QKTester OGT-01 Headspace Oxygen & CO₂ Analyzer for Cheese and Modified Atmosphere Packaging
| Brand | QKTester |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Direct Manufacturer |
| Origin Category | Domestic (China) |
| Model | OGT-01 |
| Price | USD 145 (FOB) |
| Measured Gases | O₂, CO₂ |
| Measurement Principle | Electrochemical (O₂), Non-Dispersive Infrared (NDIR) Absorption (CO₂) |
| Sample Volume | O₂ < 3 mL |
| Resolution | 0.01% (v/v) |
| Accuracy | ±0.2% (v/v) |
| O₂ Range | 0.2–21.0% (v/v) |
| Standard Sampling Volume | ≥5 mL at 101.3 kPa |
| Dimensions | 350 × 330 × 200 mm (L×W×H) |
| Power Supply | 220 VAC 50 Hz / 120 VAC 60 Hz |
| Net Weight | 5.5 kg |
Overview
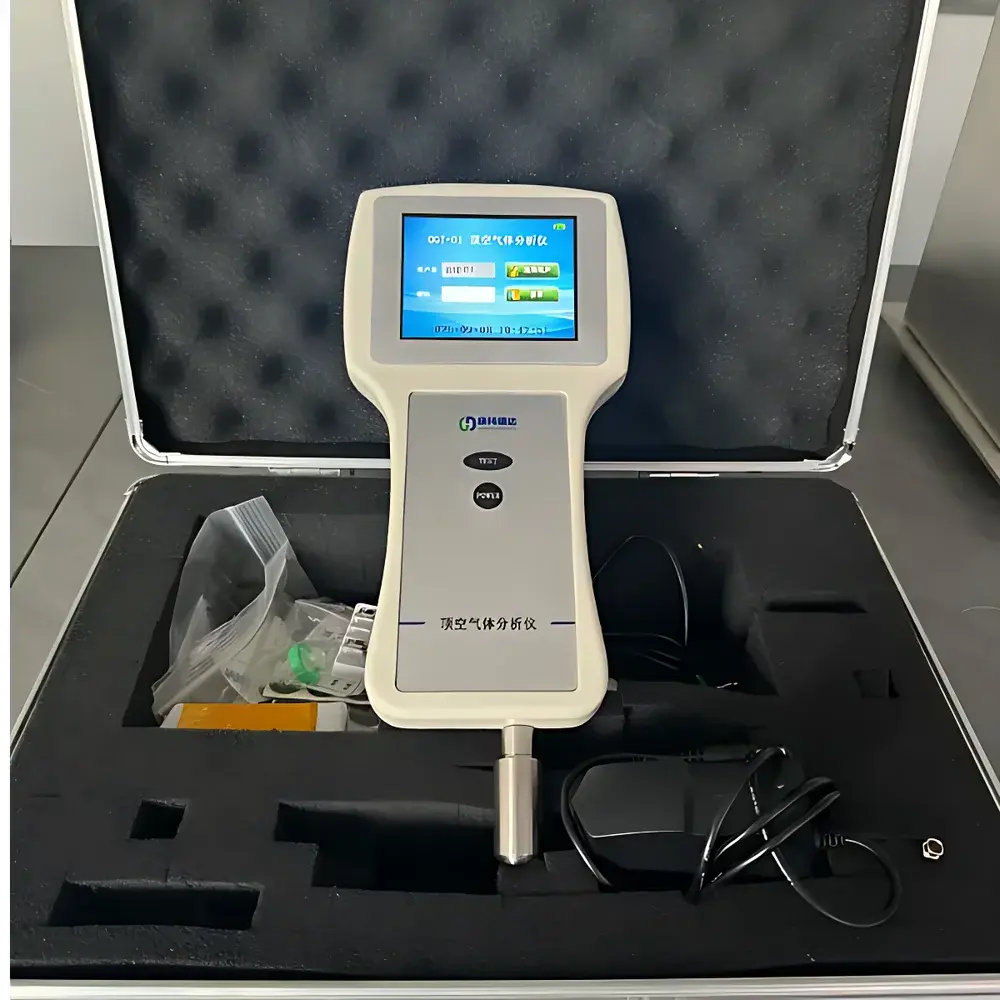
The QKTester OGT-01 Headspace Oxygen & CO₂ Analyzer is an engineered solution for quantitative gas composition analysis in the headspace of sealed non-vacuum packaging—specifically designed for quality assurance in modified atmosphere packaging (MAP) applications. It employs dual-sensor technology: an electrochemical cell for precise O₂ quantification and a non-dispersive infrared (NDIR) detector for CO₂ measurement. This principle-based architecture ensures traceable, repeatable results without requiring carrier gases or calibration gases during routine operation. The instrument operates on the principle of partial pressure-driven diffusion into the electrochemical sensor and Beer–Lambert law-based absorption for CO₂, delivering direct volumetric percentage readings under ambient pressure conditions. Its primary functional scope includes real-time verification of residual oxygen levels post-packaging, enabling manufacturers to validate seal integrity, monitor MAP process stability, and meet shelf-life prediction requirements across dairy, pharmaceutical, and ready-to-eat food sectors.
