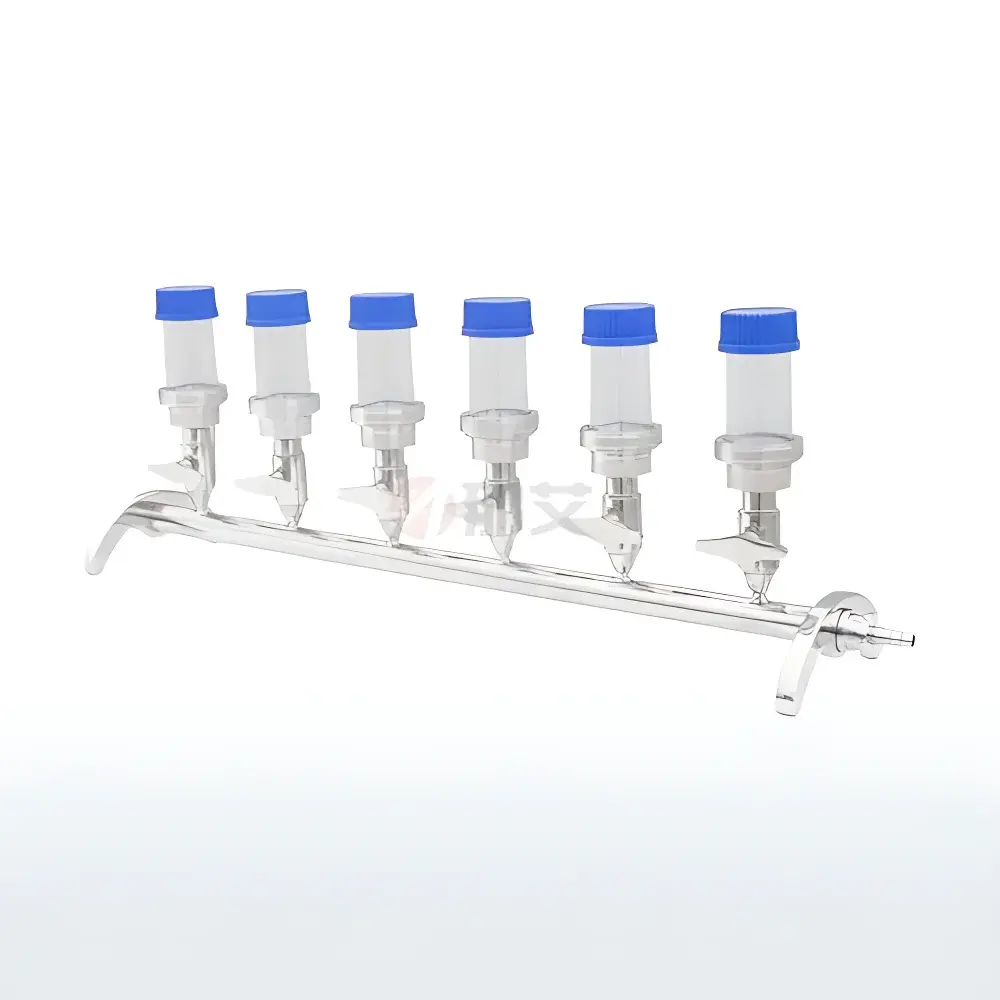
NAI-STV6 Stainless Steel Microbial Limit Filtration System with 6-Position Vacuum Manifold
| Brand | NAI Instruments |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | NAI-STV6 |
| Price | Upon Request |
| Construction Material | AISI 304 Stainless Steel |
| Filter Cup Configuration | 6 × Reusable 3B-Type Cups |
| Included Components | 6 × Filter Cups, 6 × 500 mL Borosilicate Glass Filtration Bottles, 6 × Sterile Bent Tubing Assemblies |
| Filter Membrane Compatibility | 0.45 µm or 0.22 µm, Ø50 mm |
| Max Vacuum Level | −0.08 MPa (−0.8 bar) |
| Air Flow Rate | ≥50 L/min at Rated Vacuum |
| Sterilization | Autoclavable (121 °C, 20–30 min, full manifold assembly) |
Overview
The NAI-STV6 Stainless Steel Microbial Limit Filtration System is a precision-engineered vacuum manifold designed for quantitative microbial recovery in compliance with pharmacopoeial methods—including the Chinese Pharmacopoeia (2015 Edition), USP <61>, EP 2.6.12, and ISO 8573-7. It implements membrane filtration via controlled negative-pressure aspiration to isolate viable microorganisms from liquid samples onto standardized microporous membranes. The system operates on the principle of size-exclusion filtration: when a defined volume of sample is drawn through a sterile 0.45 µm (or optionally 0.22 µm) cellulose ester or PVDF membrane under regulated vacuum, bacteria, yeasts, molds, and other particulate bioburden are retained on the membrane surface while filtrate passes into the collection bottle. Subsequent membrane transfer to selective or non-selective solid media enables colony enumeration and identification—forming the foundational step in sterility assurance, environmental monitoring, and product release testing.
