Rainin E4 XLS Adjustable Spacing Electronic Multichannel Pipette
| Brand | Rainin |
|---|---|
| Origin | USA |
| Model | E4 XLS |
| Instrument Type | Multichannel Pipette |
| Automation Level | Electronic Pipette |
| Channel Count | 6 or 8 |
| Volume Range | 5–1200 µL |
| Maximum Capacity | 1200 µL |
| Accuracy | ±0.8% at 1.2 µL and 9.6 µL |
| Compliance | GLP/GMP-ready, password-protected settings and service alerts, tamper-proof usage logs |
Overview
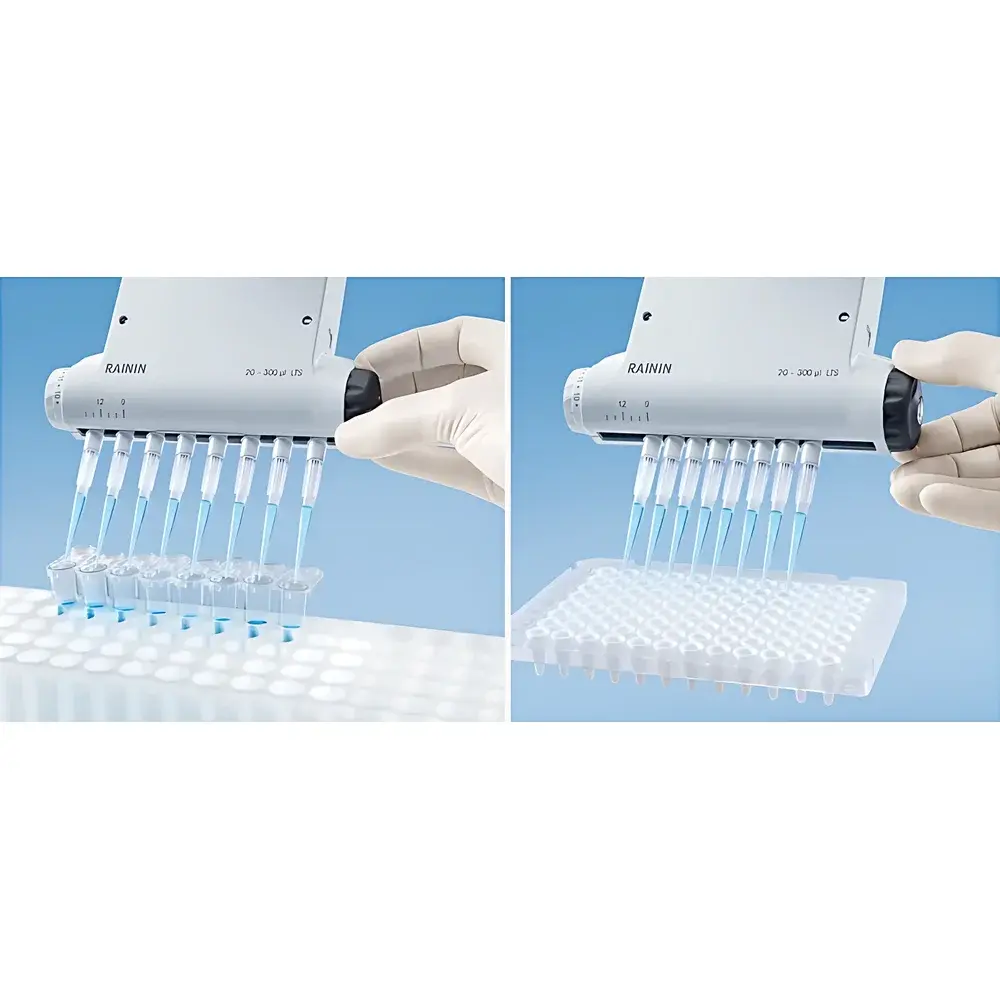
The Rainin E4 XLS Adjustable Spacing Electronic Multichannel Pipette is an engineered solution for high-throughput, precision liquid handling in regulated and research-intensive laboratory environments. Built on a microprocessor-controlled electromechanical architecture, the E4 XLS employs closed-loop stepper motor actuation (4,000-step resolution) to drive piston displacement with deterministic repeatability across its full volume range (5–1200 µL). Unlike manual or basic electronic pipettes, the E4 XLS integrates real-time positional feedback and automatic zero-point recalibration after each aspiration/dispense cycle—ensuring consistent plunger travel distance and velocity regardless of ambient temperature drift, operator fatigue, or repeated use. Its core design principle centers on spatial adaptability: the continuously adjustable inter-channel spacing enables seamless transition between standard microplate formats (e.g., 96-well, 24-well, 384-well), tube racks, gel combs, and custom labware—without requiring hardware reconfiguration or accessory swaps.
