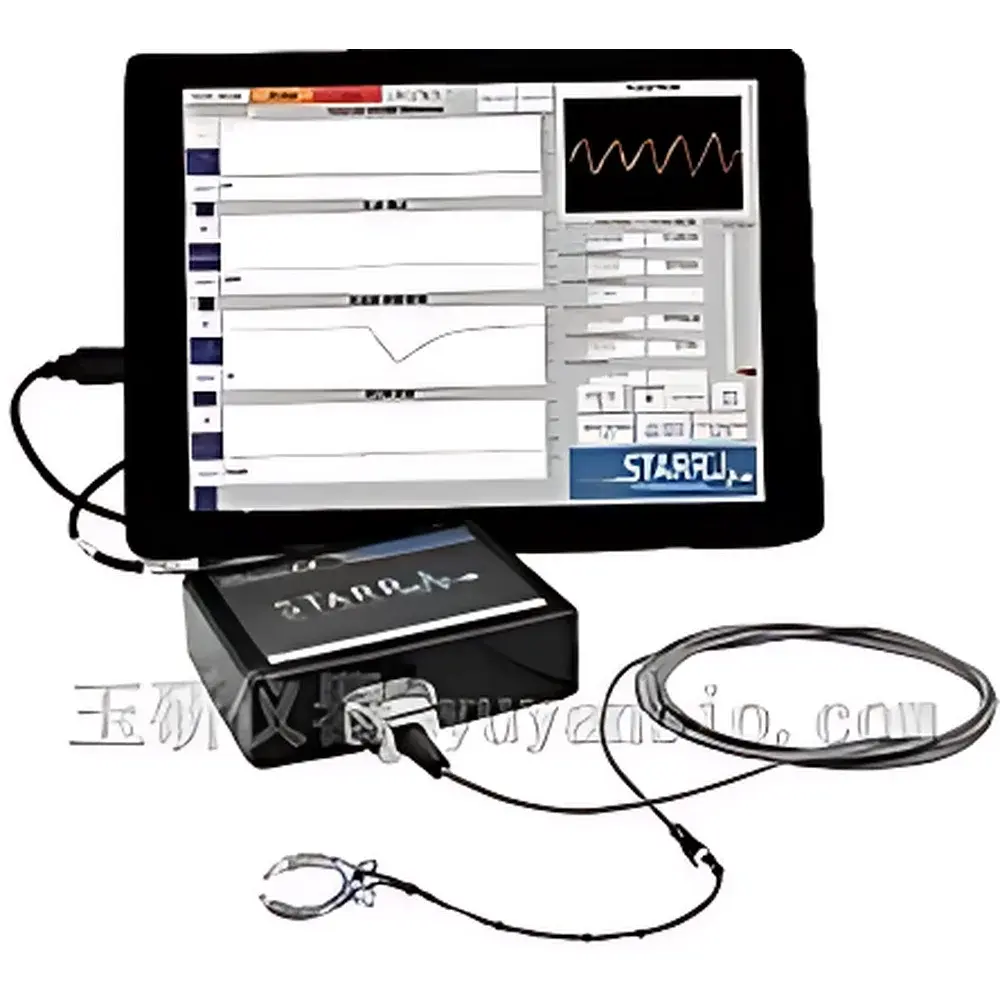
STARR MouseOx Plus Non-Invasive Pulse Oximeter for Small Animals
| Brand | STARR Instruments |
|---|---|
| Origin | USA |
| Manufacturer | STARR Instruments, Inc. |
| Model | MouseOx Plus |
| Device Type | Small Animal Physiological Monitor |
| Measurement Principles | Reflectance Pulse Oximetry (735 nm & 850 nm dual-wavelength LED photoplethysmography), Thermal Sensing (for temperature) |
| Regulatory Status | CE-marked, FDA 510(k)-cleared (K122924), ISO 13485-certified manufacturing |
| Compliance | Designed to support GLP-compliant preclinical studies |
Overview
The STARR MouseOx Plus is a research-grade, non-invasive physiological monitor engineered specifically for longitudinal and intraoperative monitoring of small laboratory mammals—including mice, rats, guinea pigs, and rabbits. It employs dual-wavelength reflectance pulse oximetry (735 nm and 850 nm LEDs) combined with high-sensitivity photodetectors to extract arterial oxygen saturation (SpO₂), pulse rate (PR), respiratory rate (RR), pulse amplitude (PA), and respiratory amplitude (RA) from a single, minimally perturbing sensor interface. Temperature is measured separately via a calibrated thermistor probe. Unlike clinical human oximeters optimized for finger or earlobe perfusion, the MouseOx Plus is optically and algorithmically tuned for low-perfusion, high-heart-rate rodent physiology—enabling reliable detection of subtle hemodynamic and ventilatory changes during anesthesia, pharmacological challenge, hypoxia modeling, or recovery-phase observation. Its real-time signal processing architecture supports dynamic physiological assessment without requiring surgical instrumentation or blood sampling, thereby reducing inter-animal variability and enhancing experimental reproducibility in compliance with the 3Rs principles.
