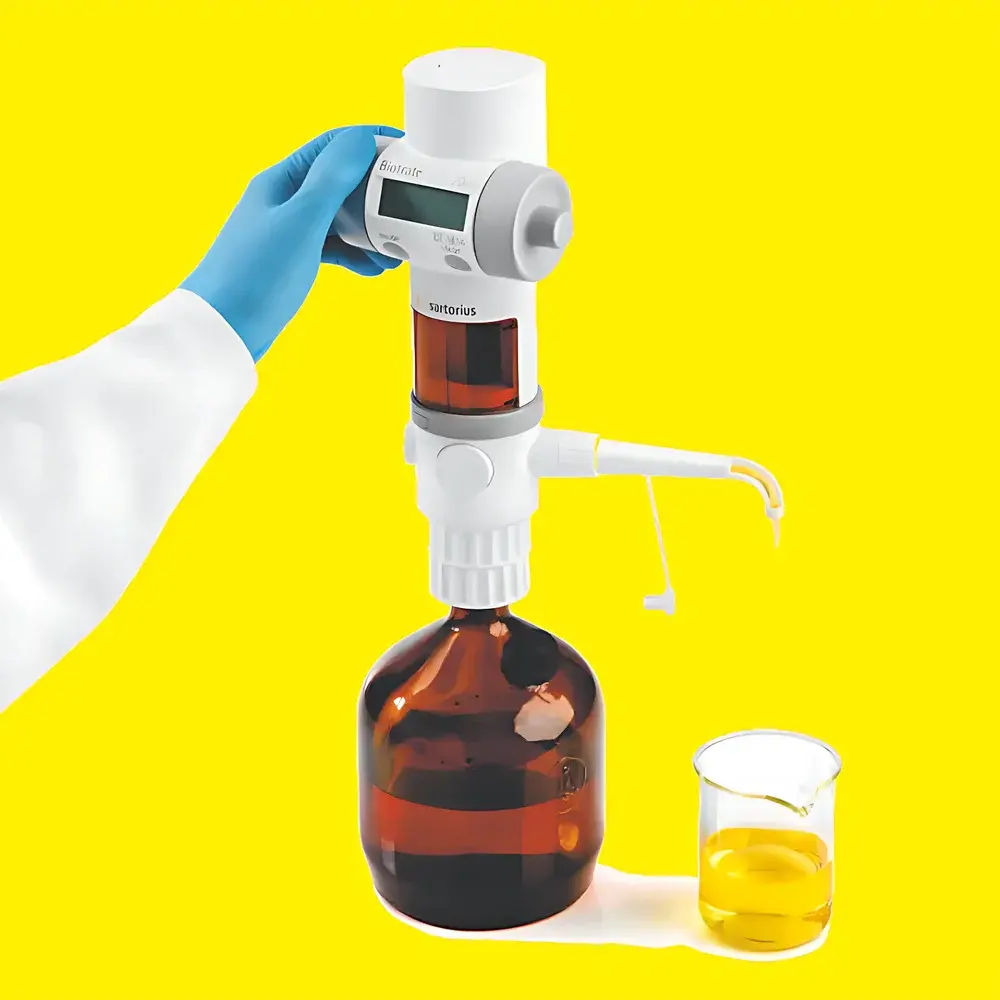
Sartorius Biotrate Premium Digital Titrator
| Brand | Sartorius |
|---|---|
| Origin | Germany |
| Model | Biotrate |
| Category | Imported Electronic Titrator |
| Compliance | CE, ISO 9001, GLP-ready |
| Display | High-contrast LCD with volume readout (aspirated/dispensed) |
| Dispensing Head | 360° rotatable Luer-lock interface |
| Fluid Path | Chemically resistant PTFE, FFKM, and sapphire components |
| Piston Drive | Precision-machined ceramic-coated piston with low-friction linear actuation |
| Power | Replaceable AA batteries (≥2,000 operations per set) |
| Sterilization | Fully autoclavable fluid path (121 °C, 20 min, steam) |
| Accessories | Minisart® NML syringe filters (0.2 µm, 0.45 µm), bottle adapters, UV-protective amber window kit, desiccant tube port |
Overview
The Sartorius Biotrate Premium Digital Titrator is an electronically controlled, precision liquid dispensing instrument engineered for volumetric titration in regulated laboratory environments. Operating on a positive-displacement principle, it utilizes a high-precision ceramic-coated piston driven by a micro-stepped motor to deliver reproducible volumes across a wide dynamic range—without reliance on air displacement or gravity-dependent flow. Its fully sealed, chemically inert fluid path—constructed from PTFE, perfluoroelastomer (FFKM), and sapphire—is validated for compatibility with aggressive solvents, strong acids, bases, and halogenated reagents. Designed in accordance with ISO 8655-7 (volumetric instruments — piston-operated instruments) and aligned with GLP documentation requirements, the Biotrate supports traceable, auditable titration workflows in QC, R&D, and compliance-driven settings including pharmaceutical, chemical, and environmental testing laboratories.
