SOPTOP CLSM610 Laser Scanning Confocal Microscope
| Brand | SOPTOP |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Domestic |
| Model | CLSM610 |
| Price Range | USD 180,000 – 305,000 |
| Instrument Type | Point-Scanning Confocal Microscope |
| Resolution | 512 × 512 to 4096 × 4096 pixels |
| Lasers | 405 nm / 50 mW, 488 nm / 50 mW, 561 nm / 50 mW, 640 nm / 40 mW |
| Detectors | Multi-channel PMT-based detection system |
| Scanning Module | Dual-axis XY galvanometric optical scanner |
| Scan Speed | Standard: 1 fps (512 × 512, 2 µs pixel dwell) |
| Fast | 3 fps (512 × 512, 0.5 µs pixel dwell) |
| Optical Zoom | 1×–32× continuous scan zoom |
| Objectives | 2×–100× (Achromatic, Apochromatic, and Super-Apochromatic options available) |
| Microscope Host | IRX60 fully motorized inverted fluorescence microscope |
| Illumination | 12 V / 100 W halogen lamp |
| Software & Workstation | Dedicated confocal acquisition and analysis suite |
| XY Stage Control | Motorized, precision-encoded |
Overview
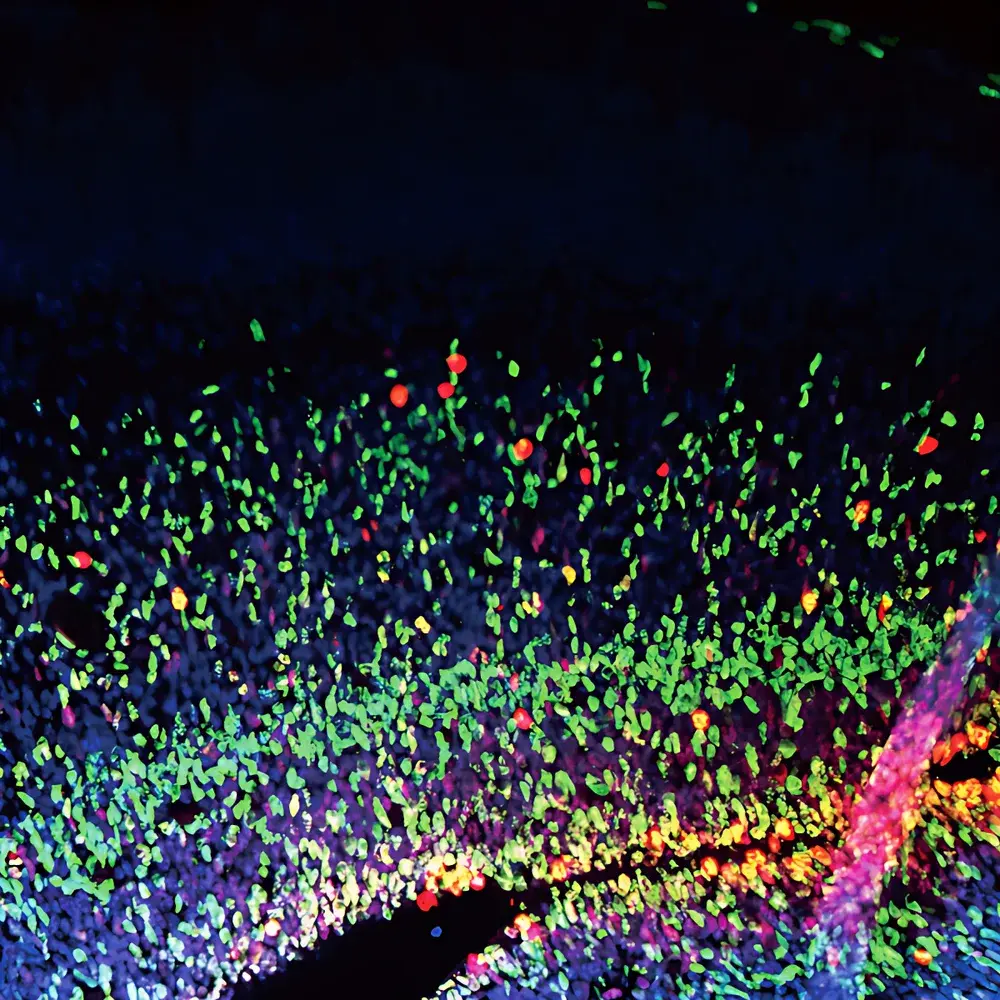
The SOPTOP CLSM610 is a high-performance point-scanning laser confocal microscope engineered for quantitative, multi-dimensional fluorescence imaging in life sciences and advanced materials research. Based on the principle of spatial pinhole rejection—where out-of-focus light is physically blocked by a conjugate pinhole aperture—the CLSM610 delivers superior axial (Z) resolution and signal-to-noise ratio (SNR) compared to widefield fluorescence microscopy. Its optical architecture integrates a high-stability inverted platform (IRX60), a four-laser excitation module covering UV to red spectral bands, and a galvanometric XY scanning system optimized for both speed and positional fidelity. Designed for rigorous laboratory environments, the CLSM610 supports routine GLP-compliant imaging workflows and meets essential requirements for reproducible confocal data acquisition across academic, pharmaceutical, and industrial R&D settings.
