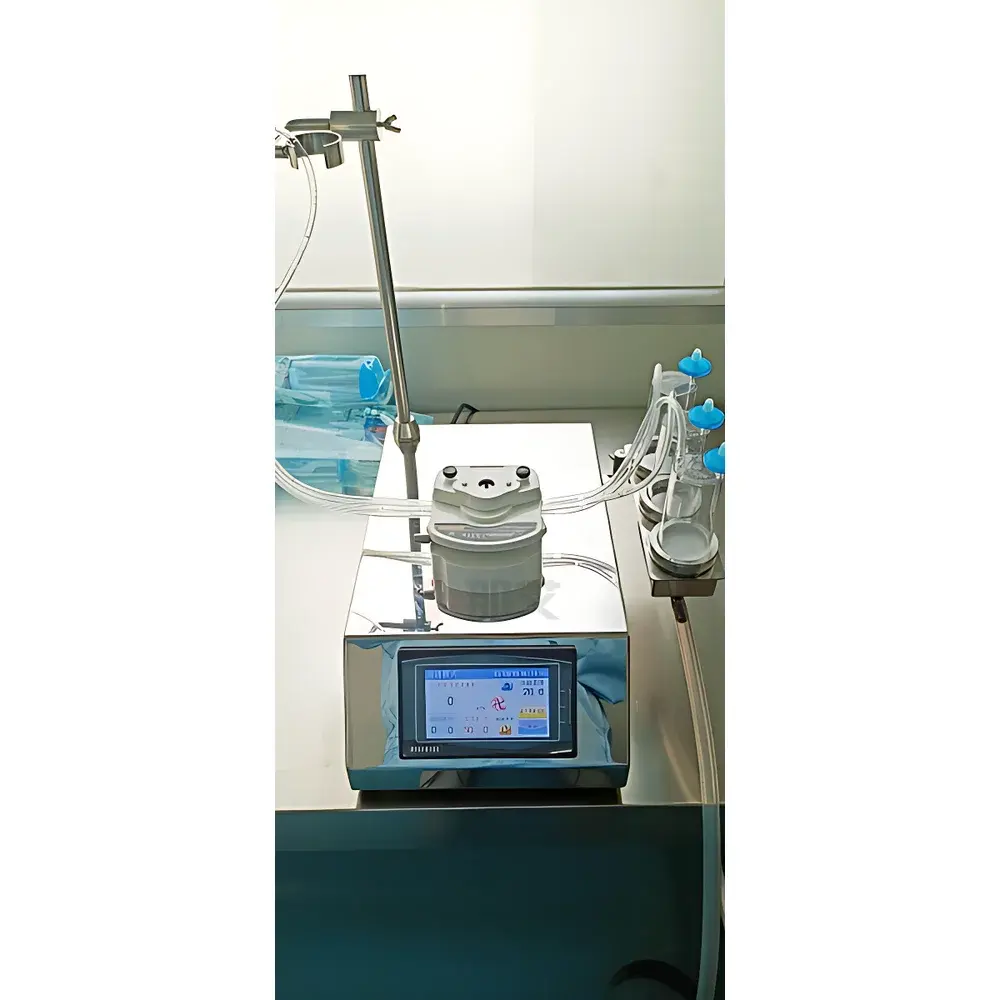
NAI-JJY-Z Portable Sterile Filtration System for Microbial Limit Testing – Mirror-Finish Stainless Steel Bacterial Collection Instrument
| Brand | NAI Instruments |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Regional Category | Domestic (China) |
| Model | NAI-JJY-Z |
| Pricing | Available Upon Request |
Overview
The NAI-JJY-Z Portable Sterile Filtration System is a precision-engineered bacterial collection instrument designed specifically for microbial limit testing in regulated pharmaceutical, biotechnology, food, and beverage laboratories. It operates on the principle of pressure-driven sterile filtration using peristaltic pump technology to deliver controlled, consistent flow across hydrophilic or hydrophobic microporous membranes (typically 0.45 µm or 0.22 µm pore size). The system facilitates the quantitative recovery and retention of viable microorganisms from liquid samples onto filter surfaces—enabling subsequent membrane transfer to appropriate culture media for enumeration or identification. Fully compliant with the Chinese Pharmacopoeia (ChP) 2020 Edition, Section 1101 “Sterility Tests” and Section 1105 “Microbial Limits Tests”, it supports both membrane filtration and direct inoculation protocols under aseptic conditions. Its compact footprint, mirror-finish stainless steel housing, and seamless surface geometry minimize particle entrapment and support rigorous cleaning validation in ISO Class 5 (Grade A) environments or laminar flow hoods.
