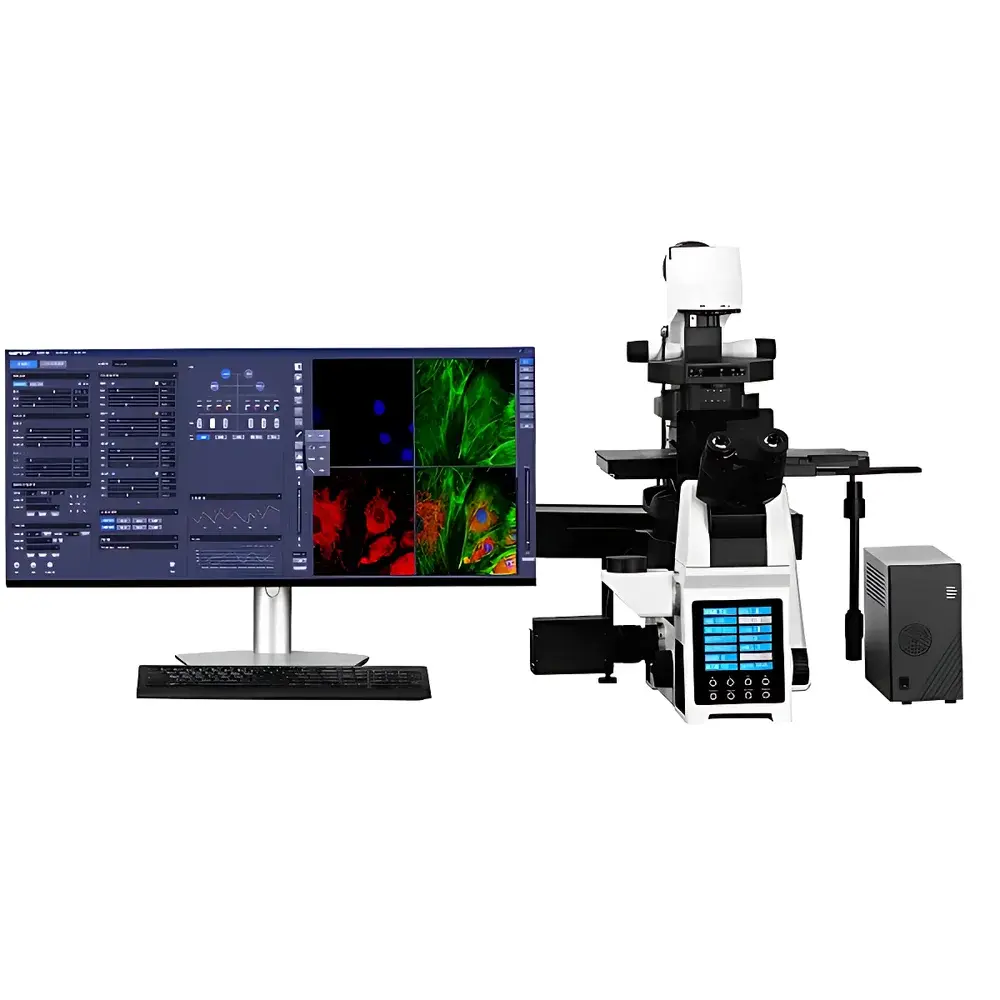
SOPTOP M-SIM6000 Structured Illumination Microscopy (SIM) System
| Brand | SOPTOP |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Origin | Domestic (China) |
| Model | M-SIM6000 |
| Pricing Range | USD 118,000 – 153,000 (FOB) |
| Technology | Structured Illumination Microscopy (SIM) |
| Lateral Resolution (XY) | 240 nm |
| Axial Resolution (Z) | 600 nm |
| Imaging Speed | 100 fps @ 2048 × 2048 |
| Eyepieces | High-eyepoint wide-field plan achromat PL10X/22 mm with adjustable diopter |
| Objectives | Long-working-distance plan semi-apochromat (4×, 10×, 20×, 40×, 60×) |
| Light Source | Independently controllable four-wavelength LED fluorescence illumination |
Overview
The SOPTOP M-SIM6000 Structured Illumination Microscopy (SIM) System is a research-grade super-resolution imaging platform engineered for high-fidelity, optically sectioned 3D fluorescence microscopy of thick biological specimens. Unlike conventional widefield or confocal systems, the M-SIM6000 employs deterministic patterned illumination—projecting precisely controlled sinusoidal light patterns onto the sample at multiple phase shifts and orientations—to computationally extract spatial frequency information beyond the classical Abbe diffraction limit. This optical super-resolution technique achieves a verified lateral resolution of 240 nm and axial resolution of 600 nm, enabling subcellular structural interrogation without requiring specialized fluorophores, single-molecule blinking, or phototoxic excitation conditions typical of STORM or STED. The system is optimized for live-cell compatibility, supporting rapid volumetric acquisition at up to 100 frames per second at full 2048 × 2048 pixel resolution—making it suitable for dynamic processes such as organelle trafficking, mitotic progression, and cytoskeletal remodeling.