Key Features
- Continuously adjustable channel spacing: 9–14 mm for 8-channel configuration; 9–19 mm for 6-channel configuration—optimized for cross-format transfers (e.g., 96-well aspiration → 24-well or centrifuge tube dispensing)
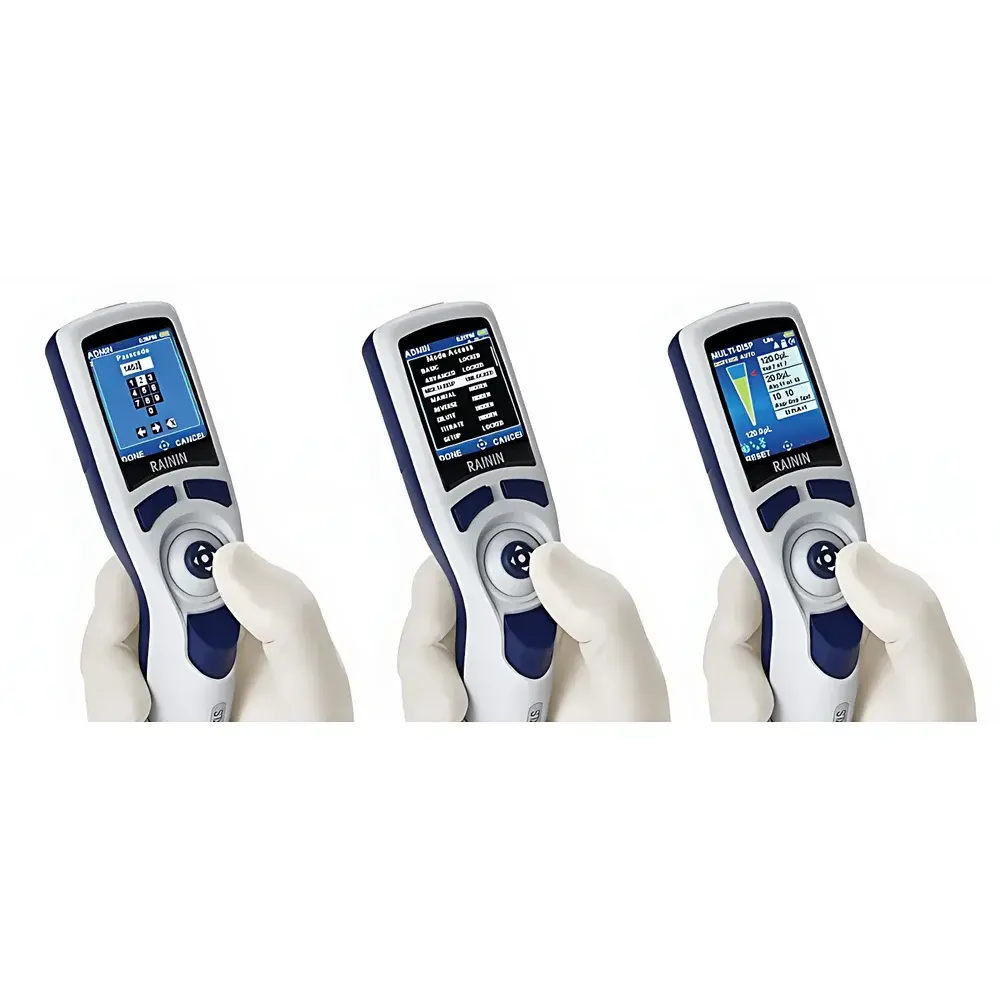
- Ergonomic lever-actuated control interface with tactile feedback for intuitive parameter selection (volume, speed, mode, repetitions)
- Onboard memory stores multiple user-defined protocols—including continuous dispensing (AutoPace), serial dilution, multi-step mixing, and repeat dispensing—with instant recall via joystick navigation
- Password-protected configuration management supports role-based access control: operators may execute preloaded methods, while supervisors configure, lock, or delete programs per SOP requirements
- Tamper-proof audit trail records all critical events—including calibration status, total pipetting cycles, error logs, and service alerts—with immutable timestamps aligned to system clock
- LTS (Low Retention Tip System) compatibility ensures secure, low-force multichannel tip attachment and ejection, minimizing tip drop-out and reducing variability in high-density workflows
Sample Compatibility & Compliance
The E4 XLS accommodates standard ANSI/SLAS-compliant tips (including filtered and low-binding variants) and interfaces reliably with common labware geometries—including polypropylene and polystyrene plates, PCR strips, conical tubes (1.5–15 mL), and electrophoresis comb assemblies. It meets essential regulatory expectations for instrument qualification in quality-controlled environments: full traceability of operational parameters, configurable lockout of unauthorized modifications, and non-erasable usage history satisfy core requirements of FDA 21 CFR Part 11 (electronic records/signatures), ISO/IEC 17025 (testing laboratories), and EU GMP Annex 11 (computerized systems). While not certified as “GMP-compliant” out-of-box, the device is fully configurable to support GLP/GMP validation protocols—including IQ/OQ/PQ documentation templates and calibration certificate integration.
Software & Data Management
