Regulatory Compliance Management System (RCMS) by SCYZ
No specifications available.
Overview
The Regulatory Compliance Management System (RCMS) by Sichuan Yanzhi Technology Co., Ltd. (SCYZ) is a purpose-built software solution designed to support quality assurance and regulatory adherence in accredited testing laboratories, clinical labs, and R&D facilities operating under ISO/IEC 17025, GLP, GMP, or FDA 21 CFR Part 11 frameworks. Built on a relational database architecture with role-based access control and audit-trail-enabled operations, the RCMS implements standardized document lifecycle management for regulatory texts—including national and international standards (e.g., ISO, ASTM, USP, GB/T), test methods, SOPs, and internal policies. It operates as a core module within broader laboratory information management ecosystems, integrating with LIMS, ELN, and instrument data acquisition layers via standardized APIs (e.g., RESTful endpoints). The system enforces version integrity through immutable revision logging, timestamped approvals, and enforced change control workflows—ensuring that every document in use is demonstrably current, authorized, and traceable to its origin and approval chain.
Key Features
- Structured Regulatory Repository: Hierarchical taxonomy-driven storage for regulations, standards (national, regional, industry-specific), validated test methods, and internal SOPs—with metadata tagging for jurisdiction, scope, applicability, and effective date.
- Automated Regulatory Intelligence Feed: Configurable periodic checks against authoritative sources (e.g., CNAS announcements, ISO updates, NIST bulletins, SAMR releases) with configurable alert thresholds and escalation paths for pending revisions.
- Version Control & Audit Trail: Full revision history including author, approver, effective date, superseded version reference, and rationale for change—compliant with ISO 17025:2017 Clause 8.3 and FDA 21 CFR Part 11 Subpart B requirements for electronic records.
- Change Control Workflow Engine: Multi-stage review/approval routing with mandatory sign-offs, parallel review options, and automatic notification upon status transition (e.g., “Under Review”, “Approved”, “Obsolete”).
- Compliance Self-Assessment Module: Predefined checklists aligned with ISO/IEC 17025, GLP Principles, or internal QA protocols; generates auditable gap reports with assignable corrective actions and deadline tracking.
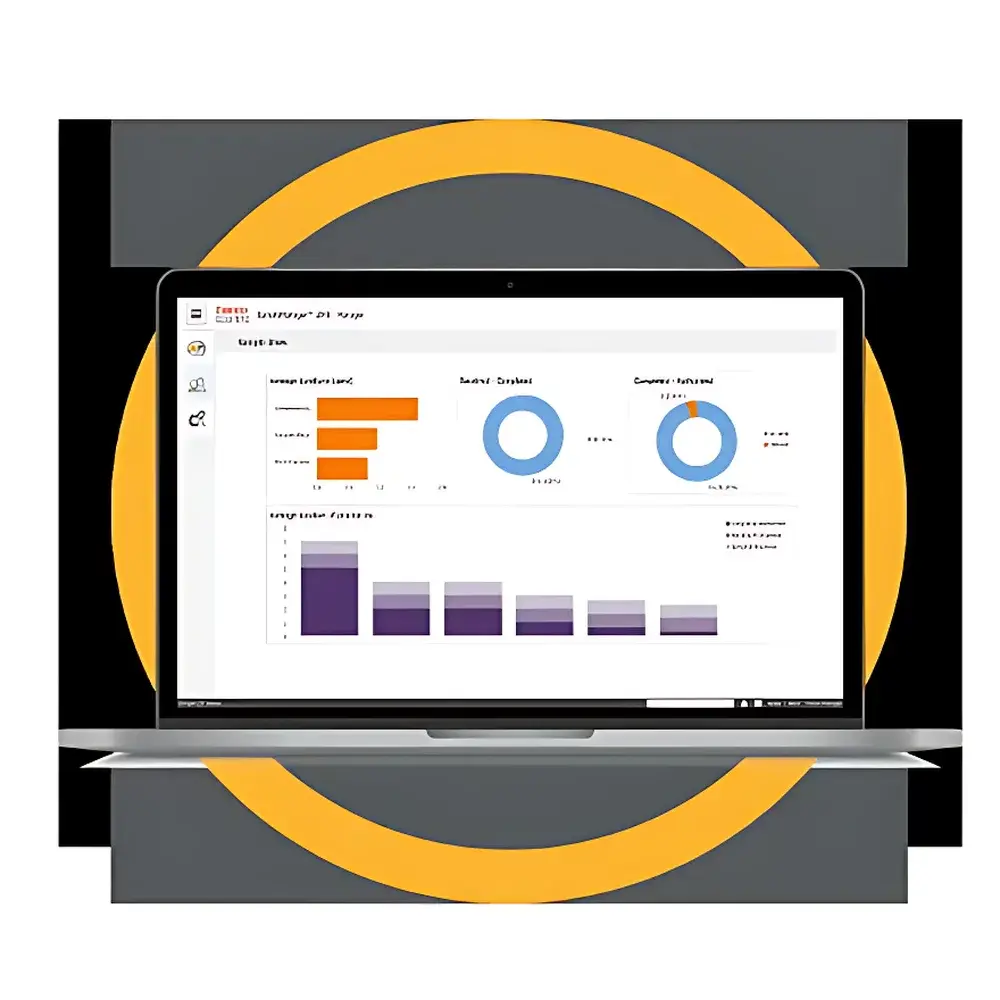
- Document Status Dashboard: Real-time visualization of document health metrics—including % of standards overdue for review, % of active documents lacking latest revision, and average time-to-approval per workflow type.
