RAININ QuickFlow 4L Vacuum Liquid Aspiration System
| Brand | RAININ |
|---|---|
| Origin | Jiangsu, China |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Domestic (China) |
| Model | QuickFlow 4L |
| Max. Aspiration Rate | 17 mL/s |
| Noise Level | <60 dB(A) |
| Collection Bottle Capacity | 4 L |
| Sterilization Compatibility | Autoclavable (121°C, 20 min) |
| Compliance | Designed for GLP-compliant labs |
Overview
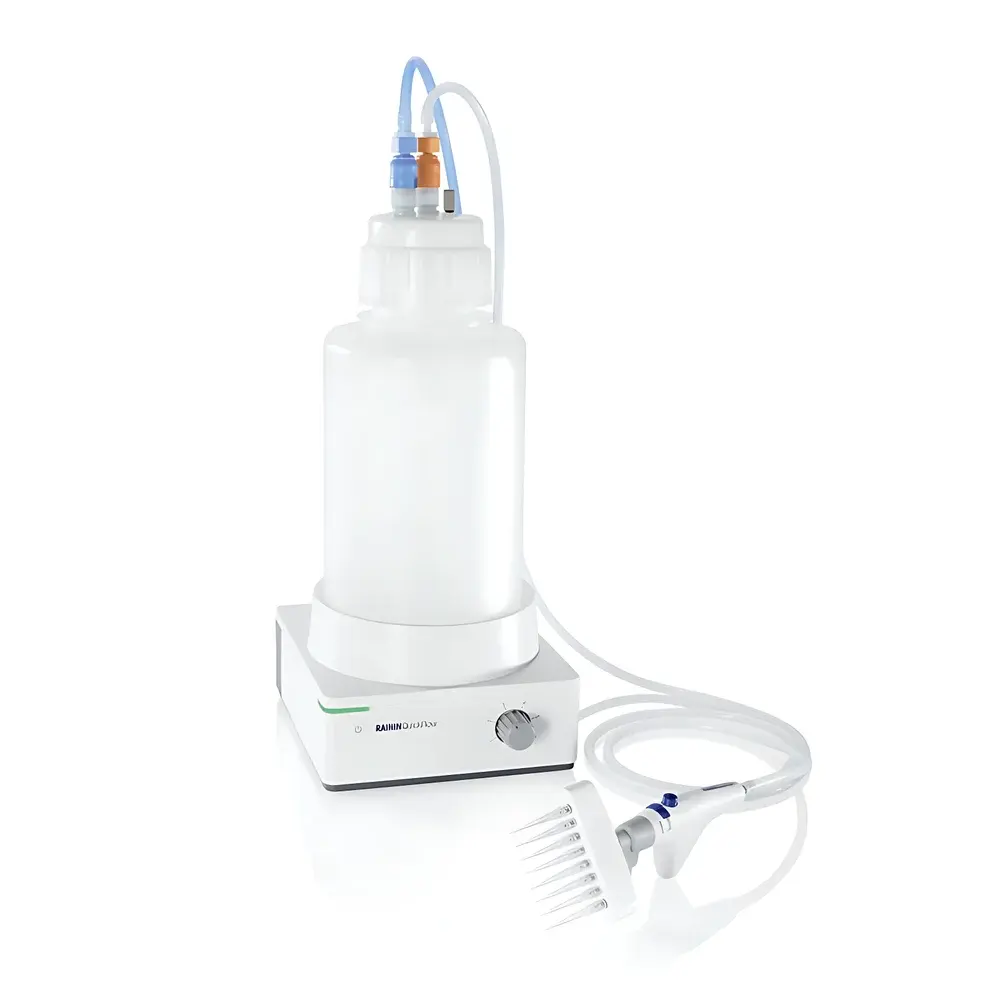
The RAININ QuickFlow 4L Vacuum Liquid Aspiration System is an engineered solution for precise, ergonomic, and contamination-controlled liquid removal in life science, clinical diagnostics, and pharmaceutical laboratories. Operating on a regulated vacuum principle, the system utilizes a diaphragm pump to generate consistent negative pressure—enabling controlled aspiration of aqueous, viscous, or mildly corrosive media without risk of pump fluid back-contamination. Its core architecture integrates safety-critical hardware—including non-contact optical liquid level detection, dual-stage filtration, and self-sealing quick-connect interfaces—to ensure operational integrity across high-throughput workflows such as cell culture maintenance, ELISA plate processing, and buffer exchange protocols. Designed for seamless integration into biosafety cabinets, laminar flow hoods, and modular lab benches, the QuickFlow 4L meets functional requirements for ISO/IEC 17025-accredited testing environments where traceability, repeatability, and operator safety are foundational.
