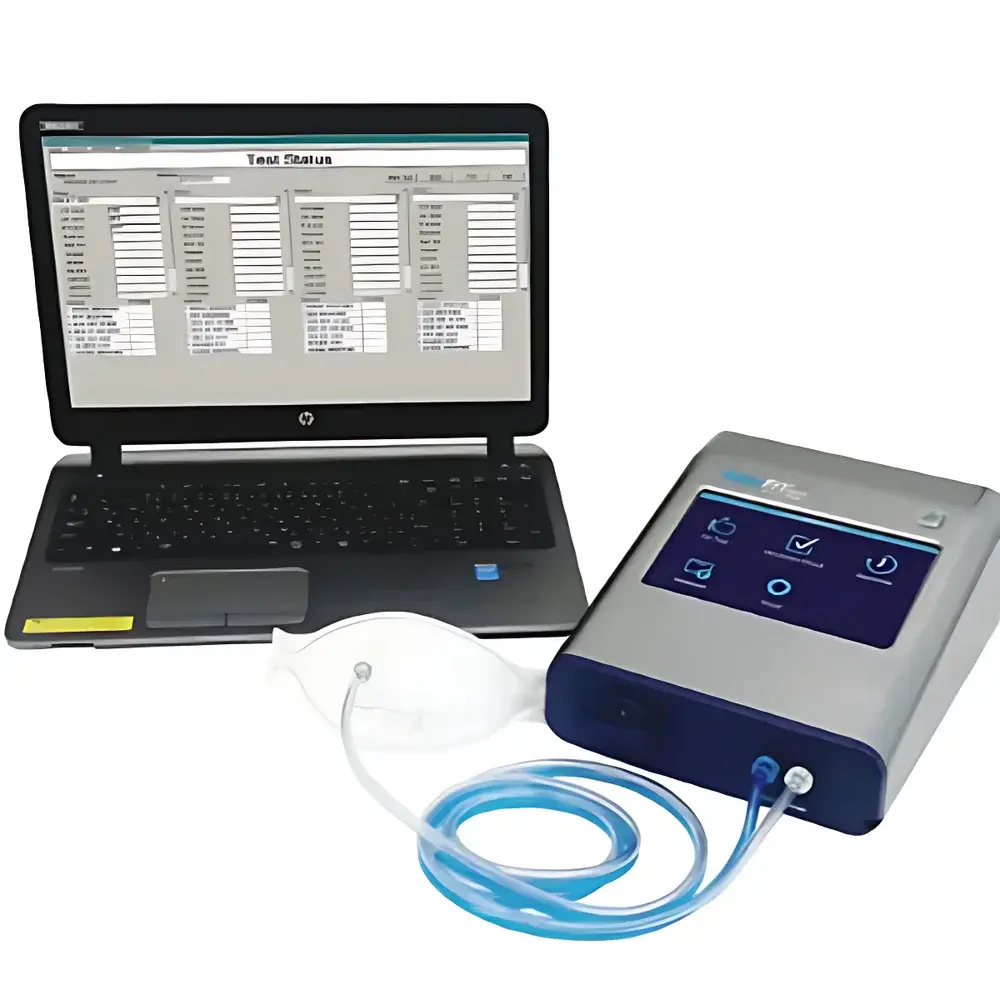
Starwood 3000-0C Respirator Fit Testing System
| Brand | Starwood |
|---|---|
| Model | 3000-0C |
| Detection Principle | Condensation Particle Counter (CPC) |
| Particle Size Range | 0.02–1.0 µm |
| Concentration Range | 0–100,000 particles/cm³ |
| Sample Flow Rate | 100 cm³/min |
| Total Flow Rate | 700 cm³/min |
| Fit Factor Calculation | Direct Cout/Cin Ratio |
| Challenge Agent | Isopropanol (≥99.5%) or Ethanol |
| Display | 7-inch True Color Touchscreen |
| Operating Temperature | 10–35 °C |
| Relative Humidity | 20–85% RH (non-condensing) |
| Power Supply | AC 110–240 V, 50/60 Hz |
| Dimensions | 208 × 117 × 262 mm (W×H×D) |
| Weight | 2.3 kg |
| Compliance Standards | OSHA 1910.134, CSA Z94.4, ANSI/AIHA Z88.10, UK HSE INDG479, GB 19083–2010, GB 2626–2019, GB/T 32610–2016 |
| Connectivity | USB, Ethernet, Wi-Fi |
| Software Compatibility | Windows 7/8/10, .NET Framework 3.5 |
| Data Export Format | Microsoft Excel |
| Storage Media | USB flash drive |
| Language Options | English, Chinese, Japanese, French, Spanish, Portuguese |
Overview
The Starwood 3000-0C Respirator Fit Testing System is a portable, quantitative fit testing instrument engineered for precision measurement of respirator facepiece seal integrity in occupational and clinical environments. It employs a calibrated condensation particle counter (CPC) detection principle to quantify ambient and in-mask aerosol concentrations in real time, enabling direct calculation of the fit factor (Cout/Cin). Unlike qualitative methods relying on subjective sensory response, this system delivers objective, numerical pass/fail determinations traceable to internationally recognized performance thresholds. Its CPC-based architecture ensures high sensitivity across the critical ultrafine particle range (0.02–1.0 µm), where most airborne pathogens and industrial nanoparticles reside—making it especially suitable for evaluating high-efficiency filtering facepieces (e.g., N95, FFP2, KN95) and powered air-purifying respirators (PAPRs).