Sample Compatibility & Compliance
The RCMS does not process physical samples or analytical data streams; rather, it governs the digital artifacts that define analytical validity. It supports ingestion of PDF, DOCX, XLSX, and XML-formatted regulatory documents—including machine-readable standards (e.g., ISO/IEC 17025:2017 Annex A XML schemas). All document uploads undergo checksum validation and virus scanning prior to ingestion. The system conforms to ISO/IEC 27001:2022 controls for information security, implements TLS 1.2+ encryption for data in transit, and supports optional FIPS 140-2 validated cryptographic modules for environments requiring federal-grade data protection. It satisfies documentation control requirements outlined in ISO/IEC 17025:2017 Sections 8.3 and 8.5, USP <1058> Analytical Instrument Qualification, and EU Annex 11 for computerized systems used in regulated environments.
Software & Data Management
Deployed as an on-premise or private-cloud application, the RCMS uses PostgreSQL v14+ with row-level security policies and granular permission sets (viewer, editor, approver, administrator). All user actions—including document upload, modification, approval, and deletion—are captured in an immutable audit log compliant with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). Export functionality supports CSV, PDF, and XML formats for external reporting or regulatory submission. Integration with enterprise identity providers (e.g., Active Directory, LDAP, SAML 2.0) enables centralized authentication. Backup policies adhere to ISO 22301 business continuity standards, with daily incremental backups retained for 90 days and quarterly full backups archived offline for 7 years.
Applications
- Accredited testing laboratories maintaining ISO/IEC 17025 compliance across chemical, environmental, food, and pharmaceutical domains.
- Pharmaceutical QC labs implementing ICH Q5A–Q5E and USP <1223> validation requirements for compendial methods.
- Contract research organizations (CROs) managing multi-client regulatory dossiers with client-specific access segmentation.
- National metrology institutes and reference material producers requiring traceable method documentation for calibration hierarchies.
- University core facilities supporting grant-funded research subject to NIH or NSF data management plan (DMP) stipulations.
FAQ
Does the RCMS support multilingual regulatory documents?
Yes—metadata fields (title, description, scope) are Unicode-compliant and support UTF-8 encoding. Document content remains in original language; translation management is handled externally, though version linkage between original and translated documents is supported.
Can the system integrate with our existing LIMS or ELN platform?
Yes—via documented REST APIs and configurable webhooks. Pre-built connectors are available for Thermo Fisher SampleManager LIMS, LabVantage, and IDBS E-WorkBook; custom integrations follow HL7 FHIR or ASTM E1467 standards.
How does the RCMS handle obsolete or withdrawn standards?
Withdrawn documents are flagged with “Obsoleted” status, removed from active search results by default, and retained in read-only archive mode with clear linkage to their replacement standard (if applicable). Access requires explicit administrative override.
Is electronic signature capability included?
Yes—digital signatures comply with eIDAS Level 1 and are cryptographically bound to document hash values. Signature logs include biometric time stamps, IP address, and device fingerprinting for forensic traceability.
What training and validation support is provided?
SCYZ delivers IQ/OQ documentation packages, UAT scripts, and 3-day administrator training. Validation assistance includes protocol development, execution oversight, and final summary report generation aligned with GAMP 5 guidelines.