Key Features
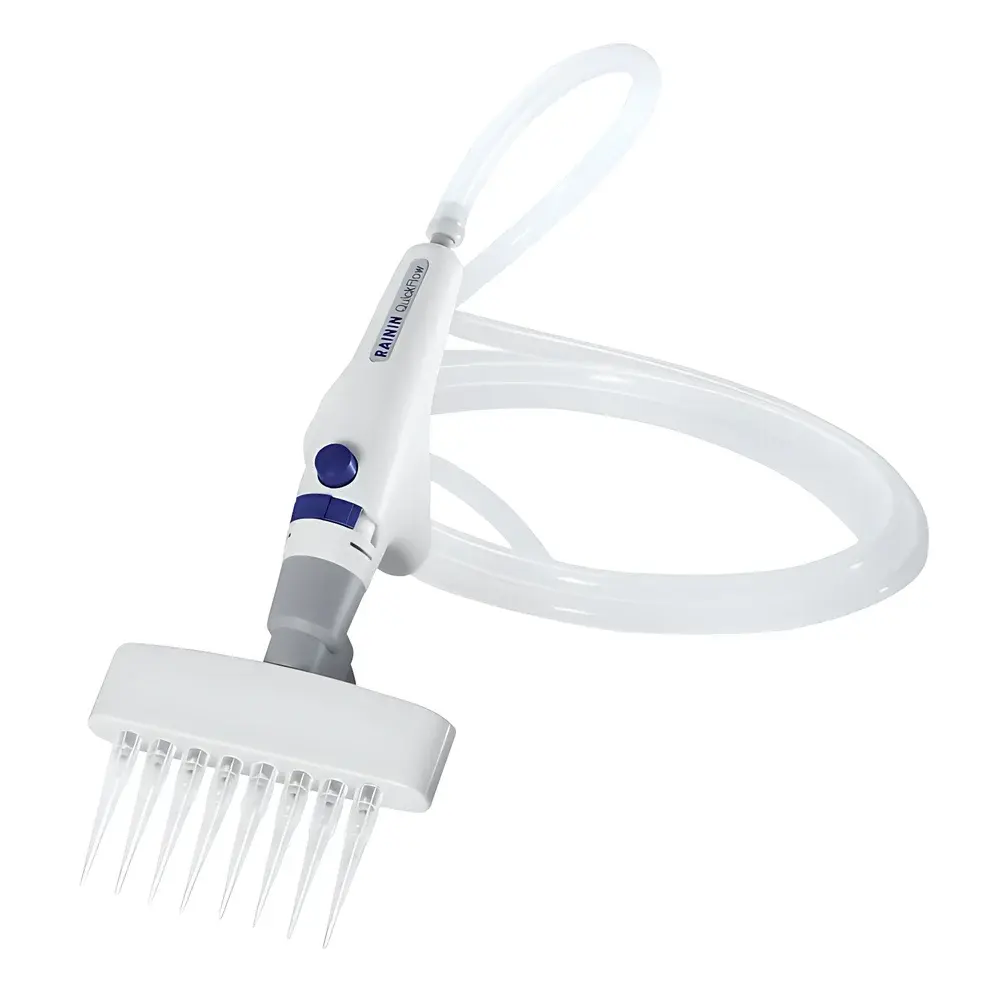
- Ergonomic dual-mode handpiece: Switches instantly between manual trigger actuation and continuous aspiration mode—reducing repetitive strain injury (RSI) risk during prolonged use.
