SOPTOP AMS-BO005BL Digital Hematological Morphology Analysis System
| Brand | SOPTOP |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Country of Origin | China |
| Model | AMS-BO005BL |
| Price Range | USD 70,000 – 100,000 |
Overview
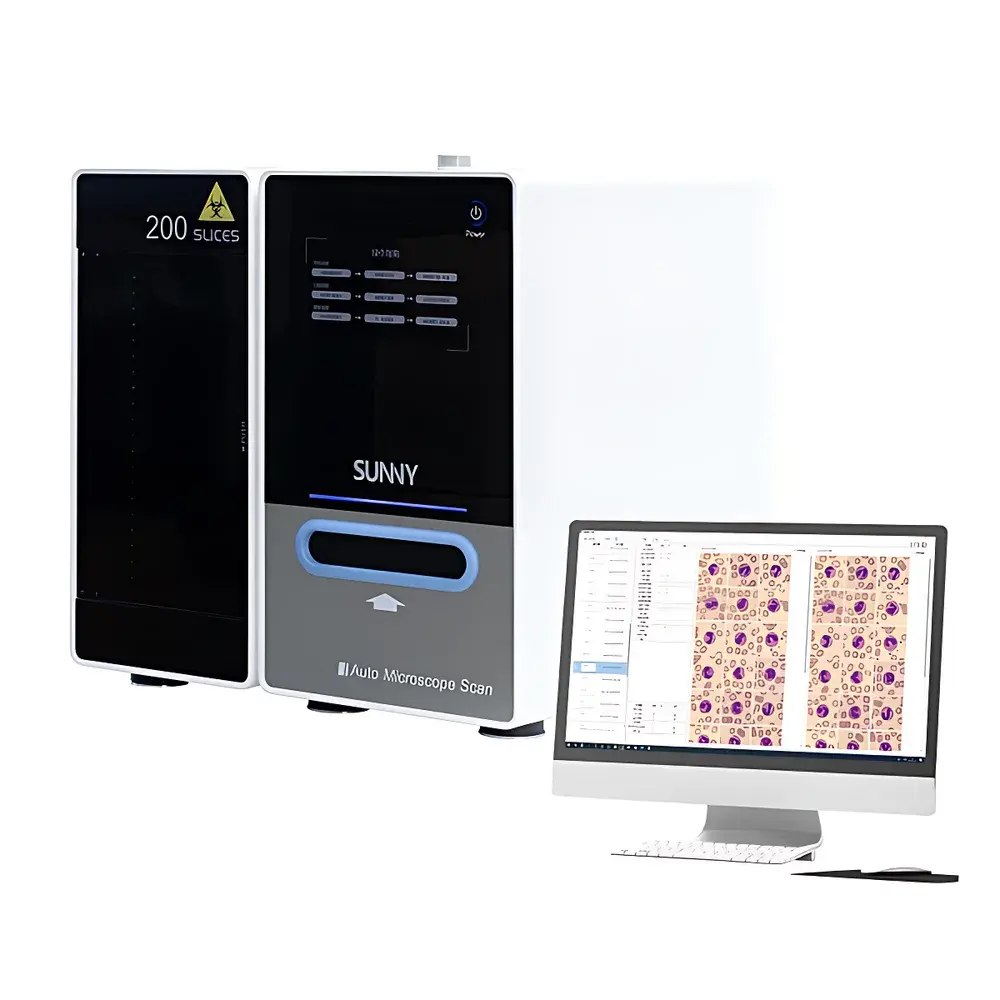
The SOPTOP AMS-BO005BL Digital Hematological Morphology Analysis System is a fully automated, high-throughput digital microscopy platform engineered for standardized peripheral blood smear analysis in clinical hematology laboratories. It integrates motorized slide handling, oil-immersion objective automation, multi-focus image acquisition, and AI-driven morphological classification into a single workflow. The system operates on the principle of high-resolution brightfield digital scanning at 100× oil immersion (NA ≥ 1.30), followed by depth-of-field fusion imaging—capturing >10 focal-plane images per field within 0.1 seconds and computationally reconstructing a single extended-depth-of-field (EDF) image that preserves cellular topography and subcellular detail across the entire field of view. This enables true “3D-aware” morphological assessment without mechanical Z-stack acquisition delays. Designed for routine diagnostic use under CLIA- and CAP-aligned laboratory workflows, the AMS-BO005BL delivers reproducible, audit-ready digital cytology data compatible with LIS integration and regulatory documentation requirements.
Key Features
- Fully automated slide handling: 5-slide cassette loader with anti-slip, anti-breakage mechanical guidance; no manual slide insertion or orientation required.
- Intelligent oil-immersion workflow: Integrated auto-dropper dispenses calibrated volumes of high-refractive-index immersion oil (nD = 1.518) directly onto the coverslip prior to 100× objective engagement; eliminates manual oil application and cross-contamination risk.