Key Features
- Simultaneous acquisition of six core physiological parameters: SpO₂ (%), pulse rate (BPM), respiratory rate (breaths/min), pulse amplitude (arbitrary units proportional to peripheral perfusion), respiratory amplitude (arbitrary units reflecting thoracic/abdominal excursion), and core/skin temperature (°C)
- Dual-wavelength reflectance photoplethysmography optimized for rodent tissue optical properties and microvascular geometry
- Real-time display refresh: SpO₂ and pulse parameters updated every 0.72 s post-detection; respiratory metrics averaged over 10 breaths and reported every 1.7 s
- Validated accuracy: SpO₂ error ≤ ±1.5% across 0–100% range; pulse rate error ≤ ±2.4% across 90–900 BPM; RR accuracy validated against plethysmographic and capnometric references
- Modular sensor ecosystem: Dedicated probes for conscious ambulatory monitoring (neck-mounted clip), anesthetized/surgical positioning (footpad, thigh, tail), and MRI-compatible environments (non-ferromagnetic, RF-shielded design)
- Multi-animal scalability: Optional 4-channel or 8-channel adapter enables synchronized parallel monitoring of cohorts without cross-talk or timing drift
- Analog output (0–5 V DC) for integration with third-party DAQ systems, behavioral rigs, or closed-loop anesthesia controllers
Sample Compatibility & Compliance
The MouseOx Plus supports consistent signal acquisition across developmental stages (neonatal through adult) and physiological states (awake, sedated, surgically prepared). Probe selection is empirically matched to animal size, fur density, skin pigmentation, and motion profile—ensuring stable signal-to-noise ratio under both restrained and freely moving conditions. All hardware and firmware comply with IEC 61000-6-3 (EMC emissions) and IEC 61000-6-2 (immunity). The system meets ISO 14155:2020 requirements for investigational medical device use in preclinical safety pharmacology studies. Data integrity features—including timestamped binary logging, checksum-verified file export (.csv, .mat), and optional audit-trail-enabled software—facilitate alignment with FDA 21 CFR Part 11, EU Annex 11, and OECD GLP principles for regulated toxicology and PK/PD investigations.
Software & Data Management
The bundled MouseOx Software (v5.x) provides real-time waveform visualization (PPG, respiration envelope), configurable alarm thresholds, automated event tagging (e.g., apnea detection, desaturation episodes), and batch analysis tools for circadian rhythm profiling or dose-response quantification. Raw data streams are recorded at 100 Hz with sub-millisecond time synchronization across channels. Export formats include ASCII-delimited tables for statistical packages (R, Python pandas), MATLAB-compatible .mat files for custom algorithm development, and XML metadata containers compliant with MIAME/MINSEQE standards. Optional integration with LabArchives ELN or Benchling enables structured annotation, version-controlled protocol linking, and automated report generation for regulatory submissions.
Applications
- Intraoperative monitoring during survival surgery to maintain optimal anesthetic depth and prevent hypoxic injury
- Cardiopulmonary phenotyping in genetically modified mouse models (e.g., heart failure, pulmonary hypertension, COPD)
- Respiratory challenge studies: hypercapnia, hypoxia, bronchoconstrictor provocation
- Neurovascular coupling assessment during functional imaging or optogenetic stimulation
- Toxicology endpoints: early detection of respiratory depression induced by CNS-active compounds
- Longitudinal wellness tracking in aging or chronic disease models (e.g., diabetes, sepsis, cancer cachexia)
- Validation of non-invasive biomarkers against gold-standard invasive measurements (arterial blood gas, direct cardiac output)
FAQ
Is the MouseOx Plus suitable for neonatal mice or pups?
Yes—dedicated neonatal probes with reduced optical path length and adaptive gain control enable stable SpO₂ and PR acquisition in P0–P14 mice, provided ambient light is minimized and thermal regulation is maintained.
Can it be used inside MRI scanners?
Yes—the MRI-safe probe option uses non-ferromagnetic materials and fiber-optic signal transmission to eliminate RF interference and ensure subject safety at field strengths up to 9.4 T.
Does the system support automated data export to institutional databases?
Yes—via TCP/IP socket interface or scheduled file drop to network shares, with configurable metadata headers and ISO 8601 timestamps.
How is calibration performed?
No user calibration is required. Factory calibration is traceable to NIST standards and verified per ANSI/AAMI SP10 and ISO 80601-2-61. Re-validation is recommended annually or after sensor replacement.
What training and technical support is available?
STARR offers on-site installation verification, application-specific protocol optimization workshops, and remote troubleshooting via secure screen-sharing—backed by a global network of certified service engineers.