- Non-contact optical liquid level sensor: Mounted externally on the collection bottle, it triggers audible and visual alarms plus automatic pump shutoff when liquid reaches 90% capacity—eliminating overflow and cross-contamination events.
- Self-sealing quick-connect interface: Patented spring-loaded coupling seals the bottle port upon disconnection, preventing aerosol release and maintaining containment integrity during bottle changes.
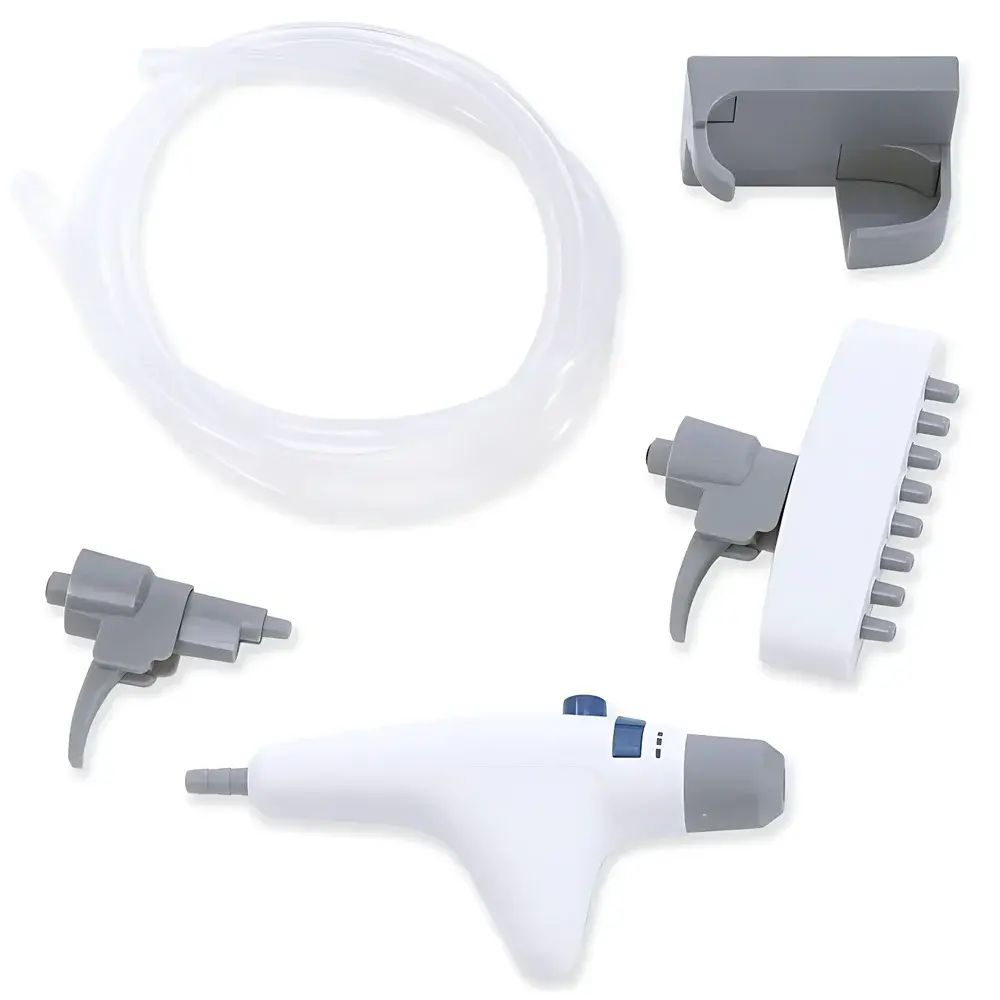
- Triple-adapter compatibility: Includes single-channel, 8-channel (for 96-well plate aspiration), and pipette-tip suction adapters—supporting flexible transition between low- and high-density sample formats.
- Variable aspiration speed control: Continuously adjustable vacuum output (0–100%) enables optimization for delicate cell monolayers, viscous media (e.g., serum-containing solutions), or rapid bulk waste removal.