Key Features
- Integrated structured illumination module with motorized grating translation and phase control for artifact-free pattern generation and precise optical sectioning.
- Full-electric motorized microscope platform with independent Z-stage, filter turret, shutter, and objective revolver—enabling reproducible multi-dimensional acquisition sequences (x, y, z, λ, t).
- Dual-path fluorescence optics: one path dedicated to structured illumination projection; the other for high-quantum-efficiency image detection—minimizing crosstalk and maximizing signal fidelity.
- Modular objective library including long-working-distance semi-apochromats (4×–60×) for cleared tissue or chambered samples, and high-NA oil-immersion super-apochromats (60×/100× oil) for maximal resolution in fixed or live adherent cells.
- Four-channel independently addressable LED light engine (typically 405/488/561/640 nm) with microsecond-level TTL synchronization, intensity linearity ±2%, and thermal stabilization for photometric consistency across time-lapse experiments.
Sample Compatibility & Compliance
The M-SIM6000 supports diverse specimen formats: standard glass slides, #1.5 coverslips, multi-well plates (including 96- and 384-well), and custom chambered coverslips compatible with environmental control (temperature, CO₂, humidity). Its optical sectioning capability enables robust imaging through scattering media—including 50–100 µm thick brain slices, organoids, and hydrogel-embedded spheroids—without physical sectioning. The system architecture conforms to ISO 10993-5 biocompatibility guidelines for optical components in contact with live samples. All firmware and software modules comply with GLP/GMP documentation requirements, including audit-trail-enabled user login, parameter logging, and timestamped metadata embedding (EXIF + custom JSON) per acquired stack. While not FDA-cleared as a diagnostic device, the platform meets essential design controls per IEC 61000-6-2 (EMC) and IEC 61000-6-4 (emissions) standards for laboratory instrumentation.
Software & Data Management
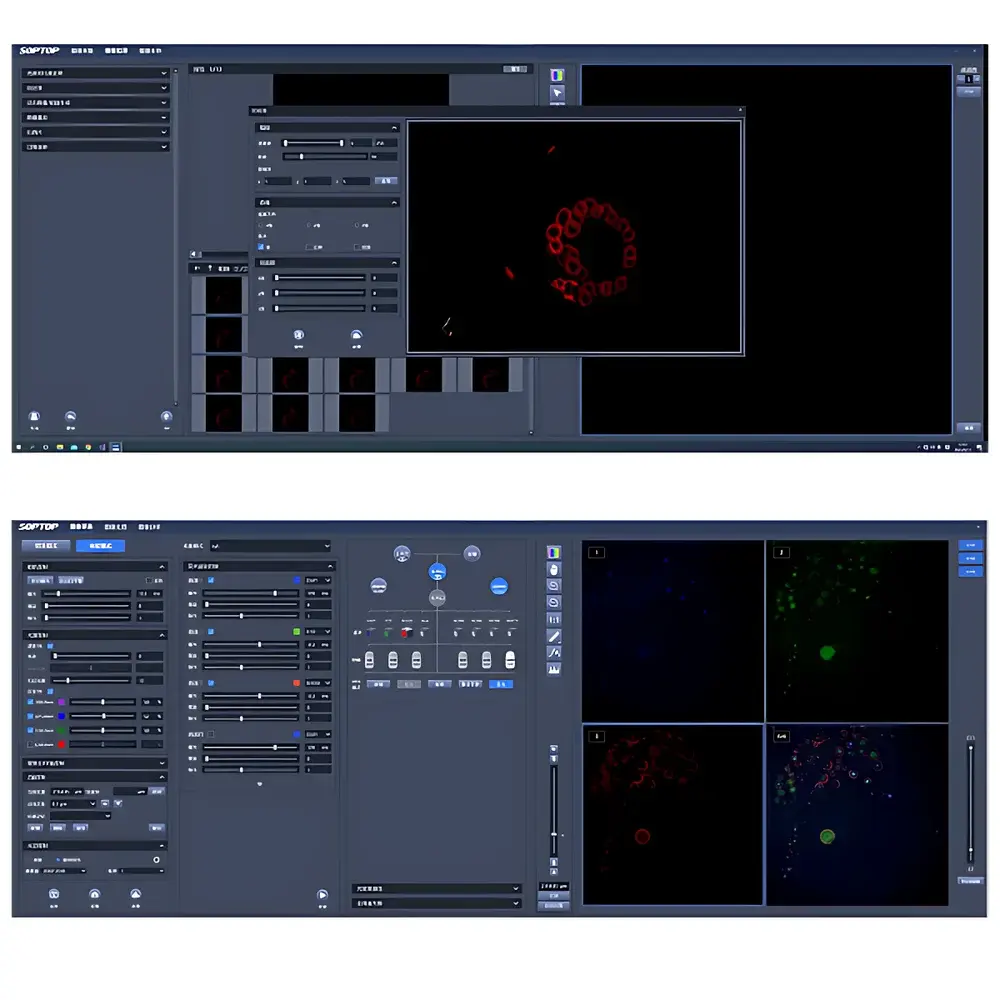
The SRF (Super-Resolution Framework) software suite provides end-to-end workflow integration—from acquisition to quantitative analysis. Acquisition modules support five-dimensional (x, y, z, λ, t) scheduling with hardware-triggered synchronization, drift correction via fiducial tracking, and real-time SIM reconstruction using GPU-accelerated deconvolution algorithms (Wiener-filter-based and constrained iterative solvers). Image processing includes isotropic 3D rendering (volume, maximum-intensity projection, orthogonal slicing), background subtraction (rolling-ball and