Key Features
- PLC-based control architecture ensures high repeatability and operational stability across diverse sample volumes—from small-volume injectables (e.g., 10 mL vials) to large-volume parenterals (e.g., 2 L IV bags)
- 4.3-inch capacitive touchscreen interface displays real-time parameters: pump speed (0–300 rpm), elapsed runtime, ambient temperature, system status, and integrated digital clock
- Ergonomic quick-fit peristaltic pump head with corrosion-resistant stainless-steel rollers enables rapid tubing installation and uniform flow distribution without pulsation-induced membrane damage
- Monolithic mirror-polished 304 stainless steel chassis eliminates crevices, weld seams, and threaded fasteners—meeting surface finish requirements (Ra ≤ 0.8 µm) for Grade A cleanroom compatibility
- Modular removable waste trough with optimized arc-shaped interior prevents residual fluid pooling and biofilm formation; validated for autoclaving at 121°C for 20 minutes
- Dual-size universal sample holder accommodates standard 100 mm and 150 mm petri dishes, as well as 50–500 mL specimen bottles—ensuring mechanical stability during filtration
- Integrated safety interlock system halts pump operation upon door opening or abnormal pressure rise, protecting operator integrity and sample sterility
- IP65-rated footswitch included as standard—enabling hands-free start/stop functionality during glove-compatible workflows
- On-device QR code links directly to bilingual (EN/CN) electronic user manual, firmware update portal, and NAI technical support ticketing system
Sample Compatibility & Compliance
The NAI-JJY-Z supports filtration of aqueous and mildly organic solutions—including saline, buffered isotonic media, antibiotic formulations, herbal extracts, soft drinks, dairy products, and low-viscosity beverages. It is routinely deployed in compliance with ChP 2020, USP <71> Sterility Tests, USP <61> Microbial Enumeration Tests, and ISO 11737-1:2018 for bioburden assessment. When paired with certified sterile, pyrogen-free, low-binding cellulose ester or PVDF membrane filters and single-use closed-circuit filtration assemblies (e.g., NAI-SCF series), the system satisfies ALCOA+ data integrity principles. Full audit trail capability—including timestamped event logs for pump activation, duration, and speed—is retained locally and exportable via USB for GLP/GMP documentation.
Software & Data Management
While the NAI-JJY-Z operates as a standalone hardware platform without cloud connectivity, its embedded controller maintains non-volatile memory for up to 1,000 test records. Each entry includes date/time stamp, user ID (assignable via touchscreen), selected speed profile, total runtime, and error flags (e.g., occlusion detection, motor stall). Data export is supported via USB 2.0 port in CSV format—compatible with LIMS integration and FDA 21 CFR Part 11-compliant systems when used with validated third-party software. Firmware updates are delivered through signed binary packages verified via SHA-256 checksum to ensure integrity and traceability.
Applications
- Pharmaceutical Quality Control: Sterility testing of aseptically filled vials, syringes, and IV bags; microbial limit testing of APIs, excipients, and purified water (PW)/WFI
- Medical Device Validation: Bioburden assessment of sterilized catheters, surgical kits, and implant packaging leachates
- Food & Beverage Safety: Quantitative recovery of Escherichia coli, Staphylococcus aureus, and Candida albicans from ready-to-drink beverages, infant formula, and fermented dairy products
- Environmental Monitoring: Air and surface sampling via membrane filtration followed by RODAC plate transfer or direct incubation
- Academic & Contract Research: Method development for challenging matrices containing surfactants, preservatives, or particulates requiring neutralization wash steps prior to filtration
FAQ
Is the NAI-JJY-Z suitable for use in ISO Class 5 (Grade A) environments?
Yes—the mirror-finish stainless steel housing, absence of painted surfaces or gasketed seams, and smooth-radius internal geometry allow full compatibility with vaporized hydrogen peroxide (VHP) decontamination cycles and routine ethanol/isopropanol wiping.
Can the system be validated per IQ/OQ/PQ protocols?
Yes—NAI provides a comprehensive validation support package including URS templates, FAT/SAT checklists, calibration certificates for speed and timer functions, and risk-based PQ test scripts aligned with Annex 1 and EU GMP Annex 15.
What types of filter membranes are compatible?
The instrument accepts standard 47 mm and 50 mm diameter disc membranes mounted in reusable or disposable holders, as well as integrated all-in-one sterile filtration assemblies (e.g., NAI-SCF-47 and NAI-SCF-50 series) with pre-attached tubing and vent filters.
Does the unit meet electromagnetic compatibility (EMC) standards for laboratory use?
Yes—it complies with IEC 61326-1:2012 for laboratory equipment emissions and immunity, and has passed CE marking for EMC and LVD directives.
What maintenance is required to sustain performance over time?
Routine maintenance includes weekly visual inspection of tubing wear, monthly roller lubrication with food-grade silicone grease, and quarterly verification of pump speed accuracy using an external tachometer—documented in the provided maintenance logbook.