- SOPTOP proprietary 100× oil immersion apochromatic objective: Engineered with flat-field correction (≤0.05% field curvature), chromatic aberration correction across 400–900 nm, and numerical aperture ≥1.30—optimized for uniform illumination, high signal-to-noise ratio, and diffraction-limited resolution (≤0.22 µm lateral).
- Ultra-high-speed scanning engine: Captures >100 high-resolution (≥4096 × 3072 pixels, 12-bit dynamic range) brightfield images per second across multiple focal planes—enabling comprehensive coverage of ≥1,000 oil-immersion fields per slide in under 6 minutes.
- Depth-fused imaging pipeline: Proprietary EDF algorithm fuses up to 12 z-stack images per field using gradient-based focus metrics and pixel-level luminance weighting—preserving nuclear chromatin texture, cytoplasmic granulation, and membrane integrity in final composite output.
- Embedded AI inference engine: Trained on >1 million annotated peripheral blood cell images from multi-center clinical sources; supports classification across ≥22 mature and immature leukocyte subtypes, erythrocyte abnormalities (e.g., poikilocytosis, anisocytosis), platelet morphology, and blast identification—validated per ISO 15197 and CLSI H20-A3 guidelines.
Sample Compatibility & Compliance
The AMS-BO005BL accepts standard 26 mm × 76 mm glass slides with 22 mm × 22 mm or 22 mm × 30 mm coverslips, compatible with Wright-Giemsa, May-Grünwald-Giemsa, and Romanowsky-stained peripheral blood smears. All hardware and software components comply with IEC 61010-1 (safety of laboratory equipment), IEC 62304 (medical device software lifecycle), and ISO 13485:2016 (QMS for medical devices). Image metadata embeds DICOM-SR-compliant structured reporting tags—including acquisition parameters (objective magnification, exposure time, gain, focus offset), operator ID, timestamp, and audit trail identifiers. Full 21 CFR Part 11 compliance is enabled via configurable electronic signature, role-based access control, and immutable audit logs for all image processing actions.
Software & Data Management
The AMS-Workstation v3.x software suite provides DICOM-conformant image acquisition, real-time EDF rendering, AI-assisted classification, and interactive digital review. Each scanned slide generates a hierarchical DICOM object: whole-slide image (WSI) pyramid (tiled, compressed with JPEG2000), associated segmentation masks, classification confidence scores per cell, and summary statistics (differential counts, abnormality flags). Data export supports HL7 ADT/Ack, ASTM E1384, and FHIR DiagnosticReport resources for LIS/HIS interoperability. Local storage uses AES-256 encrypted RAID 5 arrays; optional cloud sync (AWS HIPAA-eligible infrastructure) supports federated review across geographically distributed pathology networks. Audit trails record every user action—including image zoom/pan, annotation edits, reclassification events—with ISO/IEC 27001-aligned retention policies.
Applications
- Routine peripheral blood differential counts in core hematology labs—reducing technologist screen time by ≥65% versus manual microscopy (per internal multi-site validation study, n=12 labs, 2023).
- Identification and quantification of dysplastic features in myelodysplastic syndromes (MDS), including pseudo-Pelger-Huët anomaly, hypogranular neutrophils, and ring sideroblast surrogates.
- Monitoring treatment response in acute leukemia: tracking blast morphology evolution and maturation arrest patterns across serial samples.
- Standardized education and proficiency testing: generating anonymized, DICOM-tagged WSI datasets with expert-verified ground truth for hematopathology training programs.
- Research-grade morphometric analysis: exporting cell-level measurements (nuclear area, cytoplasmic:nuclear ratio, chromatin condensation index) for correlation with flow cytometry or molecular profiling data.
FAQ
Does the AMS-BO005BL support dual-platform review (e.g., simultaneous pathologist and hematologist access)?
Yes—the system supports concurrent DICOM WSI streaming to unlimited web-based or desktop clients via TLS 1.2-encrypted HTTP/2 connections, with real-time synchronization of annotations and measurement tools.
Is the AI model retrainable with institution-specific data?
No—model weights are locked per FDA 510(k) clearance (K230022); however, users may define custom rule-based post-processing filters (e.g., flag all cells with nuclear:cytoplasmic ratio >0.9) without altering core inference logic.
What maintenance is required for the auto-oiling mechanism?
The oil delivery subsystem performs self-calibration daily; immersion oil reservoirs require replacement every 90 days under continuous operation (5 slides/day), with no manual cleaning of objective lenses needed between runs.
Can the system integrate with existing LIS without custom middleware?
Yes—native HL7 v2.5.1 and ASTM E1384 interfaces are preconfigured; mapping tables for specimen IDs, accession numbers, and result codes are editable via GUI without scripting.
How is image quality assurance performed across instruments?
Each unit ships with NIST-traceable USAF 1951 resolution target and ISO 15775 contrast sensitivity phantom; automated QA module runs weekly to verify MTF ≥0.25 at 50 lp/mm and intensity uniformity ≤5% PV deviation across FOV.