Key Features
- Full AISI 304 stainless steel construction ensures corrosion resistance, mechanical rigidity, and compatibility with autoclaving (121 °C, 20–30 min, full assembly including manifold, cups, and support rods)
- Modular 6-position manifold supports parallel processing of up to six samples without cross-contamination; each filter cup operates independently with individual on/off valve control
- Reinforced 3B-type reusable filter cups engineered for repeated sterilization cycles and long-term dimensional stability—reducing consumable cost per test versus disposable alternatives
- Integrated ergonomic membrane retrieval mechanism allows rapid, aseptic removal of the filter disc using standard forceps or dedicated lift tools—minimizing handling-induced contamination risk
- Optimized flow-path geometry and low-resistance bent tubing assemblies maintain consistent vacuum distribution across all six channels, ensuring reproducible filtration rates and uniform membrane wetting
Sample Compatibility & Compliance
The NAI-STV6 is validated for use with aqueous and low-viscosity non-aggressive liquids across pharmaceutical, biotechnology, food & beverage, cosmetics, and environmental sectors. Compatible sample types include purified water, WFI, bulk drug substances, oral suspensions, injectables, bottled water, process rinse solutions, and surface rinse extracts. All filtration procedures align with regulatory expectations for method validation per ICH Q5A(R2) and data integrity requirements under FDA 21 CFR Part 11 and EU Annex 11. The system supports GLP/GMP-compliant documentation workflows when paired with validated electronic lab notebooks or LIMS-integrated reporting tools. Membrane specifications meet pharmacopoeial mandates: pore size ≤0.45 µm (standard), nominal diameter 50 mm, low extractables, and high bubble point consistency.
Software & Data Management
As a hardware-only filtration platform, the NAI-STV6 does not incorporate embedded firmware or onboard software. Its design prioritizes mechanical reliability and regulatory transparency—eliminating unvalidated digital layers that complicate audit trails. Vacuum source operation (e.g., diaphragm or oil-free rotary vane pumps) is managed externally via analog pressure gauges or third-party pump controllers with analog/digital outputs. Users integrate the system into broader quality management ecosystems by logging critical parameters manually or via connected sensors: filtration start/stop timestamps, applied vacuum level (kPa), total filtered volume (mL), membrane lot number, operator ID, and incubation conditions. This approach satisfies ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) for raw data capture in regulated environments.
Applications
- Pharmaceutical QC labs performing microbial limit tests per ChP 1107, USP <61>, and JP 4.06
- Biologics manufacturing facilities conducting routine environmental monitoring of compressed gases, HVAC systems, and cleanroom surfaces
- Contract testing laboratories supporting GMP audits with traceable, repeatable filtration protocols
- FDA-registered food processors verifying microbiological safety of finished beverages and potable water sources
- Cosmetic formulation labs assessing preservative efficacy (Challenge Testing) and raw material bioburden
- Public health laboratories screening municipal water supplies, wastewater effluents, and recreational water bodies per ISO 9308-1 and EPA Method 1603
FAQ
Is the NAI-STV6 compliant with USP <61> and USP <62>?
Yes—the system’s physical configuration, membrane specifications (0.45 µm, Ø50 mm), and operational methodology fully support the membrane filtration procedure described in USP chapters <61> (Microbiological Examination of Nonsterile Products) and <62> (Microbiological Examination of Sterile Products).
Can the manifold be sterilized in an autoclave?
Yes—the entire stainless steel assembly, including the manifold base, support rods, and filter cup housings, is rated for steam sterilization at 121 °C for 20–30 minutes. Ensure all elastomeric seals (e.g., silicone O-rings) are rated for repeated autoclaving.
What vacuum pump specifications are recommended?
A vacuum pump capable of sustaining ≥−0.08 MPa (−0.8 bar) with a free-air displacement of ≥50 L/min is required. Oil-free diaphragm pumps are preferred for cleanroom applications to avoid hydrocarbon contamination.
Are replacement membranes and accessories available through NAI Instruments?
Yes—NAI provides certified 0.45 µm and 0.22 µm cellulose nitrate, mixed ester, and PVDF membranes (Ø50 mm), as well as sterile bent tubing kits, glass filtration bottles, and silicone gaskets compatible with the STV6 platform.
Does the system support integration with LIMS or ELN platforms?
While the NAI-STV6 itself has no digital interface, its manual operation protocol is fully compatible with LIMS/ELN workflows when users record filtration metadata—including operator, date/time, sample ID, membrane lot, vacuum reading, and volume—using structured electronic forms or barcode-scanned templates.