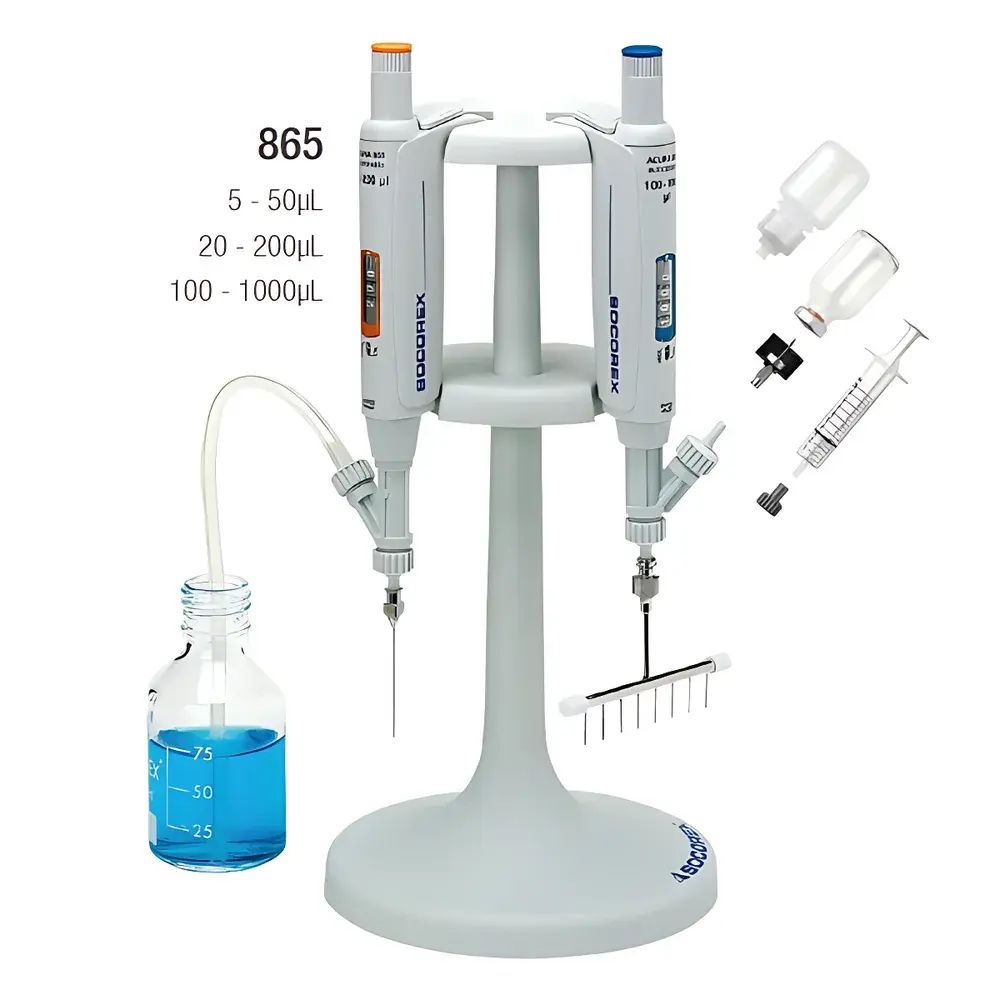
SOCOREX Acura® 865 Continuous Dispensing Pipette
| Brand | SOCOREX |
|---|---|
| Origin | Switzerland |
| Model | 865 |
| Pipette Type | Manual Single-Channel Continuous Dispenser |
| Volume Range | 5–1000 µL |
| Accuracy (E%) | ±0.6% to ±1.5% |
| Precision (CV%) | ≤0.2% to ≤0.4% |
| Sterilization | Autoclavable (121°C, 20 min) |
| Display | Digital LED Volume Readout |
| Calibration | User-Adjustable One-Touch System |
| Materials in Contact | PTFE, PP, Stainless Steel (316), Chemically Inert |
| Compliance | ISO 8655-1:2022, ISO 8655-2:2022, GLP/GMP-ready |
Overview
The SOCOREX Acura® 865 is a precision-engineered, manually operated continuous dispensing pipette designed for high-reproducibility liquid handling in regulated and research-intensive laboratory environments. Based on positive displacement principle with an integrated three-way air valve system, the Acura® 865 eliminates air cushion variability—enabling true air-free, piston-driven dispensing directly from reservoirs such as reagent bottles, syringes, or vials. Unlike traditional air-displacement pipettes, its sealed fluid path ensures consistent delivery across viscosity gradients and volatile solvents, minimizing evaporation-induced error and cross-contamination risk. The device operates within a broad, digitally adjustable volume range of 5–1000 µL, supporting both single-step and multi-step dispensing protocols without tip changes. Its modular architecture allows seamless integration with optional multi-channel adapters (4- and 8-channel stainless steel manifolds), making it suitable for serial dilution, plate filling, assay setup, and QC sample preparation workflows requiring traceable, repeatable volumetric output.
