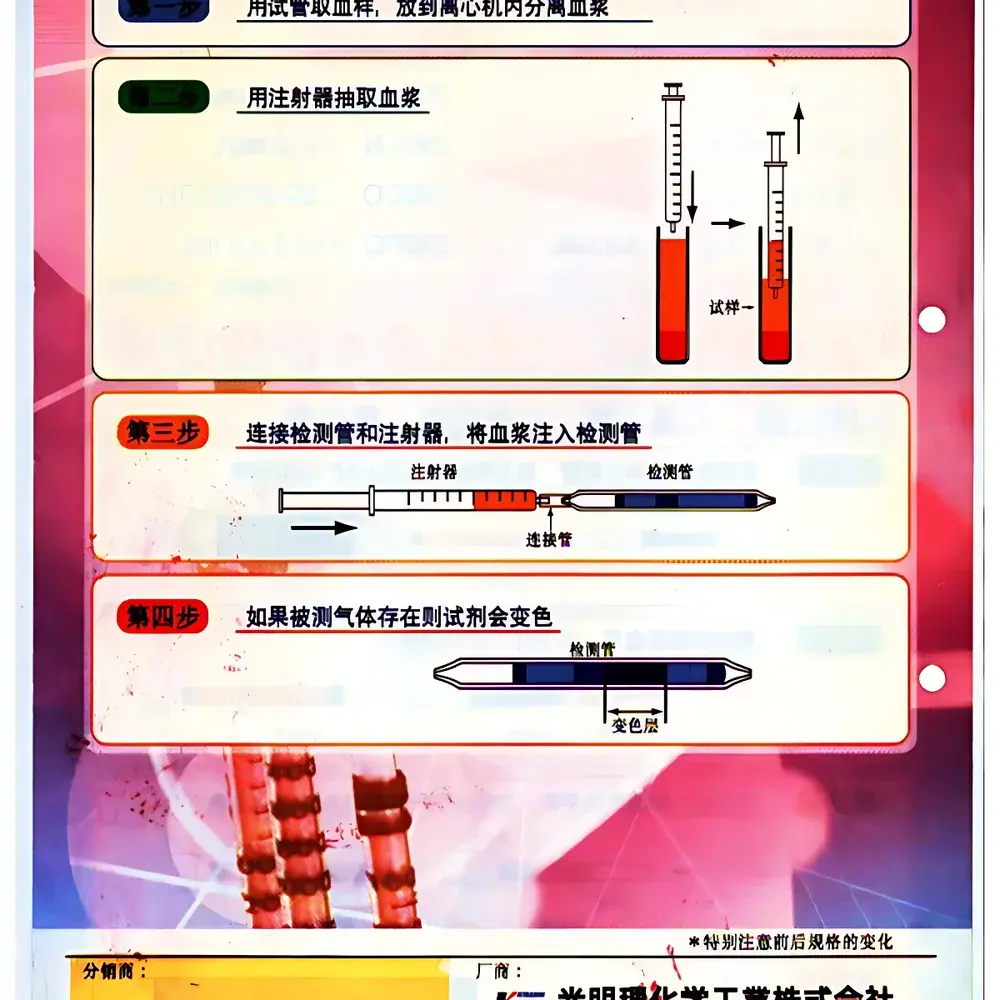
NorthKawa Rapid Blood Toxicant Detection Tube Kit (Model 290CN, 290EA, 290CO, 290H2S, 290PQ, 290OP, 290PA)
| Brand | Meiko Rikka |
|---|---|
| Origin | Japan |
| Model Numbers | 290CN, 290EA, 290CO, 290H2S, 290PQ, 290OP, 290PA |
| Detection Time | ≤60 s per test |
| Sample Volume | 10 mL whole blood |
| Sample Type | Human peripheral venous or capillary blood |
| Loading Mode | Semi-automatic manual insertion |
| Detection Limits | Malathion ≥0.004% (w/v), Dichlorvos ≥0.008% (w/v), Glufosinate ≥0.02% (w/v), Glyphosate ≥0.02% (w/v) |
| Regulatory Compliance | Designed per Japanese National Police Agency Forensic Guidelines (NPA-FG-2018), ISO 15197:2013 principles for qualitative point-of-care toxicant screening |
Overview
The NorthKawa Rapid Blood Toxicant Detection Tube Kit is a field-deployable, qualitative colorimetric assay system engineered for immediate presumptive identification of life-threatening toxicants in human blood specimens. Developed in close collaboration with the Japanese National Police Agency’s Forensic Science Laboratories, this kit implements solid-phase chemical reaction cartridges—each tube contains pre-immobilized reagents optimized for specific toxicant classes. Upon contact with hemolyzed whole blood, target analytes trigger irreversible chromogenic reactions, producing visually discernible color transitions within 60 seconds. The system operates on the principle of competitive inhibition and enzymatic hydrolysis (for organophosphates) or metal-catalyzed oxidation (for cyanide, carbon monoxide, and hydrogen sulfide), enabling rapid, equipment-free triage at crime scenes, emergency departments, or disaster response units. It is not intended for quantitative diagnosis or replacement of confirmatory methods such as GC-MS or LC-MS/MS but serves as a critical first-line decision support tool to prioritize sample routing and clinical intervention.
