed as a modular, expandable platform, the 904 Titrando serves as the analytical backbone for centralized titration workflows in QC labs, pharmaceutical development, food safety testing, and environmental monitoring where regulatory compliance, audit readiness, and method portability are non-negotiable.
Key Features
- Integrated *e-Titration* architecture with embedded chips in electrodes, cables, and dosing units for automatic identification, firmware-aware operation, and electronic log generation
- 806 Dosino dispensing unit delivering 1:20,000 volumetric resolution (0.005% relative precision) and active temperature compensation for burette volume correction
- Triple titration mode support: DET (Dynamic Equivalence Titration), MET (Multi-Endpoint Titration), and SET (Setpoint Titration), including programmable pause, re-dose, and conditional branching logic
- Native support for Karl Fischer volumetric water determination (KF) and STAT (controlled-potential coulometric or potentiostatic) titrations via user-editable methods
- 90 mm × 120 mm full-color capacitive touchscreen with multilingual UI (English, German, French, Spanish, Chinese, Japanese), configurable soft keys, and real-time titration curve overlay
- Dual isolated measurement channels: primary pH/mV/°C input with secondary galvanically isolated input for simultaneous ISE concentration calculation or dual-parameter monitoring (e.g., pH + conductivity)
- Comprehensive GLP functionality: electrode health diagnostics (including slope, offset, and response time validation), electronic signatures, password-protected access levels, and audit-trail-enabled method/data versioning
Sample Compatibility & Compliance
The 904 Titrando accommodates aqueous and non-aqueous matrices across diverse sample types—including pharmaceutical actives, catalysts, petrochemical distillates, dairy products, and wastewater effluents—via interchangeable electrode configurations (glass pH, Ag/AgCl, Pt, ion-selective, redox, and KF-specific electrodes). Its hardware and software stack are validated against international regulatory frameworks: data integrity adheres to FDA 21 CFR Part 11 requirements through mandatory electronic signatures, role-based permissions, immutable audit trails, and tamper-evident file encryption. The system supports full GMP/GLP alignment via tiamo™ software integration, enabling compliant method lifecycle management, instrument qualification documentation (IQ/OQ/PQ templates), and seamless linkage to LIMS via LabLink Ethernet or Wi-Fi. All electrical interfaces conform to IEC 61000-4 EMC standards; temperature sensing complies with IEC 60751 (Pt1000) or DIN 43760 (NTC).
Software & Data Management
Control and data handling are centralized through Metrohm’s tiamo™ software suite—a Windows-based platform certified for 21 CFR Part 11 compliance. tiamo™ provides structured database architecture for method storage (including KF and STAT protocols), sample registration, result reporting (PDF/CSV/XLS export), and statistical evaluation (RSD, confidence intervals, trend analysis). It enables parallel titration scheduling across multiple 904 units, remote supervision via LabLink network deployment, and direct synchronization with enterprise SQL servers. The integrated PCMCIA slot supports secure method backup, user credential cards, and encrypted flash storage for offline operation. All raw measurement data (100 ms sampling interval), intermediate calculations, and operator actions are timestamped and cryptographically hashed to ensure forensic traceability from sample receipt to final report.
Applications
- Pharmaceutical QA/QC: assay of active ingredients (e.g., amine titration in APIs), residual chloride quantification, excipient purity verification, and KF water content per USP <921>
- Food & Beverage: titratable acidity in wines and juices, salt content (argentometric), preservative (sorbate/benzoate) determination, and peroxide value in oils
- Environmental Testing: alkalinity and hardness profiling in surface water, cyanide and fluoride analysis by ISE, and chloride in industrial effluents per ISO 9297
- Chemical Manufacturing: catalyst metal content (e.g., Ni, Co via complexometric EDTA titration), acid number in lubricants (ASTM D974), and saponification value (ISO 3657)
- Academic Research: mechanistic studies of multi-step redox systems, pKa determination via Gran plot analysis, and kinetic endpoint modeling in slow-reaction titrations
FAQ
Does the 904 Titrando support ASTM or ISO standard-compliant titration methods?
Yes—preconfigured method templates for ASTM D974 (acid number), ISO 8587 (sensory analysis correlation), ISO 9297 (chloride), and USP <921> (water by KF) are available in tiamo™ and fully editable to match lab-specific SOPs.
Can the system operate without a PC connection?
Yes—the embedded touchscreen interface allows standalone method execution, real-time curve visualization, and local result export via USB; however, full GLP audit trail, database management, and advanced statistics require tiamo™.
What electrode diagnostics are included under GLP-certified testing?
The system performs automated slope/offset verification, response time measurement (t95), junction potential stability checks, and memory-based electrode history tracking—including calibration dates, usage cycles, and failure alerts.
Is wireless connectivity limited to printers and balances?
Bluetooth 4.0 supports Class 1 peripherals (balances, printers, barcode scanners); for secure network integration, LabLink uses TLS-encrypted Ethernet/Wi-Fi with VLAN segmentation and firewall-compatible port configuration.
How is method security enforced during multi-user operation?
tiamo™ implements hierarchical user roles (Administrator, Supervisor, Analyst, Viewer), cryptographic signing of method modifications, and automatic rollback to last-approved versions upon unauthorized change detection.