Key Features
- Dual-gas capability: Simultaneous or sequential O₂ and CO₂ analysis using industry-standard electrochemical and NDIR sensing technologies
- High-resolution readout: 0.01% (v/v) resolution across full O₂ range (0.2–21.0%), supporting compliance with ISO 11607-2 and ASTM F2622 for package integrity validation
- Low sample volume requirement: As little as 2.8 mL for O₂ measurement—ideal for small-format cheese pouches, stick packs, and blister trays where headspace volume is constrained
- Integrated color touchscreen interface: 5-inch capacitive display with intuitive navigation, English/Chinese bilingual firmware, and context-sensitive help prompts
- Onboard data management: Internal memory stores up to 10,000 test records with timestamp, operator ID, sample ID, and environmental pressure compensation metadata
- Robust mechanical design: Aluminum alloy chassis with ESD-safe housing, IP20-rated enclosure suitable for production floor, warehouse, and QC lab environments
Sample Compatibility & Compliance
The OGT-01 is validated for use with rigid and flexible non-negative-pressure containers—including aluminum-laminated pouches, PET trays, tinplate cans, Tetra Pak® cartons, glass jars, and HDPE tubs—commonly used in cheese ripening, coffee degassing, infant formula storage, and pharmaceutical blister packaging. It complies with key regulatory expectations for analytical instrumentation in GMP-adjacent environments: audit-trail-ready data logging (user-accessible via USB export), password-protected method editing, and configurable auto-zero cycles per ISO 17025 clause 7.7. While not certified to IEC 61000-4 electromagnetic immunity standards, its analog signal conditioning circuitry meets EN 61326-1 Class B emission limits for laboratory equipment. All sensor calibrations are traceable to NIST-certified gas standards (e.g., 1.00% O₂ in N₂, 10.0% CO₂ in air), and factory calibration certificates include uncertainty budgets per ISO/IEC 17025 Annex A.
Software & Data Management
Data acquisition and reporting are managed through embedded firmware v3.2.1, supporting CSV export via USB-A port (no proprietary software required). Each record contains: date/time stamp (RTC-backed), atmospheric pressure (measured internally), temperature-compensated sensor output, operator code, and pass/fail flag based on user-defined upper specification limit (USL). Optional PC-based QKDataLink™ v2.4 (Windows/macOS) enables batch report generation compliant with FDA 21 CFR Part 11 requirements—including electronic signatures, role-based access control, and immutable audit logs. Raw sensor voltage outputs and diagnostic flags (e.g., low battery, sensor saturation, flow error) are retained for root-cause analysis during CAPA investigations.
Applications
- Cheese packaging QA: Monitoring residual O₂ in vacuum-sealed or nitrogen-flushed cheddar, mozzarella, and brie packaging to prevent lipid oxidation and mold proliferation
- Pharmaceutical primary packaging: Verifying O₂ levels in desiccant-containing blister packs for hygroscopic APIs sensitive to oxidative degradation
- Instant beverage powders: Ensuring ≤0.5% O₂ in nitrogen-purged sachets of matcha, whey protein, or soluble coffee to preserve flavor volatiles and prevent Maillard browning
- Ready-to-eat meals: Validating CO₂/O₂ ratios in MAP trays for chilled prepared foods where microbial inhibition relies on precise gas balance
- Process validation: Supporting IQ/OQ documentation for MAP line qualification per ISO 22000 Clause 8.2.3 and EU Annex 15
FAQ
What gas standards are required for field calibration?
A single-point calibration using certified 1.00% O₂ in N₂ (±0.02%) and/or 10.0% CO₂ in air (±0.1%) is sufficient for daily verification. Full two-point calibration is recommended quarterly.
Can the OGT-01 measure gases other than O₂ and CO₂?
No. The hardware architecture supports only O₂ (electrochemical) and CO₂ (NDIR) detection. Other gases (e.g., N₂, Ar) are inferred by difference and not directly measured.
Is pressure compensation automatic?
Yes. An integrated absolute pressure transducer (0–120 kPa range, ±0.5 kPa accuracy) adjusts O₂ partial pressure calculations in real time for altitude or barometric variation.
How often must the O₂ sensor be replaced?
Typical service life is 24 months under normal use (≤10 measurements/day). Sensor replacement requires recalibration and is documented in the maintenance log.
Does the instrument support GLP-compliant reporting?
Yes—when used with QKDataLink™ v2.4 and configured with electronic signature enforcement, it satisfies ALCOA+ data integrity principles for regulated environments.