Key Features
- True-color 7-inch touchscreen interface with intuitive navigation and six-language support (English, Chinese, Japanese, French, Spanish, Portuguese) for global deployment.
- Integrated challenge agent delivery using ≥99.5% isopropanol or ethanol—no external nebulizer or compressed air source required.
- Dual-flow design: 100 cm³/min sampling flow for precise particle counting and 700 cm³/min total system flow to maintain stable breathing resistance during testing.
- Compact form factor (208 × 117 × 262 mm) and lightweight construction (2.3 kg) enable field use in hospitals, manufacturing facilities, fire stations, and regulatory laboratories.
- Multi-unit network capability: A single Windows PC running the dedicated fit test software can concurrently control up to four 3000-0C units—ideal for high-throughput worker screening programs.
- Robust environmental tolerance: Operational within 10–35 °C and 20–85% RH (non-condensing), with storage capability down to –20 °C.
Sample Compatibility & Compliance
The 3000-0C supports comprehensive compatibility across respirator classes, including disposable filtering facepiece respirators (N95, P2, FFP3), reusable half- and full-face elastomeric respirators, PAPRs, SCBAs, and chemical cartridge respirators. Its built-in test protocols align with major international regulatory frameworks: OSHA 1910.134 Appendix A (quantitative fit test requirements), CSA Z94.4–18, ANSI/AIHA Z88.10–19, UK HSE INDG479, and key Chinese national standards GB 19083–2010 (medical protective masks), GB 2626–2019 (particulate-filtering respirators), and GB/T 32610–2016 (daily protective masks). All test sequences are preconfigured to enforce mandatory pass criteria (e.g., minimum fit factor of 100 for half-masks, 500 for full-facepieces), ensuring audit-ready compliance in GLP and GMP-aligned workflows.
Software & Data Management
The proprietary Starwood FitTest Suite operates on Windows platforms (7/8/10) under .NET Framework 3.5 and provides full lifecycle management of fit test records. Each session captures subject ID, respirator model, test protocol, environmental conditions, real-time particle concentration curves, calculated fit factors per exercise, and final pass/fail status. Data export is natively supported in Microsoft Excel (.xlsx) format for integration into enterprise HRIS or occupational health databases. All test logs include embedded timestamps, operator ID, and instrument serial number—meeting foundational requirements for FDA 21 CFR Part 11–compliant audit trails when deployed in regulated healthcare settings. Raw data and reports may be stored directly onto removable USB drives; optional Ethernet/Wi-Fi connectivity enables centralized logging in networked laboratory infrastructures.
Applications
- Annual or periodic respirator fit verification for healthcare workers handling infectious aerosols (e.g., TB, influenza, SARS-CoV-2).
- Pre-deployment screening of first responders wearing SCBA or CBRN respirators.
- Validation of respirator selection and user training effectiveness in manufacturing and construction sectors.
- Third-party conformity assessment by accredited testing laboratories issuing CNAS or ILAC-MRA–recognized certificates.
- Research applications investigating facial anthropometry–respirator seal relationships or evaluating novel mask materials under controlled challenge conditions.
FAQ
What particle size range does the 3000-0C detect, and why is that relevant?
The system detects particles from 0.02 to 1.0 µm using condensation particle counting—a range encompassing the most penetrating particle size (MPPS) for HEPA and N95-grade filters. This ensures worst-case evaluation of respirator leakage pathways.
Can the 3000-0C perform both qualitative and quantitative fit tests?
No—it is strictly a quantitative instrument. It does not support saccharin, Bitrex®, or irritant smoke qualitative protocols. Its output is exclusively numeric fit factor data.
Is calibration traceable to national standards?
Yes. The CPC sensor is factory-calibrated against NIST-traceable aerosol generators and includes an integrated zero-count filter for routine baseline verification prior to each test series.
Does the system require consumables beyond isopropanol?
Only the supplied isopropanol (≥99.5%), disposable sampling tubes, and optional respirator-specific test kits (e.g., for 3M, Honeywell, MSA models) are required. No replacement optics or detector components are needed under normal operation.
How is data security maintained during networked operation?
When connected via Ethernet or Wi-Fi, communication uses TCP/IP with configurable firewall rules; no cloud upload or remote access is enabled by default. Local data residency is preserved unless explicitly exported by authorized personnel.