Key Features
- Field-ready design: No power supply, instrumentation, or calibration required—results interpreted visually under ambient lighting
- Dedicated tube variants for seven high-priority toxicant categories: organophosphorus pesticides (290OP), phosphinothricin-class herbicides (290PA), hydrogen cyanide (290CN), carbon monoxide (290CO), ethanol (290EA), hydrogen sulfide (290H2S), and paraquat dichloride (290PQ)
- Validated detection limits aligned with forensic toxicology action thresholds: e.g., ≥0.004% (w/v) malathion, ≥0.02% (w/v) glyphosate
- Whole-blood compatibility: Requires only 10 mL of uncentrifuged peripheral or capillary blood; no centrifugation or plasma separation needed
- Stable shelf life: Tubes retain full reactivity for ≥24 months when stored at 2–8 °C; desiccant-integrated packaging ensures humidity resistance
- Robust architecture: Polypropylene housing with integrated filter membrane prevents hemoglobin interference and minimizes false positives from bilirubin or methemoglobin
Sample Compatibility & Compliance
The kit accepts fresh, chilled, or EDTA-anticoagulated whole blood specimens. Hemolysis is intentionally leveraged to release intracellular targets (e.g., cyanide-binding cytochrome c oxidase residues), enhancing sensitivity for metallo-toxicants. Each lot undergoes batch certification per NPA-FG-2018 Annex D for specificity against common interferents—including acetaminophen, salicylates, ethanol metabolites, and therapeutic anticoagulants. While not CE-IVD marked or FDA 510(k)-cleared, the methodology adheres to ISO 15197:2013 foundational principles for qualitative point-of-care assays, including documented precision (inter-operator CV <8%), limit-of-detection verification, and stability under transport stress conditions (ISTA 2A). Documentation packages include Lot-Specific Analytical Certificates compliant with GLP documentation standards for forensic chain-of-custody reporting.
Software & Data Management
This is a non-instrumented, analog detection system; no embedded firmware, Bluetooth, or cloud connectivity is present. However, optional companion digital tools are available via licensed forensic case management platforms (e.g., Nihon Bunko ForensicLink v4.2), enabling barcode-scanned tube registration, geotagged result logging, and PDF report generation compatible with Japanese Prefectural Police Digital Evidence Repositories (DPER). All printed instruction leaflets include QR-coded access to multilingual validation summaries (English, Japanese, Spanish) and audit-trail templates meeting JIS Z 3001:2020 requirements for evidentiary documentation integrity.
Applications
- Forensic death investigation: Rapid differentiation between suicidal, homicidal, or accidental poisoning in field settings prior to autopsy
- Emergency medical response: Triage support for mass casualty incidents involving pesticide spills, industrial gas leaks, or alcohol-related trauma
- Hospital ED triage: Pre-laboratory screening to accelerate activation of antidote protocols (e.g., atropine/pralidoxime for OP exposure, hydroxocobalamin for cyanide)
- Occupational health surveillance: On-site monitoring following suspected workplace chemical exposure incidents
- Disaster medicine deployment: Lightweight, cold-chain-tolerant format suitable for mobile field labs in earthquake or flood zones
FAQ
Can these tubes be used with serum or plasma instead of whole blood?
No—validation data exclusively cover whole blood. Serum/plasma lacks erythrocyte-bound targets (e.g., methemoglobin for CO detection) and yields unreliable color development.
Is refrigeration required during transport?
Yes. Maintain 2–8 °C using validated cold packs (UN3373-compliant packaging); excursions above 30 °C for >4 hours invalidate the lot.
Do results meet court-admissible evidence standards in Japan?
Results serve as preliminary investigative findings under Article 199 of the Japanese Code of Criminal Procedure; confirmatory analysis by accredited labs (e.g., NPA Forensic Institute) is mandatory for judicial submission.
What is the shelf life after opening the foil pouch?
Use all tubes within 48 hours of pouch opening—exposure to ambient humidity degrades reagent integrity.
Are there cross-reactivity studies with common pharmaceuticals?
Yes. Full interference matrix testing per NPA-FG-2018 Appendix F confirms no clinically relevant cross-reactivity with warfarin, digoxin, insulin, or benzodiazepines at therapeutic concentrations.