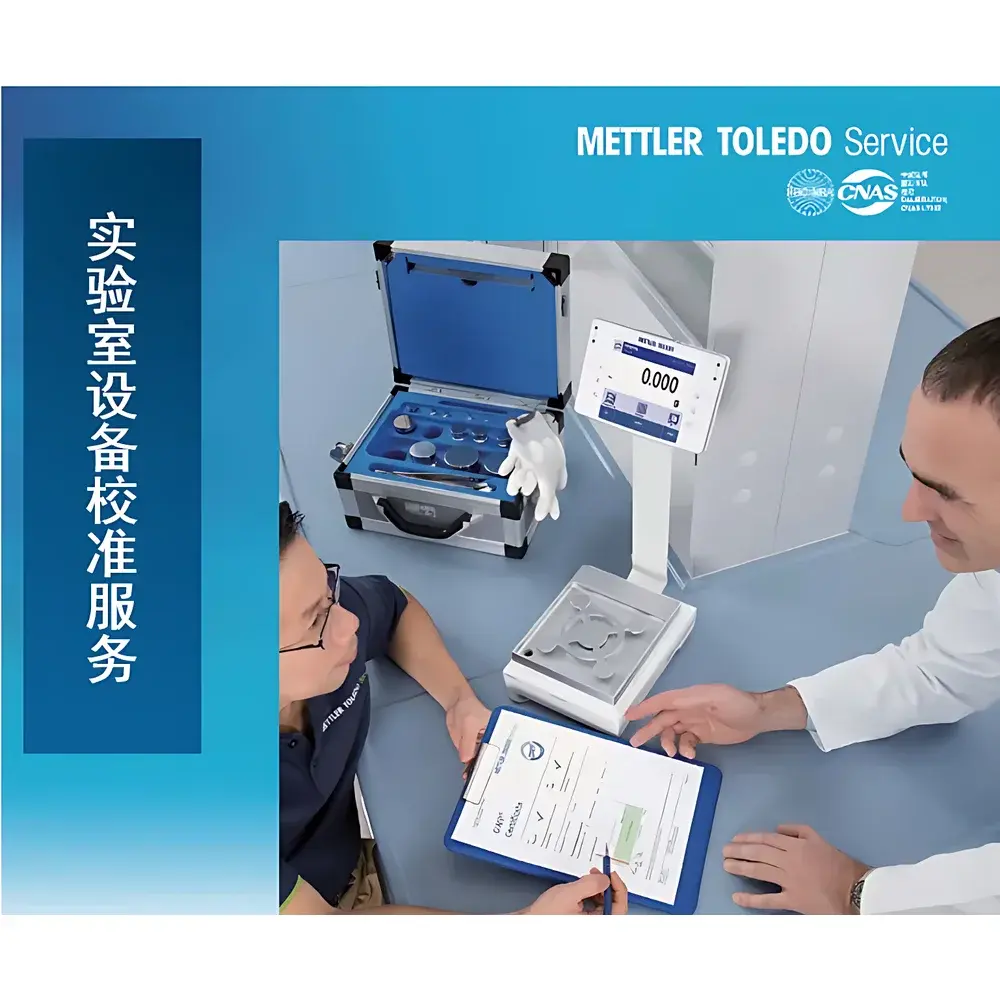
METTLER TOLEDO Pipette Calibration and Maintenance Service
| Brand | METTLER TOLEDO |
|---|---|
| Origin | Switzerland |
| Manufacturer Status | Authorized Distributor |
| Product Origin | Imported |
| Service Type | Instrument Calibration |
| Equipment Category | Laboratory Common Equipment |
| Service Experience | 100 Years |
| Response Time | Within 24 Hours |
| Scope of Certification/Calibration | Performance Verification, Uncertainty-Evaluated Calibration, and Preventive Maintenance per ISO 8655 and JJG 646 |
Overview
The METTLER TOLEDO Pipette Calibration and Maintenance Service is a traceable, uncertainty-quantified metrological service designed to ensure volumetric accuracy, repeatability, and long-term reliability of single-channel and multichannel air-displacement pipettes used across pharmaceutical QC, clinical diagnostics, academic research, and contract testing laboratories. Grounded in the fundamental principles of gravimetric measurement—where dispensed liquid mass is converted to volume using certified reference water density, temperature-controlled weighing, and buoyancy-corrected balances—the service complies with internationally recognized standards including ISO 8655 (Pipettes – Gravimetric and Photometric Methods), ISO/IEC 17025 (General Requirements for the Competence of Testing and Calibration Laboratories), and national verification regulation JJG 646 (China National Metrological Verification Regulation for Piston-Type Volumetric Apparatus). As an ISO/IEC 17025-accredited calibration laboratory under CNAS (China National Accreditation Service), METTLER TOLEDO’s service delivers legally defensible calibration certificates with full measurement uncertainty budgets, enabling compliance with FDA 21 CFR Part 11, EU Annex 11, and GLP/GMP data integrity requirements.