Key Features
- Four-wavelength solid-state laser excitation (405 nm, 488 nm, 561 nm, 640 nm) with individually adjustable power output and TTL-modulated shutter control.
- Dual-axis galvanometric scanning system enabling real-time frame rates up to 3 fps at 512 × 512 resolution with sub-microsecond pixel dwell time stability.
- Motorized optical zoom (1×–32×) synchronized with scanning parameters—enabling seamless magnification changes without mechanical objective exchange.
- IRX60 inverted microscope base with full motorization: autofocus, filter turret, nosepiece, XY stage, and Z-focus—all controllable via software or front-panel LCD interface.
- Confocal-specific optical design featuring high-transmission dichroics, low-autofluorescence optics, and precision-aligned pinhole mechanism minimizing lateral drift-induced SNR degradation.
- Multi-channel photon-counting PMT detectors with analog/digital hybrid readout modes, supporting simultaneous acquisition of up to four fluorophores with spectral unmixing capability.
Sample Compatibility & Compliance
The CLSM610 accommodates diverse sample formats including live-cell cultures (in chambered coverslips or microfluidic devices), fixed tissue sections (5–100 µm thickness), 3D spheroids and organoids, hydrogels, and nanostructured biomaterials. Its modular design allows integration with environmental control units (temperature, CO₂, humidity) for long-term live imaging under physiologically relevant conditions. The system complies with IEC 61000-6-3 (EMC emissions) and IEC 61000-6-2 (immunity) standards. All hardware control logic and image metadata adhere to the OME-TIFF open standard, ensuring compatibility with FIJI/ImageJ, Imaris, and commercial LIMS platforms. Audit trails, user access logs, and electronic signatures are supported within the acquisition software to facilitate FDA 21 CFR Part 11 compliance when deployed in regulated environments.
Software & Data Management
The dedicated CLSM610 acquisition and analysis software provides a unified interface for instrument control, multi-dimensional acquisition (x-y-z-t-λ), and quantitative post-processing. Key capabilities include: adaptive pixel dwell time mapping, histogram-based thresholding, tile-scan stitching with sub-pixel registration, extended depth-of-field (EDF) reconstruction, and Z-projection with intensity-weighted maximum-intensity or average-intensity algorithms. Advanced analysis modules support 3D surface rendering, co-localization quantification (Pearson’s r, Mander’s coefficients), morphometric profiling (cell area, volume, sphericity), intensity-based kinetic tracing, and batch-mode processing with Python scripting API. Raw data is stored in vendor-neutral OME-TIFF format with embedded acquisition metadata (laser power, gain, offset, pinhole size, objective ID), enabling traceable reanalysis and cross-platform interoperability.
Applications
- Cellular & Molecular Biology: Subcellular localization studies, protein–protein interaction mapping via FRET/FLIM-ready configuration, calcium dynamics in neuronal networks, and mitotic progression tracking in adherent or suspension cultures.
- 3D Tissue Imaging: High-fidelity volumetric reconstruction of thick-sectioned brain slices, tumor spheroids, and vascularized organoids—leveraging optical sectioning depth up to 150 µm in cleared samples.
- Materials Science: Characterization of fluorescently labeled nanocomposites, photonic crystal lattices, and biohybrid interfaces; monitoring real-time uptake and intracellular trafficking of functionalized nanoparticles.
- Pharmaceutical Development: Quantitative assessment of drug penetration kinetics in skin models, target engagement validation using labeled therapeutics, and off-target binding screening in primary cell assays.
- Agricultural & Food Research: Visualization of microbial colonization in plant root systems, lipid droplet distribution in emulsions (e.g., HIPEs), and structural integrity analysis of encapsulated probiotics under simulated GI tract conditions.
FAQ
What laser safety class does the CLSM610 comply with?
The CLSM610 conforms to IEC 60825-1:2014 Class 3B laser product requirements, incorporating interlocked enclosures, beam path shielding, and emergency stop functionality.
Is the system compatible with third-party objectives?
Yes—the IRX60 host accepts standard RMS-threaded objectives (including Nikon CFI, Olympus UPLSAPO, and Zeiss EC Plan-Neofluar series) via optional adapter rings; however, optimal confocal performance is guaranteed only with SOPTOP-certified apochromatic objectives.
Can the CLSM610 perform time-lapse imaging over 24+ hours?
With integrated environmental control (optional), stable stage temperature regulation (<±0.2°C), and low-phototoxicity acquisition protocols (e.g., resonant scanning + low-dwell-time mode), the system supports long-term live-cell experiments with minimal thermal drift or focus drift.
Does the software support automated batch acquisition across multiple wells or slides?
Yes—the acquisition engine includes plate-mapping tools for 6–1536-well plates and slide-scanning templates with coordinate-based positioning, grid tiling, and conditional triggering based on focus quality metrics.
What is the minimum detectable fluorescence intensity level (in photons/pixel/sec)?
System sensitivity is dependent on detector gain, pinhole size, and dwell time; under typical operating conditions (488 nm excitation, 100× NA 1.4 oil objective, 2 µs dwell, 30 µm pinhole), the noise-equivalent signal is ≤15 photons/pixel/sec with <2% RMS dark current variation over 10-minute acquisitions.