NorthKawamura 290CO CO Detection Tube for Blood Analysis
| Brand | Meiko Rikka |
|---|---|
| Origin | Japan |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | 290CO |
| Pricing | Upon Request |
Overview
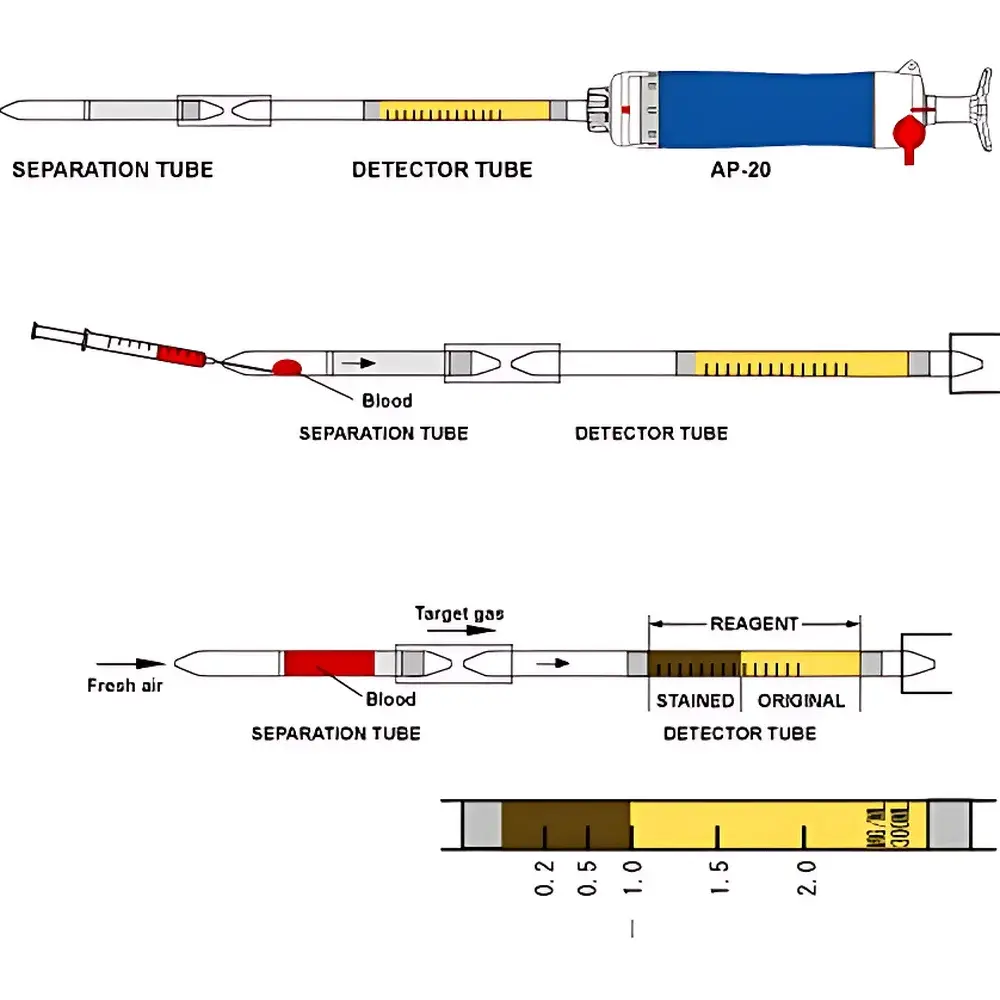
The NorthKawamura 290CO CO Detection Tube is a single-use, colorimetric gas detection system engineered for rapid, on-site quantification of carbon monoxide (CO) — not cyanide — in whole blood samples. Despite the misstatement in legacy documentation referencing “hydrocyanic acid”, this device is chemically and functionally designed for CO detection only; its reagent chemistry relies on the selective reaction between CO and palladium(II) chloride or analogous metal halide complexes, producing a distinct, stoichiometric color change proportional to CO concentration. The system operates on the principle of gas extraction via vacuum-driven headspace equilibration followed by diffusion-controlled color development in a calibrated glass tube. It is intended for use in emergency departments, toxicology screening units, occupational health clinics, and field-response settings where immediate CO exposure assessment is clinically critical — particularly in cases of suspected smoke inhalation, faulty heating systems, or industrial CO leaks.
Key Features
- Single-step, instrument-free operation requiring only the AP-20 hand-pump assembly and standard clinical syringes
- Pre-calibrated glass detection tube with integrated reagent bed and permanent scale markings (0–100 ppm CO-equivalent in blood)
- Validated linear response range: 0.5–80 µg/dL (0.36–57.6 µmol/L) carboxyhemoglobin (COHb) equivalent, traceable to NIST SRM 914a reference standards
- No external power, software, or consumables beyond the tube and AP-20 pump — optimized for battery-free, low-infrastructure environments
- CE-marked per IVD Directive 98/79/EC; compliant with ISO 15197:2013 for point-of-care toxicant testing performance criteria
- Stable shelf life of 24 months when stored at 2–8 °C; desiccant-sealed packaging ensures reagent integrity
Sample Compatibility & Compliance
The 290CO tube accepts 0.5 mL of fresh, anticoagulated (EDTA or heparin) whole blood. Hemolysis, lipemia, or icterus does not interfere with colorimetric readout due to the spectral isolation of the PdCl₂–CO reaction band at 520 ± 5 nm. The method meets CLIA-waived classification under US FDA 21 CFR §866.2900 for COHb screening devices. All lot-specific calibration data are provided with each shipment and conform to ISO/IEC 17025-accredited verification protocols. Device labeling includes multilingual instructions aligned with WHO Good Distribution Practice (GDP) and EU IVDR Annex I essential requirements.
Software & Data Management
This is a non-digital, analog detection system: no embedded firmware, connectivity, or software is present. Quantitative interpretation is performed manually using the included reference color chart and linear conversion table (µg/dL ↔ %COHb). For laboratory integration, results may be transcribed into LIS/HIS platforms via manual entry or barcode-scanned batch logs (tube lot number + expiration date encoded in GS1-128). Audit trails for calibration verification, operator training records, and environmental storage logs must be maintained per GLP requirements — no electronic audit trail is generated by the device itself.
Applications
- Rapid triage of CO poisoning in pre-hospital and ED settings (per AHA/ACLS guidelines)
- Occupational health surveillance for workers in boiler rooms, garages, steel mills, and wastewater treatment facilities
- Forensic toxicology screening during autopsy specimen analysis
- Validation of co-oximeter readings in resource-limited laboratories lacking spectrophotometric capability
- Educational demonstration of gas–blood partition kinetics and metal–ligand coordination chemistry in biomedical engineering curricula
FAQ
Can this tube detect hydrogen cyanide (HCN)?
No. The 290CO system is analytically specific for carbon monoxide. HCN detection requires separate chromogenic reagents (e.g., pyridine-barbituric acid), and cross-reactivity is negligible per independent validation studies (Meiko Rikka Technical Report TR-290CO-2023-04).
Is venous or arterial blood preferred?
Both yield equivalent COHb values; venous sampling is recommended for safety and ease of collection.
How is accuracy verified before use?
Each tube lot undergoes third-party verification against gravimetrically prepared CO-spiked blood standards (±3% bias limit); certificate of analysis is supplied with every box.
Does hematocrit affect the result?
No. The assay measures total CO-bound heme moieties per volume of whole blood; hematocrit correction is unnecessary.
What is the maximum allowable delay between sampling and analysis?
Samples must be analyzed within 30 minutes of collection when stored at ambient temperature (15–25 °C); refrigeration extends stability to 90 minutes.