No external PC or proprietary software is required for routine operation—the E4 XLS functions autonomously with embedded firmware supporting full protocol execution, parameter logging, and diagnostics. All user-defined programs reside in non-volatile flash memory and persist through power cycles. For labs implementing centralized asset management, Rainin’s Liquidator Connect platform (optional) enables remote firmware updates, fleet-wide configuration synchronization, and export of usage reports in CSV format—compatible with LIMS and ELN systems. Audit data exports include ISO 8601-formatted timestamps, operator IDs (if integrated with network authentication), and cryptographic hash verification for integrity assurance.
Applications
The E4 XLS is routinely deployed in applications demanding both spatial flexibility and metrological rigor: genomic library preparation (e.g., tagmentation, PCR setup across variable well densities), cell culture passaging into multi-well differentiation plates, ELISA assay development requiring serial dilutions across heterogeneous plate layouts, and QC testing of biologics where inter-lot consistency depends on identical dispensing kinetics. Its ability to maintain constant aspiration/dispense velocity—regardless of volume or tip resistance—reduces shear-induced degradation of sensitive biomolecules (e.g., mRNA, protein complexes) during transfer. The AutoPace function further mitigates repetitive strain injury (RSI) risk in long-duration screening campaigns by eliminating manual trigger repetition.
FAQ
Does the E4 XLS require annual calibration certification?
Yes—while the device performs self-recalibration at zero point post-cycle, formal metrological verification against gravimetric or photometric standards is required per ISO 8655 and institutional SOPs. Rainin-certified service technicians provide documented calibration certificates traceable to NIST standards.
Can spacing be adjusted mid-operation without interrupting workflow?
Yes—continuous mechanical adjustment is possible during active use via the ergonomic side-mounted dial; no power cycling or menu navigation is needed.
Is LTS tip compatibility mandatory, or can universal tips be used?
LTS tips are strongly recommended for optimal accuracy and reproducibility; universal tips may cause inconsistent seal formation and increased carryover, particularly below 50 µL.
How many protocols can be stored onboard?
Up to 20 fully parameterized methods—including volume, speed profile, step count, pause intervals, and dispensing pattern—can be saved and recalled without external storage.
What happens if the battery depletes during a multi-step protocol?
The E4 XLS retains all active program states in non-volatile memory; upon recharge, it resumes from the last completed step—not from the beginning—preserving experimental integrity.