Persee GMP Data Integrity Compliance Solution for Pharmaceutical Manufacturers
| Brand | Persee |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Regional Classification | Domestic (China) |
| Model | Persee GMP Data Integrity Compliance Solution |
| Pricing | Available Upon Request |
Overview
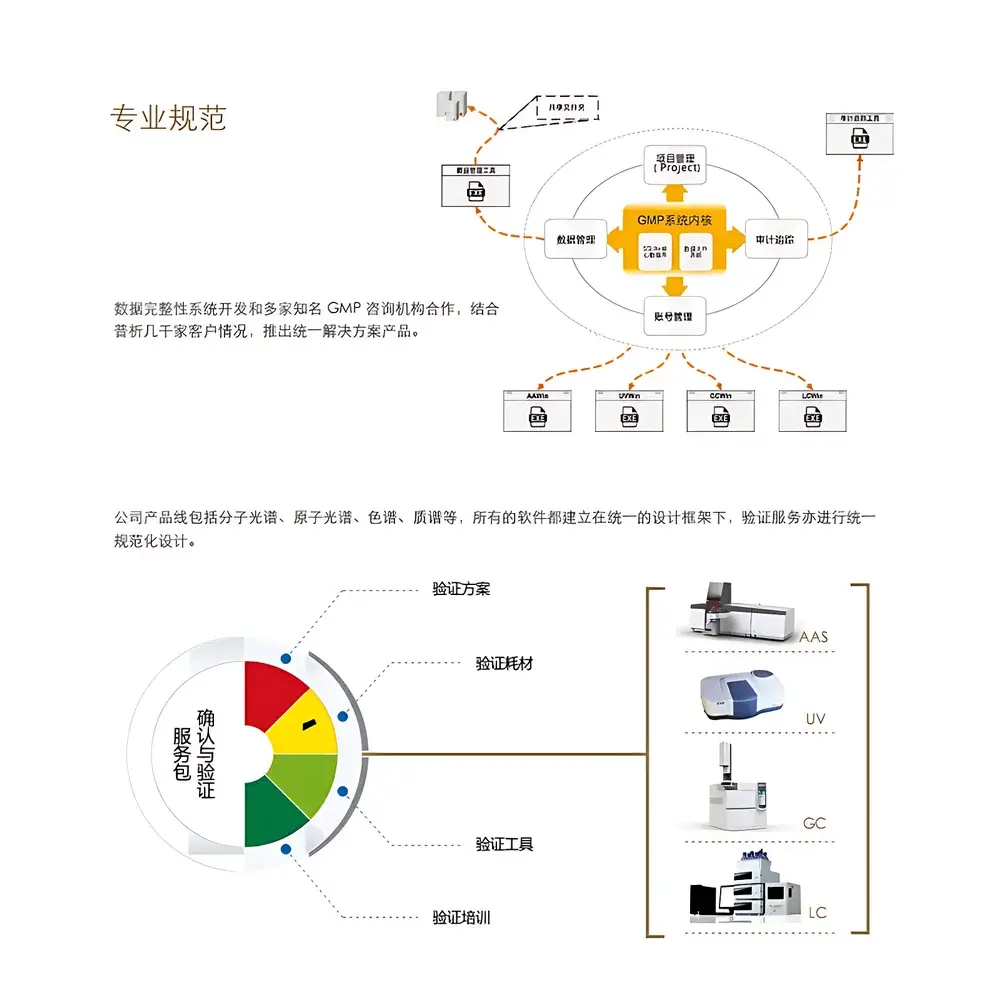
The Persee GMP Data Integrity Compliance Solution is a purpose-built, integrated software and service framework engineered to support pharmaceutical manufacturers in achieving and sustaining compliance with global Good Manufacturing Practice (GMP) regulations—specifically Annex 11 (Computerized Systems) and Annex 15 (Qualification and Validation) of the EU GMP Guidelines, as well as FDA 21 CFR Part 11 and ICH GCP/ICH Q5A/Q7 requirements. Unlike standalone validation tools or generic IT backup systems, this solution embeds data integrity principles directly into the operational lifecycle of analytical instrumentation—including UV-Vis spectrophotometers, atomic absorption spectrometers, gas chromatographs (GC), and liquid chromatographs (HPLC/UHPLC). It implements ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) through a validated architecture that governs data generation, handling, retention, and retrieval across heterogeneous instrument platforms.