Key Features
- Full-scope gravimetric calibration per ISO 8655–2 (volumetric accuracy) and ISO 8655–5 (repeatability), covering nominal volumes from 0.1 µL to 10 mL
- Measurement uncertainty evaluation performed according to GUM (Guide to the Expression of Uncertainty in Measurement), with expanded uncertainty (k=2) reported for each calibrated volume point
- Preventive maintenance protocol including tip ejector mechanism inspection, piston seal integrity test, internal contamination assessment, and ergonomic adjustment verification
- Traceable calibration certificates issued with unique certificate ID, calibration date, next due date, environmental conditions, operator ID, and full uncertainty statement
- On-site or return-to-lab service options, with guaranteed response time ≤24 hours upon service request submission
- Customized calibration intervals and multi-point verification schemes aligned with risk-based assessment (e.g., high-use vs. low-use pipettes in regulated environments)
Sample Compatibility & Compliance
This service supports all major brands of air-displacement pipettes—including METTLER TOLEDO E4/X4 series, Rainin LTS/XLS+, Gilson PIPETMAN, Thermo Fisher Finnpipette, and BrandTech Transferpette—regardless of channel count (single, 8-, 12-, or 16-channel) or volume range. All calibrations are conducted in climate-controlled laboratories (20.0 ± 0.5 °C, 45–65% RH) using Class I analytical balances traceable to NIST or BIPM standards. The service meets regulatory expectations for instrument qualification under USP <1058>, ASTM E2913, and ISO/IEC 17025 clause 6.5.2 on equipment verification. Certificates include statements of conformance to ISO 8655 and JJG 646, with optional audit-ready digital records compatible with LIMS integration.
Software & Data Management
Calibration records are managed via METTLER TOLEDO LabX® Calibration Module—a secure, 21 CFR Part 11-compliant platform supporting electronic signatures, role-based access control, automated certificate generation, and audit trail logging for all calibration events. Historical performance trends, drift analysis, and recalibration alerts are automatically generated. Data exports comply with CSV, PDF/A-1b, and XML formats for seamless import into enterprise quality management systems (QMS) or electronic lab notebooks (ELN). Full traceability is maintained from individual pipette serial number through calibration event to raw gravimetric data files.
Applications
This service is routinely deployed in settings where volumetric accuracy directly impacts analytical validity: drug substance potency assays, ELISA and PCR assay preparation, cell culture media dispensing, reference standard dilution workflows, and method validation studies. It is especially critical for laboratories undergoing regulatory inspections (e.g., FDA pre-approval inspections, MHRA audits, or PMDA GMP reviews), where documented evidence of pipette performance—supported by uncertainty-evaluated calibration—is required to demonstrate measurement traceability and data reliability.
FAQ
What standards does this calibration service follow?
It adheres strictly to ISO 8655 (Parts 2, 5, and 7), JJG 646, and ISO/IEC 17025 requirements for accredited calibration laboratories.
Is measurement uncertainty reported on the certificate?
Yes—each calibrated volume point includes expanded uncertainty (k = 2) calculated per GUM principles, covering contributions from balance repeatability, temperature variation, water density uncertainty, and evaporation correction.
Can preventive maintenance be performed without full calibration?
Yes—preventive maintenance is offered as a standalone service and includes mechanical inspection, cleaning, and functional verification; however, only full calibration provides metrological traceability and uncertainty quantification.
How often should pipettes be calibrated?
Frequency depends on usage intensity, criticality of application, and quality system requirements; typical intervals range from quarterly (high-use QC labs) to annually (low-frequency research use), with risk-based justification supported by LabX trend reports.
Do you calibrate positive displacement pipettes?
This service covers air-displacement pipettes exclusively; positive displacement pipettes require specialized photometric or syringe-pump-based methods and are addressed under separate service protocols.