- Autoclavable wetted path: All fluid-contact components—including handpiece tubing, adapters, and 4 L borosilicate glass or polypropylene collection bottle—are rated for steam sterilization at 121°C for 20 minutes, ensuring compliance with GMP cleaning validation protocols.
- Low-noise operation: Diaphragm pump design achieves <60 dB(A) sound pressure level at 1 m distance—minimizing acoustic interference in shared lab spaces and sensitive imaging suites.
Sample Compatibility & Compliance
The QuickFlow 4L is validated for aspiration of deionized water, PBS, cell culture media (with or without 10% FBS), ethanol (≤70%), and common laboratory buffers (HEPES, Tris-HCl, etc.). It is not intended for use with strong oxidizers, organic solvents exceeding Class II flammability limits (e.g., acetone, chloroform), or highly corrosive acids/bases (pH 12). The system adheres to IEC 61000-6-3 (EMC emissions) and IEC 61000-6-2 (immunity) standards. Its mechanical design supports audit readiness under FDA 21 CFR Part 11 (when paired with compliant LIMS data logging), EU Annex 11, and WHO TRS 961 Annex 6 for aseptic process support equipment. Dual hydrophobic/hydrophilic filter cartridges (0.45 µm + 0.22 µm) provide redundant barrier protection against aerosol ingress into the pump head—extending service intervals and reducing total cost of ownership.
Software & Data Management
While the QuickFlow 4L operates as a standalone hardware platform without embedded firmware or touchscreen UI, its operational parameters—including runtime, total aspirated volume (via optional external flow meter integration), and alarm event logs—can be captured via analog signal outputs (0–10 V) or dry-contact relay triggers. When deployed within validated laboratory networks, it interfaces with electronic lab notebooks (ELNs) and asset management systems through standard RS-232 or USB-to-serial adapters. Audit trail generation complies with ALCOA+ principles when linked to time-stamped, user-authenticated LIMS entries—supporting 21 CFR Part 11 Subpart B requirements for electronic records and signatures in regulated environments.
Applications
- Cell culture maintenance: Gentle aspiration of spent media from T-flasks, roller bottles, and bioreactors without disturbing adherent monolayers.
- High-throughput assay preparation: Rapid removal of supernatant from 96- and 384-well plates during ELISA, cytotoxicity, and binding assays.
- Microbiology workflows: Safe decanting of broth cultures and wash steps in sterility testing and antimicrobial susceptibility testing (AST).
- Quality control laboratories: Controlled aspiration of rinse solutions during HPLC column conditioning or dissolution testing sample handling.
- Cleanroom operations: Compact footprint and autoclavable components enable routine use inside ISO Class 5 laminar flow cabinets without compromising environmental classification.
FAQ
Is the QuickFlow 4L suitable for aspiration of infectious or biohazardous liquids?
Yes—when used with certified 0.22 µm hydrophobic filters and properly maintained collection bottles, it meets BSL-2 containment requirements per CDC/NIH Biosafety in Microbiological and Biomedical Laboratories (BMBL), 6th Edition.
Can the handpiece be sterilized using ethylene oxide (EtO)?
No—only autoclaving (121°C, 20 min, saturated steam) is validated for the handpiece and adapters. EtO may degrade elastomeric seals and compromise vacuum integrity.
What is the recommended maintenance interval for the dual-filter assembly?
Filters should be replaced after every 50 L of total aspirated volume or immediately following any visible breakthrough event—whichever occurs first.
Does the system include documentation for IQ/OQ qualification?
Yes—factory-issued Installation Qualification (IQ) templates and Operational Qualification (OQ) test protocols are provided with each unit, aligned with ASTM E2500-13 and ISO/IEC 17025:2017 Annex A.2 guidelines.
Can the aspiration speed be calibrated traceably to NIST standards?
Yes—using a certified volumetric flow meter (e.g., Brooks Instrument SLA series) and gravimetric verification per ISO 5167, users can perform in-house calibration with documented uncertainty budgets meeting ISO/IEC 17025 clause 6.5 requirements.