Key Features
- Digital Volume Display & Ergonomic Interface: High-contrast LED window provides real-time, unambiguous volume readout; rotary dial enables intuitive clockwise/counterclockwise adjustment with tactile feedback and fine resolution (0.1–1 µL increments depending on range).
- Air-Free Positive Displacement Mechanism: Patented three-way valve system automatically replenishes gas volume during aspiration, eliminating manual venting and ensuring stable piston travel—critical for accurate low-volume (<50 µL) dispensing.
- Full Autoclavability: Entire unit—including housing, piston assembly, and valve body—is rated for full-cycle steam sterilization (121°C, 20 min, 1 bar), supporting aseptic cell culture, molecular diagnostics, and GMP-compliant bioprocessing applications.
- User-Initiated Calibration: One-touch calibration system requires no tools or external software; users adjust internal volume offset via removable calibration rod while viewing real-time display—fully traceable and compliant with ISO 8655-6:2022 calibration verification requirements.
- Chemically Resistant Fluid Path: All wetted components are constructed from medical-grade PTFE, polypropylene, and passivated 316 stainless steel—validated for compatibility with organic solvents (e.g., DMSO, acetonitrile), strong acids/bases, and biological buffers (PBS, Tris-HCl, guanidine thiocyanate).
- Traceability & Documentation: Each unit ships with individual serial number, factory QC certificate (including gravimetric test data per ISO 8655-2), and CE/UKCA marking—meeting audit readiness requirements for ISO/IEC 17025, FDA 21 CFR Part 11 (when paired with validated electronic logbooks), and EU IVDR Annex II documentation standards.
Sample Compatibility & Compliance
The Acura® 865 accommodates diverse sample formats through standardized Luer-lock interfaces: direct connection to 30 mL PTFE or PP reagent bottles, silicone inlet tubing (90 cm), glass/plastic disposable syringes, or vaccine vials. Optional stainless steel multi-channel manifolds (4- or 8-tip) support parallel dispensing into microtiter plates without compromising accuracy. All configurations maintain compliance with ISO 8655 parts 1–6 (2022 edition) for metrological performance, mechanical durability, and calibration traceability. For GLP/GMP environments, the device supports instrument qualification protocols (IQ/OQ/PQ) and integrates with electronic lab notebooks (ELNs) for audit-trail generation when used with calibrated analytical balances meeting OIML R111 Class E2 or better specifications.
Software & Data Management
While the Acura® 865 operates independently as a standalone mechanical instrument, its calibration events and usage logs can be documented manually or imported into laboratory information management systems (LIMS) via structured CSV templates. Gravimetric calibration data—recorded using certified analytical balances (minimum readability: 0.01 mg for volumes ≤20 µL; 0.1 mg for ≥100 µL)—must be retained per ISO/IEC 17025 clause 7.7. Calibration intervals are recommended every 6 months (maximum 12 months), with records including date, operator ID, balance ID, ambient temperature/humidity, and measured deviation against reference values. SOCOREX provides downloadable calibration worksheets and traceable reference standards (NIST-traceable water density tables) via its global technical portal.
Applications
- High-throughput assay development requiring precise serial dilutions (e.g., ELISA standard curves, IC50 determination)
- Cell-based assays involving sensitive reagents (e.g., transfection mixes, growth factors, CRISPR ribonucleoproteins)
- QC testing in pharmaceutical manufacturing (e.g., dissolution media preparation, buffer formulation verification)
- Genomics workflows including NGS library normalization and qPCR master mix dispensing
- Environmental testing labs performing EPA Method-compliant reagent addition (e.g., TOC analysis, heavy metal digestion)
- Academic core facilities supporting shared instrumentation with multi-user calibration tracking and usage logging
FAQ
Is the Acura® 865 compatible with viscous or volatile liquids?
Yes—the positive displacement design and chemically inert fluid path ensure reliable performance with glycerol (up to 80% v/v), ethanol, acetone, and aqueous-organic mixtures without vapor lock or seal degradation.
Can I perform multi-channel dispensing without changing hardware?
Yes—by attaching the optional 4-channel (Order No. 1.170.054) or 8-channel (Order No. 1.170.058) stainless steel manifolds, the same Acura® 865 body supports parallel dispensing into 96-well or 384-well plates.
What is the recommended calibration frequency?
Per ISO 8655-6:2022 and internal SOP guidance, calibration verification is required at least every 6 months; however, frequency may increase based on usage intensity, criticality of application, or regulatory inspection findings.
Does the device require special maintenance beyond routine cleaning?
No—routine decontamination with 70% ethanol or mild detergent suffices; autoclaving serves as both sterilization and preventive maintenance. Avoid ultrasonic cleaning or exposure to chlorinated solvents.
How is traceability ensured during calibration?
Each calibration event must be recorded with balance ID, environmental conditions, raw mass readings (n = 3), calculated volume, and deviation vs. tolerance limits—retained for minimum 5 years under GLP or 21 CFR Part 11-aligned documentation practices.