Key Features
- 360° rotatable dispensing head with Luer-lock interface enables ergonomic positioning without rotating bottles or reorienting the entire unit—critical for multi-vessel titrations and confined benchtop spaces.
- Autoclavable fluid path certified for steam sterilization at 121 °C for 20 minutes, ensuring full compatibility with sterile applications and routine decontamination protocols.
- Integrated media recirculation system returns unused titrant to the source reservoir, minimizing reagent waste and eliminating priming delays between runs.
- Dedicated Luer port on rear panel for optional desiccant tube or particulate filter connection—essential for moisture-sensitive or particulate-prone titrants (e.g., Karl Fischer reagents, silver nitrate solutions).
- UV-protective amber viewing window (replaceable) safeguards light-labile titrants such as iodine solutions, ceric ammonium nitrate, or diazonium salts from photodegradation during dispensing.
- Tool-free valve replacement allows end-user maintenance without service downtime; all wetted components are modular and field-replaceable.
- Intuitive touch-sensitive control wheel with haptic feedback provides precise volume selection and real-time status indication—including mode (aspirate/dispense), volume, battery level, and calibration state.
Sample Compatibility & Compliance
The Biotrate accommodates a broad spectrum of titration media—from aqueous standards (e.g., NaOH, HCl, EDTA) to organic solvents (acetonitrile, methanol, chloroform), oxidizing agents (KMnO₄, K₂Cr₂O₇), and corrosive reagents (HF, aqua regia). Its fluid path materials meet USP Class VI biocompatibility and FDA 21 CFR Part 11 readiness criteria when paired with Sartorius LabX software (optional). Device firmware supports electronic audit trails, user-level access control, and event logging—fully compliant with ISO/IEC 17025, ASTM E2500, and EU GMP Annex 11 expectations for analytical instrumentation.
Software & Data Management
While the Biotrate operates as a standalone instrument, it integrates seamlessly with Sartorius LabX Liquid Handling software via USB or RS-232. LabX enables method storage, electronic signature capture, automatic data export to LIMS or ELN systems, and full 21 CFR Part 11 compliance—including role-based permissions, change tracking, and immutable audit logs. All volume entries, dispense events, and error codes are timestamped and linked to operator ID. Raw data files are saved in CSV and PDF formats with embedded metadata (device ID, firmware version, calibration date).
Applications
- Standardized acid–base, redox, complexometric, and precipitation titrations per ISO 3696, ASTM D1126, and USP general chapter on volumetric analysis.
- Residual moisture determination using Karl Fischer titration—leveraging integrated desiccant port and solvent compatibility.
- Pharmaceutical assay validation where volumetric accuracy, repeatability (CV ≤ 0.3% at ≥1 mL), and documentation integrity are mandated.
- Environmental water analysis (e.g., COD, chloride, hardness) requiring robust handling of turbid or particulate-containing samples via inline Minisart® NML filtration.
- Research-scale synthesis where precise stoichiometric reagent addition and reagent conservation are critical.
FAQ
Is the Biotrate suitable for use with hydrofluoric acid (HF)?
Yes—the fluid path incorporates sapphire valves and FFKM seals rated for continuous HF exposure up to 48 wt%, validated per ASTM D471.
Can the device be calibrated in-house?
Yes—gravimetric calibration is supported using certified weights and Class A volumetric flasks; calibration certificates can be generated and stored internally.
Does the Biotrate support variable dispensing speeds?
Yes—three programmable flow rates (slow/medium/fast) are available to optimize delivery for viscous media or low-volume endpoints.
What is the maximum recommended operating temperature for the fluid path?
The wetted components maintain dimensional stability and seal integrity up to 60 °C ambient; autoclaving is limited to 121 °C for defined cycles only.
How often does the piston require maintenance?
Under normal use (≤500 dispenses/day), the ceramic-coated piston requires no lubrication and exhibits <0.1% volume drift over 10,000 cycles—verified per ISO 8655-7 wear testing protocol.