morphological), adaptive contrast stretching, ROI-based time-series extraction, and batch-compatible stack alignment. Quantitative analysis tools implement ISO/IEC 17025-aligned measurement protocols: particle counting with size/shape filtering (circularity, aspect ratio, Feret diameter), co-localization analysis (Manders’ coefficients, Pearson correlation), subcellular distance mapping (e.g., nuclear envelope to centrosome), and temporal kinetics modeling (fluorescence recovery after photobleaching—FRAP—curve fitting with mono-/bi-exponential models).
Applications
The M-SIM6000 serves as a core imaging modality in academic and industrial life science laboratories where resolution, speed, and physiological relevance must coexist. In cell biology, it resolves microtubule lattice organization, nuclear pore complex distribution, and mitochondrial cristae dynamics in unperturbed cells. In immunology, it visualizes immune synapse architecture and antigen receptor clustering at synaptic nanodomains. In pharmacology, it tracks intracellular drug localization and target engagement kinetics across dose-response time courses. In hematology, it characterizes platelet granule heterogeneity and erythrocyte membrane fluctuations under shear stress. In materials science, it images polymer nanostructure morphology and biohybrid scaffold porosity at sub-250 nm scale—supporting correlative workflows with SEM or AFM.
FAQ
What is the effective optical sectioning thickness achievable with the M-SIM6000?
The system delivers an optical slice thickness of ≤1.2 µm (FWHM) in thick specimens (up to 100 µm), validated using fluorescent microsphere phantoms per ISO 19040-2.
Does the SRF software support third-party file formats for downstream analysis?
Yes—SRF exports TIFF (OME-TIFF compliant), HDF5, and N5 containers with embedded metadata; import supports LSM, ND2, CZI, and BFP formats.
Can the M-SIM6000 be integrated into automated screening platforms?
Yes—the system features standardized RS-232, USB 3.0, and Ethernet interfaces with documented API (Python/C++ SDK) for integration with robotic liquid handlers and stage controllers.
Is the SIM reconstruction algorithm open-source or proprietary?
The core reconstruction pipeline is proprietary but fully documented in the user manual; raw patterned image sequences are saved for optional external reconstruction using FIJI/SIMToolbox or custom MATLAB pipelines.
What maintenance protocols are recommended for long-term stability of the LED illumination system?
LED output is calibrated quarterly using NIST-traceable photodiode sensors; firmware auto-compensates for thermal drift during operation, and lifetime exceeds 20,000 hours at rated power.