Key Features
- Five-Cornerstone Compliance Architecture: Fully implemented access control (role-based user authentication), granular permission management (operator, supervisor, QA, admin tiers), immutable audit trail (time-stamped, user-identified, action-specific event logging), electronic signature capability compliant with 21 CFR Part 11 §11.200, and automated, version-controlled backup & recovery workflows.
- Cross-Platform Instrument Integration: Native drivers and protocol adapters for Persee’s full portfolio of regulated analytical instruments—ensuring metadata capture, raw data preservation, and method integrity across UV, AAS, GC, and LC platforms without third-party middleware.
- Validation-Ready Design: Delivered with pre-qualified IQ/OQ documentation templates, risk assessment matrices (aligned with ISO 14971), and traceability matrices linking functional requirements to test cases—reducing qualification effort by up to 60% compared to custom-developed systems.
- Automated Backup Orchestration: Supports concurrent replication to three independent storage targets (local NAS, on-premises server, cloud-based object storage); performs SHA-256 checksum validation post-backup; executes weekly full-archive packaging with timestamped encryption and retention tagging per defined data retention policies.
- Zero-IT-Overhead Deployment: Browser-based administration console with wizard-driven configuration; one-click project initialization; automated backup scheduling; and intuitive dashboard visualization of system health, audit trail volume, and backup success/failure status.
Sample Compatibility & Compliance
The solution does not process physical samples but governs digital artifacts generated during analytical testing—including chromatograms, spectral scans, calibration curves, integration reports, and raw detector outputs. It ensures all such data objects retain their original format (e.g., .D, .RAW, .CSV, .PDF/A-2b), embedded metadata (instrument ID, operator ID, timestamp, method version), and contextual annotations. The architecture conforms to ISO/IEC 17025:2017 clause 7.5.2 (Control of Records), supports GLP/GMP audit readiness via exportable audit trail packages, and enables full traceability from final report back to raw acquisition files—meeting inspection expectations from EMA, FDA, PMDA, and NMPA regulatory auditors.
Software & Data Management
Deployed as an on-premises Windows Server application (Windows Server 2016–2022, SQL Server 2019 Standard or later), the software includes built-in validation utilities for database integrity checks, cryptographic hash verification, and electronic signature certificate lifecycle management. All audit trails are stored in a write-once-read-many (WORM)-enabled relational schema with temporal tables for point-in-time reconstruction. System-generated logs comply with FDA 21 CFR Part 11 Subpart B requirements for record retention (minimum 2 years beyond product expiry), and support automated redaction workflows for privacy-sensitive fields under GDPR and China’s PIPL. Full export functionality enables CSV/XML-based transfer to LIMS or electronic lab notebook (ELN) systems while preserving audit trail lineage.
Applications
- Supporting routine QC release testing of APIs and finished dosage forms in accordance with USP , EP 2.6.12, and ChP 9101
- Enabling computerized system validation (CSV) for chromatographic data systems in multi-product facilities
- Facilitating internal and external audits by providing consolidated, searchable audit trail exports with filterable parameters (user, date range, instrument, event type)
- Meeting data retention obligations under FDA Guidance for Industry: “Data Integrity and Compliance With Drug CGMP” (2018) and MHRA GXP Data Integrity Definitions and Guidance (2023)
- Streamlining periodic review of data integrity controls during quality system assessments (e.g., CAPA initiation, change control evaluation)
FAQ
Does this solution require replacement of existing Persee instruments?
No. It integrates with current-generation Persee UV, AAS, GC, and LC systems via firmware-compatible communication protocols (e.g., TCP/IP, RS-232 with ASCII command sets) and requires no hardware modification.
Can audit trails be exported in a regulator-inspection-ready format?
Yes. Audit trail reports are exportable as PDF/A-2b or encrypted ZIP archives containing XML metadata and human-readable summary logs, fully compliant with EMA Annex 11 §4.2 and FDA Part 11 §11.10(d).
Is the electronic signature implementation certified for legal enforceability?
The signature module meets technical requirements of 21 CFR Part 11 §11.200 and EN 319 132-1:2016 for advanced electronic signatures (AdES), including identity binding, non-repudiation, and time-stamping via integrated NTP-synchronized clocks.
How is system uptime and continuity ensured during maintenance or failure events?
The solution supports high-availability clustering (optional), automatic failover to standby nodes, and rollback to last-known-good configuration snapshots—ensuring continuous logging and backup operations without interruption.
What validation support is provided for initial implementation?
Persee provides IQ/OQ documentation kits, UAT scripts, traceability matrices, and on-site validation engineering support—including execution of protocol testing, deviation documentation, and final summary report drafting aligned with client quality standards.